
Concept explainers
(a)
Interpretation: To determine whether oxaloacetate and
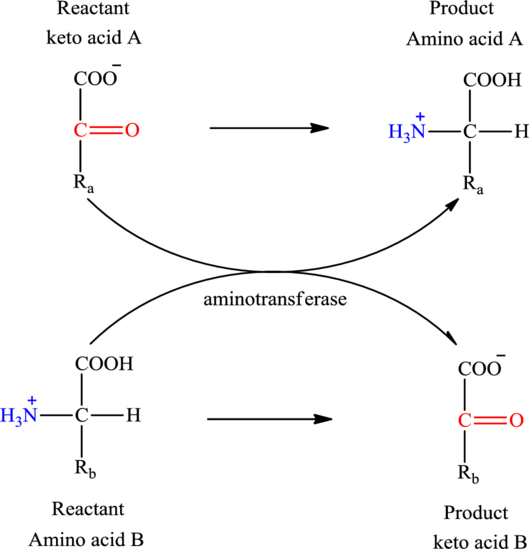
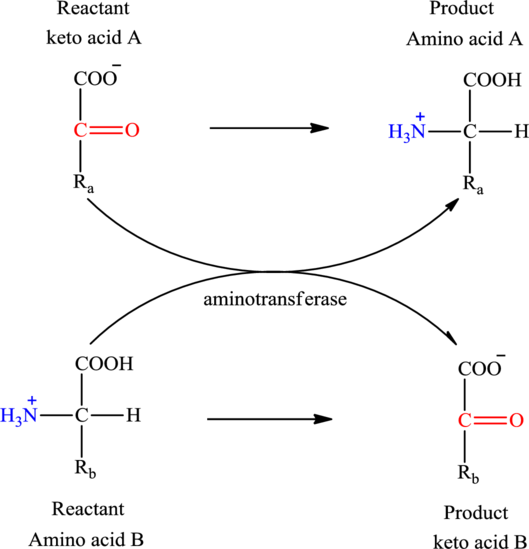
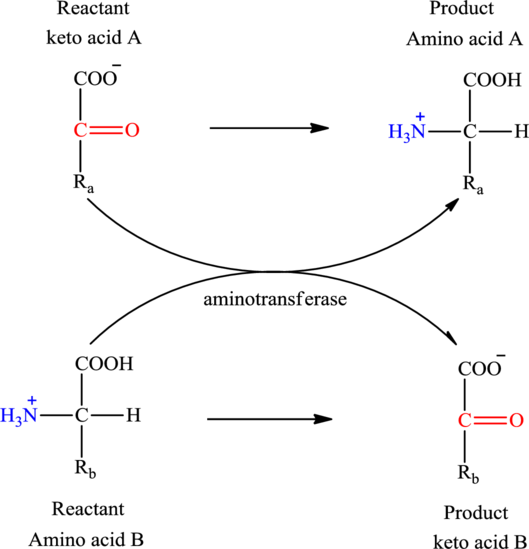
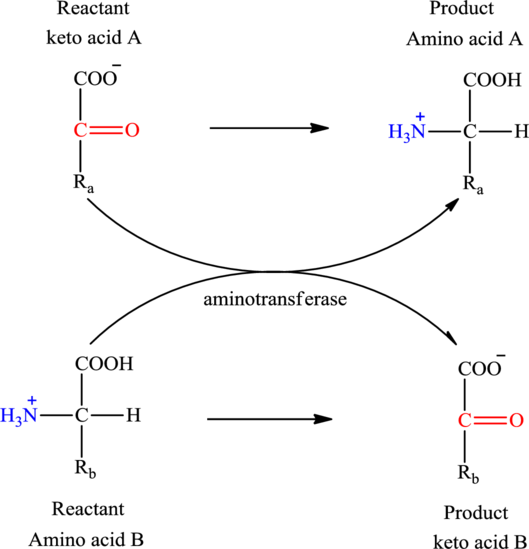
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
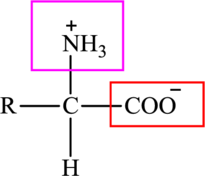
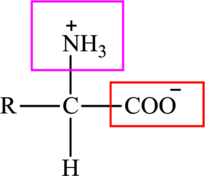
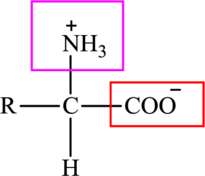
The general structure of an amino acid is:
Here,
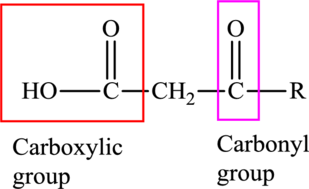
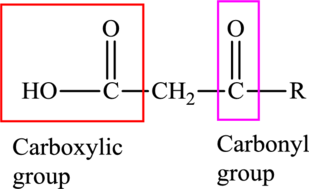
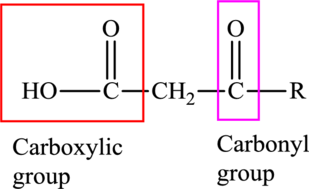
An acid containing both carbonyl and carboxyl
(a)
Answer to Problem 26.33EP
No, oxaloacetate and
Explanation of Solution
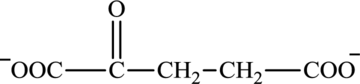
Oxaloacetate is a keto acid and its structure is:
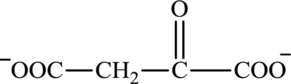
The two reactants in transamination reaction are a keto acid and an amino acid. Both oxaloacetate and
(b)
Interpretation: To determine whether glutamate and oxaloacetate could function as the two reactants in a transamination reaction or not.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
The general structure of an amino acid is:
Here,
An acid containing both carbonyl and carboxyl functional group is known as a keto acid. A general representation of a keto acid is:
(b)
Answer to Problem 26.33EP
Yes, glutamate and oxaloacetate can function as the reactants in a transamination reaction.
Explanation of Solution
Glutamate is an amino acid and its structure is:
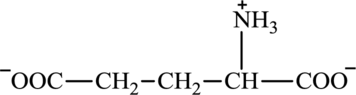
Oxaloacetate is a keto acid and its structure is:
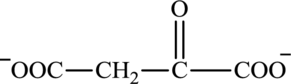
Transamination reaction involves the exchange of an amino group from an
