
Concept explainers
Interpretation:
The geometry of the carbon atoms for the given structures is to be predicted. The most nucleophilic and most electrophilic species are to be identified and the species, which react with water to give
Concept Introduction:
The VSEPR theory is used to predict the geometry of the molecules, using the number of electron pairs surrounding the central atom.
The most nucleophilic atom is the atom with the highest negative charge.
The most electrophilic atom is the atom with the highest positive charge.
Answer to Problem 102AP
Solution:
(a) The geometry of three molecules is:
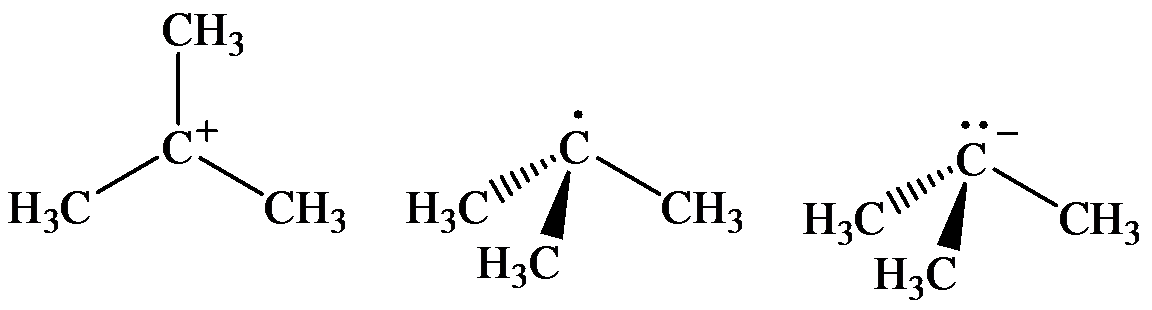
(b) The most nucleophilic atom will be tert-Butyl anion
(c) The species that reacts with water to form
(d) 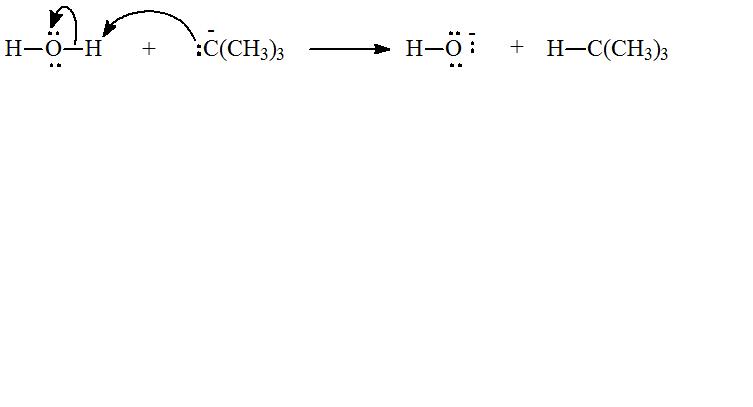
Explanation of Solution
a) The arrangement of bonds to the carton at left linear, tetrahedral, trigonal, trigonal pyramidal
The three molecules are as follows:
The geometry of tert-Butyl cation is:
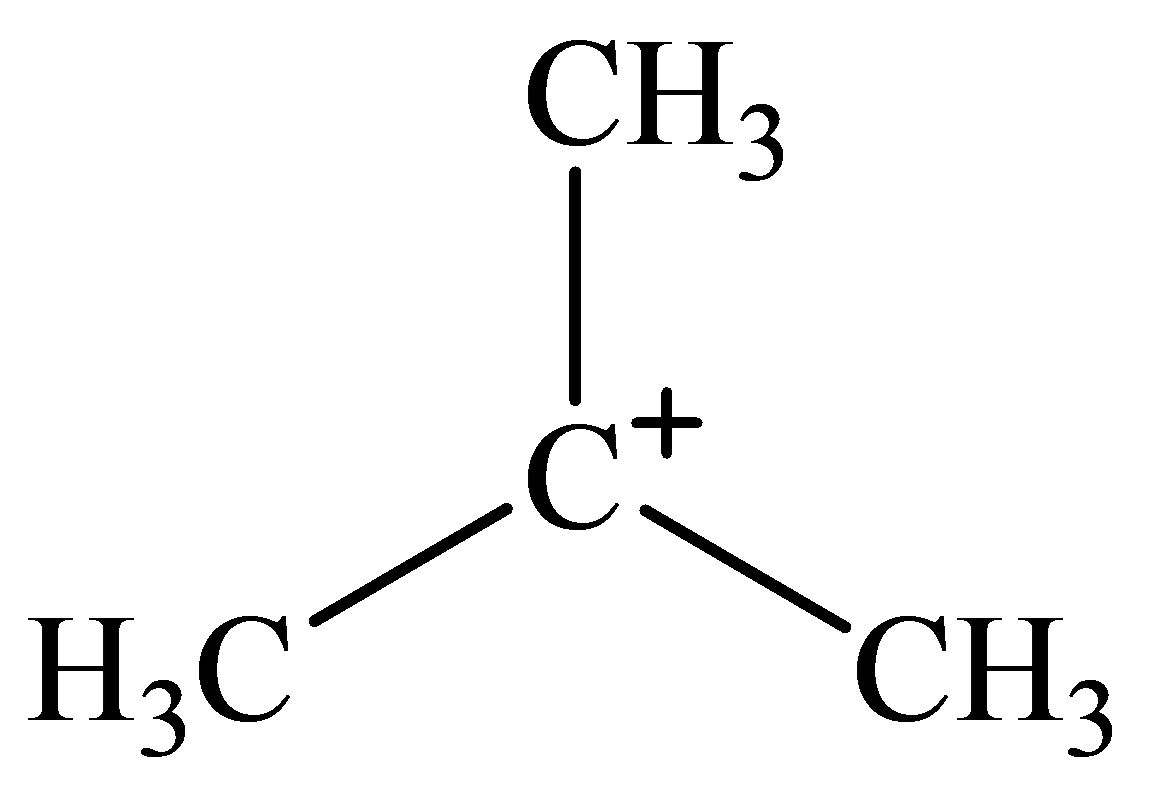
The
molecule has a total of three electron groups around the central atom. It has three bonding electron pairs and zero nonbonding electron pairs, which arrange themselves to make a bond angle of
that indicates trigonal planar geometry.
The geometry of tert-Butyl radical is:
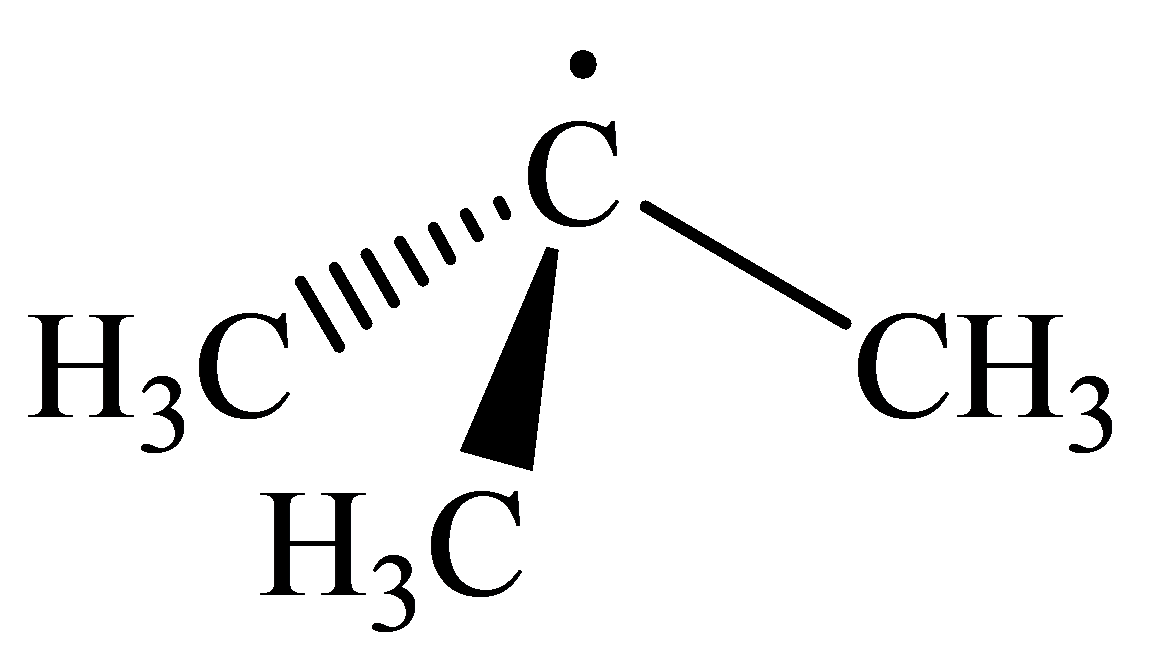
The
radicals have a total of three electron groups around the central atom. It has three bonding electron pairs and one nonbonding electron pair, which arrange themselves to make a bond angle, nearly, of
that indicates distorted trigonal planar geometry.
