
Chemistry the Central Science 13th Edition Custom for Lamar University
13th Edition
ISBN: 9781269962667
Author: Theodore L. Brown, H. Eugene LeMay Jr., Bruce E. Bursten, Batherine J. Murphy, Patrick M. Woodward, Matthew W. Stoltzfus
Publisher: Pearson Learning Center
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 48E
Which of these statements about how the percent ionization of a weak acid depends on acid concentration is true? [Section 16.6]
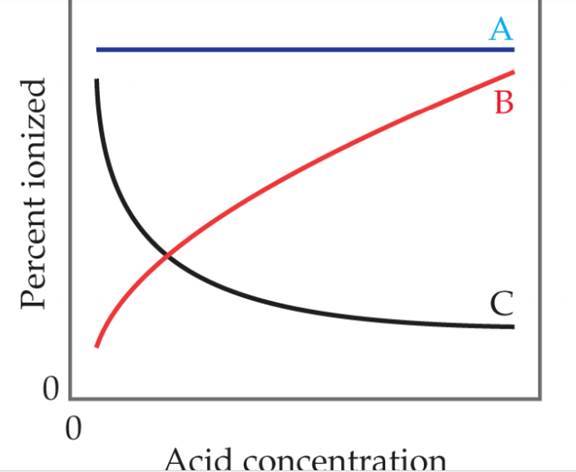
- Line A is most accurate because Ka does not depend on concentration.
- Line A is the most accurate because the percent ionization of the acid does not depend on concentration.
- Line B is the most accurate because as the acid concentration increases, a greater proportion of it is ionized.
- Line B is the most accurate because as the acid concentration increases, Ka increases.
- Line C is the most accurate because as the acid concentration.
- Line C is the most accurate because as the acid concentration increase, Ka decreases.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
PLEASE HELP! URGENT!
"Water gas" is an industrial fuel composed of a mixture of carbon monoxide and hydrogen gases. When this
fuel is burned, carbon dioxide and water result. From the information given below, write a balanced equation
and determine the enthalpy of this reaction:
CO(g) + O2(g) → CO₂(g) + 282.8 kJ
H2(g) + O2(g) → H₂O(g) + 241.8 kJ
MacBook Air
Page of 3
4. Calculate AG for the following reaction at 25°C. Will the reaction occur (be spontaneous)? How do you
know?
NH3(g) + HCl(g) → NH4Cl(s)
AH=-176.0 kJ
AS-284.8 J-K-1
Chapter 24 Solutions
Chemistry the Central Science 13th Edition Custom for Lamar University
Ch. 24.2 - Prob. 24.1.1PECh. 24.2 - The equilibrium constant Kc for C(s) +CO2 2CO(g)...Ch. 24.2 - Prob. 24.2.1PECh. 24.2 - Le Chatelier noted that many industrial processes...Ch. 24.3 - Prob. 24.3.1PECh. 24.3 - Prob. 24.3.2PECh. 24.3 - [15.91] An equilibrium mixture of H2, I2, and HI...Ch. 24.3 - Consider the hypothetical reaction A(g) + 2B(g) 2...Ch. 24.3 - Prob. 24.5.1PECh. 24.3 - Prob. 24.5.2PE
Ch. 24.4 - Prob. 24.6.1PECh. 24.4 - The following equilibria were measured at 823 K:...Ch. 24.7 - Prob. 24.7.1PECh. 24.7 - Prob. 24.7.2PECh. 24.7 - At 800 K, the equilibrium constant for the...Ch. 24.7 - Prob. 24.8.2PECh. 24 - Practice Exercise 1 A solution at 25° C has pOH =...Ch. 24 - Prob. 1ECh. 24 - Prob. 2ECh. 24 - Practice Exercise 2 Predict whether the...Ch. 24 - Prob. 4ECh. 24 - Prob. 5ECh. 24 - Prob. 6ECh. 24 - Prob. 7ECh. 24 - Prob. 8ECh. 24 - Prob. 9ECh. 24 - Prob. 10ECh. 24 - Prob. 11ECh. 24 - Prob. 12ECh. 24 - Practice Exercise 2 For each reaction, use Figure...Ch. 24 - Practice Exercise 1 In a certain acidic solution...Ch. 24 - Prob. 15ECh. 24 - Prob. 16ECh. 24 - Prob. 17ECh. 24 - Practice Exercise 1 A solution at 250C has [OH-] =...Ch. 24 - Practice Exercise 2 In a sample of lemon juice,...Ch. 24 - Prob. 20ECh. 24 - Prob. 21ECh. 24 - Prob. 22ECh. 24 - Prob. 23ECh. 24 - Practice Exercise 2 Niacin, one of the B vitamins,...Ch. 24 - Prob. 25ECh. 24 - Practice Exercise 2 A 0.020 M solution of niacin...Ch. 24 - Practice Exercise 1 What is the pH of a 0.40 M...Ch. 24 - Practice Exercise 2 The Ka for niacin (Sample...Ch. 24 - Prob. 29ECh. 24 - Practice Exercise 2 Calculate the pH of a 0.020 M...Ch. 24 - Prob. 31ECh. 24 - Practice Exercise 2 Which of the following...Ch. 24 - Prob. 33ECh. 24 - Practice Exercise 2 What is the morality of an...Ch. 24 - Practice Exercise 1 By using information from...Ch. 24 - Practice Exercise 2 Based on information in...Ch. 24 - Prob. 37ECh. 24 - Prob. 38ECh. 24 - Practice Exercise 1 How many of the following...Ch. 24 - In each pair, choose the compound that gives the...Ch. 24 - Prob. 41ECh. 24 - a. Identify the Br ted-Lowry acid and base in the...Ch. 24 - Prob. 43ECh. 24 - Prob. 44ECh. 24 - Prob. 45ECh. 24 - 16.5 The following diagrams represent aqueous...Ch. 24 - Prob. 47ECh. 24 - Which of these statements about how the percent...Ch. 24 - 16.8 Each of the three molecules shown here...Ch. 24 - Prob. 50ECh. 24 - Prob. 51ECh. 24 - Prob. 52ECh. 24 - 16.14 Which of the following statements is...Ch. 24 - Prob. 54ECh. 24 - Prob. 55ECh. 24 - Identify the Bronsted-Lowry acid and the...Ch. 24 - Prob. 57ECh. 24 - Prob. 58ECh. 24 - Prob. 59ECh. 24 - Prob. 60ECh. 24 - Prob. 61ECh. 24 - Prob. 62ECh. 24 - Prob. 63ECh. 24 - Prob. 64ECh. 24 - Prob. 65ECh. 24 - 16.29 Calcualte [H +] for each of the following...Ch. 24 - Prob. 67ECh. 24 - 16.31 At the freezing point of water (0 o C), K10...Ch. 24 - Prob. 69ECh. 24 - 16.35 Complete the following table by calculating...Ch. 24 - Prob. 71ECh. 24 - Prob. 72ECh. 24 - 16.38 Carbon dioxide in the atmosphere dissolves...Ch. 24 - Prob. 74ECh. 24 - Prob. 75ECh. 24 - Prob. 76ECh. 24 - Prob. 77ECh. 24 - Prob. 78ECh. 24 - Prob. 79ECh. 24 - Prob. 80ECh. 24 - Prob. 81AECh. 24 - Prob. 82AECh. 24 - write the chemical equation and the Ka expression...Ch. 24 - Prob. 84AECh. 24 - Prob. 85AECh. 24 - Prob. 86AECh. 24 - Prob. 87AECh. 24 - Prob. 88AECh. 24 - Prob. 89AECh. 24 - Prob. 90AECh. 24 - Prob. 91AECh. 24 - Prob. 92AECh. 24 - Prob. 93AECh. 24 - Calculate the percent ionization of hydrazoic acid...Ch. 24 - Prob. 95IECh. 24 - Prob. 96IECh. 24 - Prob. 97IECh. 24 - Prob. 98IECh. 24 - 16.68 The hypochlorite ion, CIO- , acts as a weak...Ch. 24 - Prob. 100IECh. 24 - Practice Exercise 1 Consider the following...Ch. 24 - Prob. 102IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- true or false The equilibrium constant for this reaction is 0.20. N2O4(g) ⇔ 2NO2(g) Based on the above, the equilibrium constant for the following reaction is 5. 4NO2(g) ⇔ 2N2O4(g)arrow_forwardtrue or false The equilibrium constant for this reaction is 0.20. N2O4(g) ⇔ 2NO2(g) Based on the above, the equilibrium constant for the following reaction is 0.4. 2N2O4(g) ⇔ 4NO2(g)arrow_forwardtrue or false Using the following equilibrium, if heat is added the equilibrium will shift toward the reactants. N2(g) + 3H2(g) ⇔ 2NH3(g) + heatarrow_forward
- True or False Using the following equilibrium, if heat is added the equilibrium will shift toward the products. N2O4(g) + heat ⇔ 2NO2(g)arrow_forwardtrue or false Using the following equilibrium, if solid carbon is added the equilibrium will shift toward the products. C(s) + CO2(g) ⇔ 2CO(g)arrow_forwardProvide the complete mechanism for the reaction below. You must include appropriate arrows,intermediates, and formal charges. Please also provide a reason to explain why the 1,4-adduct is preferred over the 1,3-adduct.arrow_forward
- Which of the following pairs are resonance structures of one another? I. III. || III IV + II. :0: n P !༠ IV. EN: Narrow_forwardPredict the major organic product(s) and byproducts (either organic or inorganic) for thefollowing reactions.arrow_forwardA 8.25 g sample of aluminum at 55°C released 2500 J of heat. The specific heat of aluminum is 0.900 J/g°C. The density of aluminum is 2.70 g/mL. Calculate the final temperature of the aluminum sample in °C.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY