
CNCT ORG CHEM 6 2020
6th Edition
ISBN: 9781266807244
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 27, Problem 65P
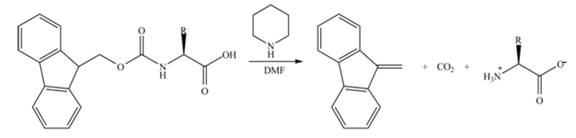
Draw the mechanism for the reaction that removes an Fmoc group from an amino acid under the following conditions:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Answer d, e, and f
If the rotational constant of a molecule is B = 120 cm-1, it can be stated that the transition from 2←1:a) gives rise to a line at 120 cm-1b) is a forbidden transitionc) gives rise to a line at 240 cm-1d) gives rise to a line at 480 cm-1
Briefly indicate the coordination forms of B and Si in borates and silicates, respectively.
Chapter 27 Solutions
CNCT ORG CHEM 6 2020
Ch. 27.1 - Prob. 1PCh. 27.1 - Problem 29.2
What form exists at the isoelectric...Ch. 27.1 - Problem 29.3
Explain why the of the group of an...Ch. 27.1 - Prob. 4PCh. 27.2 - Problem 29.5
What -halo carbonyl compound is...Ch. 27.2 - Problem 29.6
The enolate derived from diethyl...Ch. 27.2 - Problem 29.7
What amino acid is formed when is...Ch. 27.2 - Problem 29.8
What aldehyde is needed to synthesize...Ch. 27.2 - Problem 29.9
Draw the products of each...Ch. 27.3 - Prob. 10P
Ch. 27.3 - Prob. 11PCh. 27.3 - Prob. 12PCh. 27.4 - Problem 29.13
What alkene is needed to synthesize...Ch. 27.5 - Problem 29.14
Draw the structure of each peptide....Ch. 27.5 - Problem 29.15
Name each peptide using both the...Ch. 27 - Draw the product formed when the following amino...Ch. 27 - With reference to the following peptide: a...Ch. 27 - Prob. 31PCh. 27 - Histidine is classified as a basic amino acid...Ch. 27 - Tryptophan is not classified as a basic amino acid...Ch. 27 - What is the structure of each amino acid at its...Ch. 27 - What is the predominant form of each of the...Ch. 27 - 29.37 What is the predominant form of each of the...Ch. 27 - a. Draw the structure of the tripeptide A–A–A, and...Ch. 27 - 29.39 Draw the organic products formed in each...Ch. 27 - 29.40 What alkyl halide is needed to synthesize...Ch. 27 - Prob. 50PCh. 27 - Draw the structure for each peptide: (a) Phe–Ala;...Ch. 27 - 29.52 For the tetrapeptide Asp–Arg–Val–Tyr:
a....Ch. 27 - Prob. 53PCh. 27 - Prob. 54PCh. 27 - 29.55 Draw the amino acids and peptide fragments...Ch. 27 - Prob. 56PCh. 27 - Prob. 57PCh. 27 - Prob. 58PCh. 27 - 29.59 An octapeptide contains the following amino...Ch. 27 - 29.60 Draw the organic products formed in each...Ch. 27 - 29.65 Draw the mechanism for the reaction that...Ch. 27 - 29.66 Which of the following amino acids are...Ch. 27 - 29.67 After the peptide chain of collagen has been...Ch. 27 - Prob. 68PCh. 27 - Prob. 69PCh. 27 - 29.70 The anti-obesity drug orlistat works by...Ch. 27 - Prob. 71P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Can you please draw out the Lewis structure for these two formulasarrow_forwardIn a rotational Raman spectrum of a diatomic molecule it is correct to say that:a) anti-Stokes lines occur at frequencies higher than the excitatory oneb) Stokes lines occur at frequencies higher than the excitatory onec) Rayleigh scattering is not observedd) Rayleigh scattering corresponds to delta J = 0arrow_forwardOf the molecules: H2, N2, HCl, CO2, indicate which ones can give Raman vibration-rotation spectra:a) H2, N2 and HClb) H2, N2, HCl and CO2c) H2 and N2d) all of themarrow_forward
- Can you please help me with drawing the Lewis structure of each molecular formula?I truly appreciate you!arrow_forwardCan you please help me with drawing the Lewis structure of each molecular formula?I truly appreciate you!arrow_forwardCan you please help me with drawing the Lewis structure of each molecular formula?I truly appreciate you!arrow_forward
- Can you please help me with drawing the Lewis structure of each molecular formula?I truly appreciate you!arrow_forwardPlease draw and explainarrow_forwardDescribe each highlighted bond in terms of the overlap of atomic orbitals. (a) Н Н H H [References] HIC H H C H H-C-CC-N: H σ character n character (b) HIC H H H H-C-C-C HIC H Н H O-H σ character n character Submit Answer Try Another Version 3 item attempts remainingarrow_forward
- 11 Naming and drawing alcohols Write the systematic (IUPAC) name for each of the following organic molecules: structure OH HO OH Explanation Check name ☐arrow_forwardwhat is the drawn mechanism for diethyl carbonate and 4 - bromo - N, N -dimethylaniline to create crystal violet?arrow_forwardWhich of the following compounds are constitutional isomers of each other? I and II O II and III O III and IV OI and IV O II and IV CI H CI H CI H H CI H-C-C-CI C-C-C-CI H-C-C-CI H-C-C-CI H CI Ĥ ĆI A A Ĥ ĆI || IVarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY