
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 58P
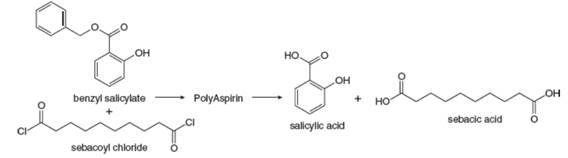
Researchers at Rutgers University have developed biocompatible
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used hand raiting and don't used Ai solution
Determine the number of protons (p), neutrons (n), or electrons (e) in each of thefollowing:
using dimensional analysis convert 15.28 lb/gallon to mg/mL
Chapter 28 Solutions
Organic Chemistry (6th Edition)
Ch. 28.1 - Prob. 1PCh. 28.2 - Prob. 2PCh. 28.2 - Prob. 3PCh. 28.2 - Draw the mechanism for the radical polymerization...Ch. 28.2 - Prob. 8PCh. 28.2 - Prob. 9PCh. 28.2 - Prob. 10PCh. 28.3 - Problem 30.12
What polymer is formed by anionic...Ch. 28.5 - Prob. 12PCh. 28.5 - Prob. 13P
Ch. 28.6 - Problem 30.15
What polyamide is formed from each...Ch. 28.6 - Prob. 19PCh. 28.7 - Prob. 20PCh. 28.8 - Prob. 21PCh. 28.9 - Prob. 22PCh. 28.9 - Prob. 23PCh. 28 - Prob. 24PCh. 28 - Prob. 25PCh. 28 - 30.30 Draw each polymer in Problem 30.29 using the...Ch. 28 - Prob. 44PCh. 28 - 30.49 Draw the products of each reaction.
a. e....Ch. 28 - Prob. 56PCh. 28 - 30.56 Compound A is a novel poly (ester amide)...Ch. 28 - 30.57 Researchers at Rutgers University have...Ch. 28 - 30.58 Melmac, a thermosetting polymer formed from...Ch. 28 - 30.59 Although chain branching in radical...Ch. 28 - Prob. 61P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- using dimensional analysis convert 0.00685 km to micrometersarrow_forwardWhat are the major products of the following reaction? Draw all the major products. If there are no major products, then there is no reaction that will take place. Use wedge and dash bonds when necessary.arrow_forwardcalculation based on mole-mole relationshiparrow_forward
- An aquarium measures 175 cm by 225 cm by 151 cm. Calculate the volume of theaquarium in yards3arrow_forwardThe density of sulfuric acid is 0.875 g/cm3. If a procedure needed 4.00 mL of sulfuricacid, how many grams would you measure out?arrow_forwardQuestion Suggest a mechanism for the following reactions. Each will require multiple types of concerted pericyclic reactions (cycloaddition, electrocyclic, and sigmatropic. Classify each reaction type. CN a. NC 180 °Carrow_forward
- Don't used Ai solution and don't used hand raitingarrow_forwardQ2: Ranking Acidity a) Rank the labeled protons in the following molecule in order of increasing pKa. Briefly explain the ranking. Use Table 2.2 as reference. Ha Нь HC H-N Ha OHe b) Atenolol is a drug used to treat high blood pressure. Which of the indicated N-H bonds is more acidic? Explain. (Hint: use resonance structures to help) Name the functional groups on atenolol. H H-N atenolol Ν H-N OH Нarrow_forwardAnswer d, e, and farrow_forward
- If the rotational constant of a molecule is B = 120 cm-1, it can be stated that the transition from 2←1:a) gives rise to a line at 120 cm-1b) is a forbidden transitionc) gives rise to a line at 240 cm-1d) gives rise to a line at 480 cm-1arrow_forwardBriefly indicate the coordination forms of B and Si in borates and silicates, respectively.arrow_forwardCan you please draw out the Lewis structure for these two formulasarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Molecular spectroscopy; Author: Vidya-mitra;https://www.youtube.com/watch?v=G6HjLIWvCQo;License: Standard YouTube License, CC-BY