(c)
Interpretation: To determine whether glutarate and glutamate could function as the two reactants in a transamination reaction or not.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
The general structure of an amino acid is:
Here,
An acid containing both carbonyl and carboxyl functional group is known as a keto acid. A general representation of a keto acid is:
(c)
Answer to Problem 26.33EP
No, glutarate and glutamate cannot function as the reactants in a transamination reaction.
Explanation of Solution
Glutarate is a diacid and its structure is:
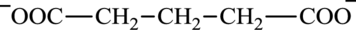
Glutamate is an amino acid and its structure is:
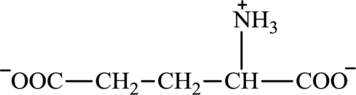
The two reactants in transamination reaction are a keto acid and an amino acid. Glutamate is an amino acid but glutarate is not a keto acid. For a transamination reaction to take place there must be one keto acid present along with an amino acid. Thus, glutarate and glutamate cannot function as the reactants in a transamination reaction.
(d)
Interpretation: To determine whether oxaloacetate and succinate could function as the two reactants in a transamination reaction or not.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
The general reaction to illustrate transamination is as follows:
The general structure of an amino acid is:
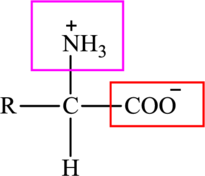
Here,
An acid containing both carbonyl and carboxyl functional group is known as a keto acid. A general representation of a keto acid is:
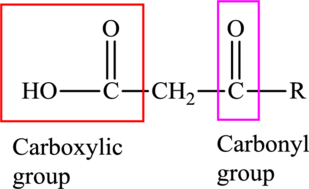
(d)
Answer to Problem 26.33EP
No, oxaloacetate and succinate cannot function as the reactants in a transamination reaction.
Explanation of Solution
Oxaloacetate is a keto acid and its structure is:
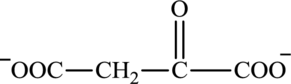
Succinate is a diacid acid and its structure is:
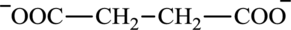
The two reactants in transamination reaction are a keto acid and an amino acid. Oxaloacetate is keto acid but succinate is not an amino acid. For a transamination reaction to take place there must be one amino acid present along with a keto acid. Thus, oxaloacetate and succinate cannot function as the reactants in a transamination reaction.
Want to see more full solutions like this?
Chapter 26 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Consider the reaction below to answer the following questions. HOCH CHOH На A B C D H₂Oarrow_forwardConsider the structures below to answer the following questions. A. Indicate the most acidic hydrogens in each of the molecules. OH CH-H CH₂C-H H&C མིངྒཱའི B. Rank the molecules above in order of increasing acidity (least acidic to most acidic). a. III, II, I b. II, III, I C. I, II, III d. II, I, IIIarrow_forwardConsider the reaction below to answer the following questions. H H+ A B CH₂OH 5% NaOCH, CH₂OH A. Which carbonyl compound functions as the electrophile in this reaction? B. Draw the structure of the enolate ion that is generated during the course of this reaction. C. This reaction is an example of: a. a mixed Claisen condensation. b. C. d. a Dieckman condensation. a Michael reaction. a mixed aldol reaction.arrow_forward
- Give the major organic product(s) of each of the following reactions or sequences of reactions. Show all relevant stereochemistry. [two only] CH3O (11 HC-C-C-CH3 A. CH3 12. NaOHarrow_forwardDiethyl malonate can be prepared by the following reaction sequence. Draw the structures of each of the missing intermediates in the boxes provided. 17 1. Br PBr H&C OH 2 H₂O CH3CH₂OH На NaCN H₂SO4 NC. CH CH₂OH на H₂O, heat CH₂ OCHCH3 ཝསི། ཡིཀྑཱམུདྡྷནྟ CH₂ OEtarrow_forwardThe reaction of a carboxylic acid with an alcohol in the presence of acid is termed Fischer esterification. OH + CH₂OH На B C A. The nucleophile in this reaction is B. Compound C functions as a. a base scavenger b. a solvent C. a catalyst d. a neutralizer C. Fischer esterification is an example of: . a. nucleophilic acyl addition b. nucleophilic acyl substitution c. nucleophilic acyl elimination d. nucleophilic acyl rearrangement in this reaction.arrow_forwardA highly useful and general method for the synthesis of alcohols is the addition of Grignard reagents to carbonyl compounds. Show what Grignard reagent and what carbonyl compound you would start with to prepare each alcohol below. List all possibilities. OH C-CH2CH3 CH3arrow_forwardRank the following groups of compounds from most acidic (1) to least acidic (4). Place the number corresponding to the compound's relative rank in the blank below the structure. NO2 a. b. NO2 NO2 CH,CH,CH,CH,OH. CHCHCH-CHOH. CH-CH-CH,CH;OH CH-CHCH-CH-OH OH OH CH₂OH COH ဒီ ပုံ ပုံ H&C CN CN ĆNarrow_forwardGiven the major organic product(s) of each of the following reactions. If none is predicted, write "N.R." answer] a. CHỊCH, CHẤT AIC13 H b. 0 Cl₂ HC- NHOCH3 FeCl3arrow_forwardGive the major organic product(s) for each of the following reactions or sequences of reactions. Show all relevant stereochemistry [2 ONLY]. A -CH2COOH 1. LIAIH THF, heat 2 HO B. C. CH₂Br Br 1. NaCN, acetone 2 H₂O, heat 1. Mg ether 3 HO Z CO₂arrow_forwardWhat is the order of increasing acidity for the following compounds? (least to most) [2 ONLY] A. COOH COOH COOH COOH 6666 a. IV, I, III, II b. III, II, I, IV с. II, III, I, IV d. III, II, IV, I slingoros CH3 IV woled noise bizarrow_forwardWith this, please answer the following questions: 1.) draw the structure of the electrophilic intermediate in this reaction. 2.) what is the role of the AlCl3 in the reaction 3.) write the complete stepwise mechanism for this reaction. Show all electron flow with arrows and include all intermediate structuresarrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning



