
Concept explainers
Locate the isoprene units in each compound.
a.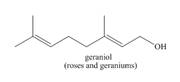 c.
c. 
b. 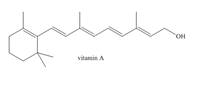 d.
d. 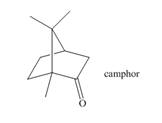
(a)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
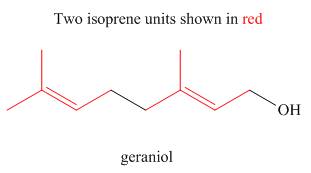
The isoprene units in the given terpene (geraniol) are located as,
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.
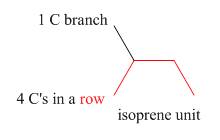
Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (geraniol) are located as,

Figure 2
The isoprene units in the given terpene (geraniol) are located in Figure 2.
(b)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
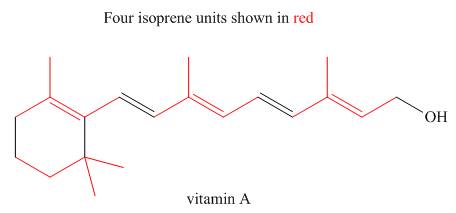
The isoprene units in the given terpene (vitamin A) are located as,
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.

Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (vitamin A) are located as,

Figure 3
The isoprene units in the given terpene (vitamin A) are located in Figure 3.
(c)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
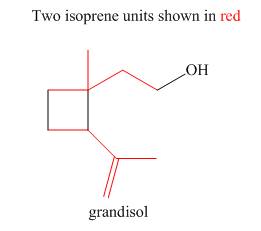
The isoprene units in the given terpene (grandisol) are located as,
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.

Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (grandisol) are located as,

Figure 4
The isoprene units in the given terpene (grandisol) are located in Figure 4.
(d)
Interpretation: The isoprene units in the given compound are to be located.
Concept introduction: An isoprene unit possesses following properties.
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
Answer to Problem 30.11P
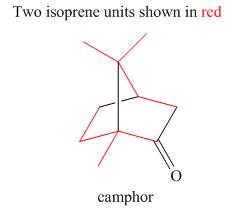
The isoprene units in the given terpene (camphor) are located as,
Explanation of Solution
Terpenes are composed of repeating five-carbon units. These units are called isoprene units.

Figure 1
An isoprene unit possesses following properties.
➢ It involves one branched carbon and
➢ It may either consist of
➢ It is always connected by one or more
➢ Each isoprene unit is consist of
➢ While locating an isoprene unit, the presence of heteroatoms is ignored.
The isoprene units in the given terpene (camphor) are located as,

Figure 5
The isoprene units in the given terpene (camphor) are located in Figure 5.
Want to see more full solutions like this?
Chapter 30 Solutions
Organic Chemistry
- A mixture of C7H12O2, C9H9OCl, biphenyl and acetone was put together in a gas chromatography tube. Please decide from the GC resutls which correspond to the peak for C7,C9 and biphenyl and explain the reasoning based on GC results. Eliminate unnecessary peaks from Gas Chromatography results.arrow_forwardIs the molecule chiral, meso, or achiral? CI .CH3 H₂C CIarrow_forwardPLEASE HELP ! URGENT!arrow_forward
- Identify priority of the substituents: CH3arrow_forwardHow many chiral carbons are in the molecule? OH F CI Brarrow_forwardA mixture of three compounds Phen-A, Acet-B and Rin-C was analyzed using TLC with 1:9 ethanol: hexane as the mobile phase. The TLC plate showed three spots of R, 0.1 and 0.2 and 0.3. Which of the three compounds (Phen-A; Acet-B or Rin-C) would have the highest (Blank 1), middle (Blank 2) and lowest (Blank 3) spot respectively? 0 CH: 0 CH, 0 H.C OH H.CN OH Acet-B Rin-C phen-A A A <arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