The geometry of tert-Butyl anion is:
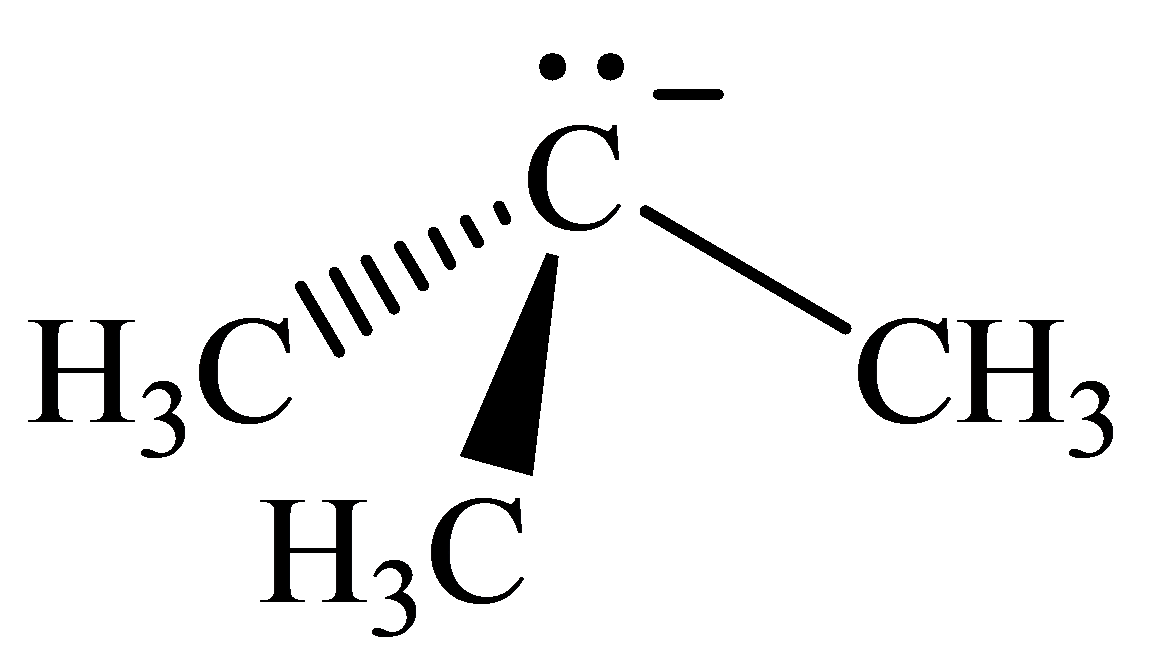
The
molecule has a total of five electron groups around the central atom. It has three bonding electron pairs and one nonbonding electron pair, which arrange themselves to make a bond angle of
that indicates trigonal pyramidal geometry.
b) The most nucleophilic and electrophilic atom
The most nucleophilic atom is the atom with the highest negative charge.
So, the most nucleophilic atom will be tert-Butyl anion,
The most electrophilic atom is the atom with the highest positive charge.
So, the most nucleophile atom will be tert-Butyl cation,
c) The species that reacts with water to give
The tert-Butyl anion has a lone pair that can pair with the proton of water, to form
Hence, the species that reacts with water to form
The reaction of water with species which gives
d) Chemical reaction for the reaction in part (c) and curve arrow to show flow of electron.
The chemical equation for reaction of tert-Butyl anion,
The flow of electrons can be shown as:

Want to see more full solutions like this?
Chapter 25 Solutions
CHEMISTRY >CUSTOM<
- 5. Write the formation reaction of the following complex compounds from the following reactants: 6. AgNO₃ + K₂CrO₂ + NH₄OH → 7. HgNO₃ + excess KI → 8. Al(NO₃)₃ + excess NaOH →arrow_forwardIndicate whether the product formed in the reaction exhibits tautomerism. If so, draw the structure of the tautomers. CO₂C2H5 + CH3-NH-NH,arrow_forwardDraw the major product of this reaction N-(cyclohex-1-en-1-yl)-1-(pyrrolidino) reacts with CH2=CHCHO, heat, H3O+arrow_forward
- Draw the starting material that would be needed to make this product through an intramolecular Dieckmann reactionarrow_forwardDraw the major product of this reaction. Nitropropane reacts + pent-3-en-2-one reacts with NaOCH2CH3, CH3CHOHarrow_forwardIndicate whether the product formed in the reaction exhibits tautomerism. If so, draw the structure of the tautomers. OC2H5 + CoHs-NH-NH,arrow_forward
- Explain how substitutions at the 5-position of barbituric acid increase the compound's lipophilicity.arrow_forwardExplain how substitutions at the 5-position of phenobarbital increase the compound's lipophilicity.arrow_forwardName an interesting derivative of barbituric acid, describing its structure.arrow_forward
- Briefly describe the synthesis mechanism of barbituric acid from the condensation of urea with a β-diketone.arrow_forwardGiven the hydrazones indicated, draw the structures of the enamines that can be formed. Indicate the most stable enamine (explain). C6H5 C6H5 H C6H5 Harrow_forward4. Propose a Synthesis for the molecule below. You may use any starting materials containing 6 carbons or less (reagents that aren't incorporated into the final molecule such as PhзP do not count towards this total, and the starting material can have whatever non-carbon functional groups you want), and any of the reactions you have learned so far in organic chemistry I, II, and III. Your final answer should show each step separately, with intermediates and conditions clearly drawn.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





