
Concept explainers
Interpretation:
All resonance structures for the product of head-to-tail addition shown in Figure 26-11 are to be drawn. Then curved-arrow notation is to be used to show the movement of electrons in the head-to-tail addition of a growing chain of poly(ethyl acrylate) to a molecule of ethyl acrylate. The resonance structures are to be drawn to explain why head-to tail addition occurs instead of head-to-head addition.
Concept introduction:
Styrene is an unsymmetric vinyl monomer, so we can differentiate between the two carbons of the vinyl group. The less substituted carbon is called the tail, and the more substituted carbon is called the head. As a result, regiochemistry comes into play during
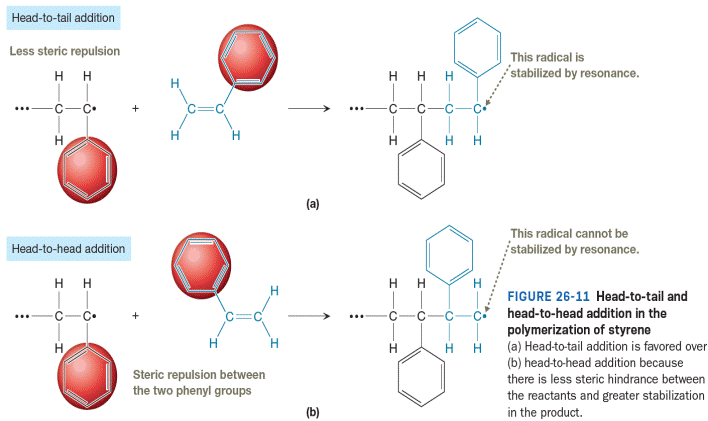
The head-to-tail addition is favored in the polymerization of polystyrene for the two reasons shown in Figure 26-11. First, steric repulsion between the two phenyl groups (one at the end of the propagating radical and one in the monomer) makes it less likely that a head-to-head collision will bring the bond-forming carbons within a bond length. Secondly, the radical formed in the head-to-tail addition is more stable. The radical produced from head-to-tail addition is resonance stabilized while the radical produced from head-to-head addition is not.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
ORG CHEM W/ EBOOK & SW5 + STUDY GUIDE
- Would the following organic synthesis occur in one step? Add any missing products, required catalysts, inorganic reagents, and other important conditions. Please include a detailed explanation and drawings showing how the reaction may occur in one step.arrow_forward(a) Sketch the 'H NMR of the following chemical including the approximate chemical shifts, the multiplicity (splitting) of all signals and the integration (b) How many signals would you expect in the 13C NMR? CH3arrow_forwardDraw the Show the major and minor product(s) for the following reaction mechanisms for both reactions and show all resonance structures for any Explain why the major product is favoured? intermediates H-Brarrow_forward
- 3. Draw ALL THE POSSBILE PRODUCTS AND THE MECHANISMS WITH ALL RESONANCE STRUCTURES. Explain using the resonance structures why the major product(s) are formed over the minor product(s). H₂SO4, HONO CHarrow_forward7. Provide the product(s), starting material(s) and/or condition(s) required for the No mechanisms required. below reaction HO + H-I CI FO Br2, FeBr3 O I-Oarrow_forward6. Design the most efficient synthesis of the following product starting from phenot Provide the reaction conditions for each step (more than one step is required) and explain the selectivity of each reaction. NO MECHANISMS ARE REQUIRED. OH step(s) CIarrow_forward
- What is the skeletal structure of the product of the following organic reaction?arrow_forwardIf a reaction occurs, what would be the major products? Please include a detailed explanation as well as a drawing showing how the reaction occurs and what the final product is.arrow_forwardWhat is the major organic product of the following nucleophilic acyl substitution reaction of an acid chloride below?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
