
Organic Chemistry, Binder Ready Version
2nd Edition
ISBN: 9781118454312
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 26.8, Problem 4LTS
Interpretation Introduction
Interpretation:
Isoprene units in camphor need to be identified.
Concept introduction:
Terpenes are naturally occurring compounds which share a common feature. All terpenes are thought to be assembled from isoprene units having five carbon atoms. So the structure of terpenes has the carbon atoms in multiples of five only.
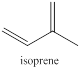
The isoprene units are identified by following few steps namely,
- Identify the number of isoprene units
- Look for methyl group
- Trial and error to find isoprene unit
To identify: the isoprene units in camphor
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the starting material in the following reaction. Click the "draw structure" button to launch the
drawing utility.
draw structure ...
[1] 0 3
C10H18
[2] CH3SCH3
H
In an equilibrium mixture of the formation of ammonia from nitrogen and hydrogen, it is found that
PNH3 = 0.147 atm, PN2 = 1.41 atm and Pн2 = 6.00 atm. Evaluate Kp and Kc at 500 °C.
2 NH3 (g) N2 (g) + 3 H₂ (g)
K₂ = (PN2)(PH2)³ = (1.41) (6.00)³ = 1.41 x 104
What alkene or alkyne yields the following products after oxidative cleavage with ozone? Click the
"draw structure" button to launch the drawing utility.
and two equivalents of CH2=O
draw structure ...
Chapter 26 Solutions
Organic Chemistry, Binder Ready Version
Ch. 26.2 - Prob. 1CCCh. 26.2 - Prob. 2CCCh. 26.3 - Prob. 1LTSCh. 26.3 - Prob. 3PTSCh. 26.3 - Prob. 4ATSCh. 26.3 - Prob. 5ATSCh. 26.3 - Prob. 6ATSCh. 26.4 - Prob. 7CCCh. 26.4 - Prob. 8CCCh. 26.4 - Prob. 2LTS
Ch. 26.4 - Prob. 9PTSCh. 26.4 - Prob. 10ATSCh. 26.4 - Prob. 11ATSCh. 26.4 - Prob. 3LTSCh. 26.4 - Prob. 12PTSCh. 26.4 - Prob. 13LTSCh. 26.4 - Prob. 14LTSCh. 26.5 - Prob. 15CCCh. 26.5 - Prob. 16CCCh. 26.5 - Prob. 17CCCh. 26.5 - Prob. 18CCCh. 26.5 - Prob. 19CCCh. 26.6 - Prob. 20CCCh. 26.6 - Prob. 21CCCh. 26.6 - Prob. 22CCCh. 26.6 - Prob. 23CCCh. 26.7 - Prob. 24CCCh. 26.8 - Prob. 4LTSCh. 26.8 - Prob. 25PTSCh. 26.8 - Prob. 26ATSCh. 26.8 - Prob. 27CCCh. 26.8 - Prob. 28CCCh. 26 - Prob. 29PPCh. 26 - Prob. 30PPCh. 26 - Prob. 32PPCh. 26 - Prob. 33PPCh. 26 - Prob. 34PPCh. 26 - Prob. 35PPCh. 26 - Prob. 36PPCh. 26 - Prob. 37PPCh. 26 - Prob. 38PPCh. 26 - Prob. 39PPCh. 26 - Prob. 40PPCh. 26 - Prob. 41PPCh. 26 - Prob. 42PPCh. 26 - Prob. 43PPCh. 26 - Prob. 44PPCh. 26 - Prob. 45PPCh. 26 - Prob. 46PPCh. 26 - Prob. 47PPCh. 26 - Prob. 48PPCh. 26 - Prob. 49PPCh. 26 - Prob. 50PPCh. 26 - Prob. 51IPCh. 26 - Prob. 52IPCh. 26 - Prob. 53IPCh. 26 - Prob. 54IPCh. 26 - Prob. 55IP
Knowledge Booster
Similar questions
- H-Br Energy 1) Draw the step-by-step mechanism by which 3-methylbut-1-ene is converted into 2-bromo-2-methylbutane. 2) Sketch a reaction coordinate diagram that shows how the internal energy (Y- axis) of the reacting species change from reactants to intermediate(s) to product. Brarrow_forward2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). C5H10 H-CI CH2Cl2 CIarrow_forwardDraw the products of the stronger acid protonating the other reactant. དའི་སྐད”“ H3C OH H3C CH CH3 KEq Product acid Product basearrow_forward
- Draw the products of the stronger acid protonating the other reactant. H3C NH2 NH2 KEq H3C-CH₂ 1. Product acid Product basearrow_forwardWhat alkene or alkyne yields the following products after oxidative cleavage with ozone? Click the "draw structure" button to launch the drawing utility. draw structure ... andarrow_forwardDraw the products of the stronger acid protonating the other reactant. H3C-C=C-4 NH2 KEq CH H3C `CH3 Product acid Product basearrow_forward
- 2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). C5H10 Br H-Br CH2Cl2 + enant.arrow_forwardDraw the products of the stronger acid protonating the other reactant. KEq H₂C-O-H H3C OH Product acid Product basearrow_forwardDraw the products of the stronger acid protonating the other reactant. OH KEq CH H3C H3C `CH3 Product acid Product basearrow_forward
- 2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). Ph H-I CH2Cl2arrow_forward3 attempts left Check my work Draw the products formed in the following oxidative cleavage. [1] 03 [2] H₂O draw structure ... lower mass product draw structure ... higher mass productarrow_forward2. Draw the missing structure(s) in each of the following reactions. The missing structure(s) can be a starting material or the major reaction product(s). H-Br CH2Cl2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY