
Concept explainers
a)
Interpretation:
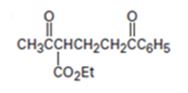
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are to be given.
Concept introduction:
Michael reaction involves the conjugate addition of a stable enolate ion derived from a β-ketoesters or β-diketones or β-ketonitriles or malonic esters (donors) to an unhindered α,β-unsaturated
To give:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown.
Answer to Problem 62AP
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are
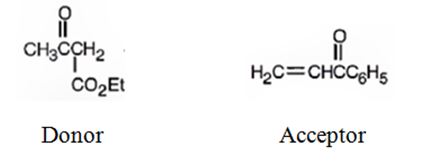
Explanation of Solution
An analysis of the structure of the compound indicates that it is formed by the reaction between the ethylacetoacetate (nucleophilic donor) and phenyl vinyl ketone (electrophilic acceptor).
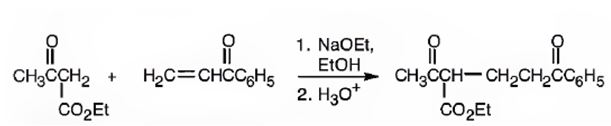
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are
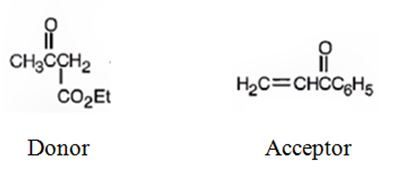
b)
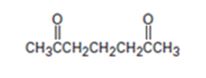
Interpretation:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are to be given.
Concept introduction:
Michael reaction involves the conjugate addition of a stable enolate ion derived from a β-ketoesters or β-diketones or β-ketonitriles or malonic esters (donors) to an unhindered α,β-unsaturated ketones or aldehydes or esters or thioesters or nitriles or amides or nitro compounds (acceptors). The enolate ion from the donor attacks the double bond in acceptor. A new bond is formed between the α-carbon of the donor and the β-carbon of the unsaturated ester.
To give:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown.
Answer to Problem 62AP
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

Explanation of Solution
An analysis of the structure of the compound indicates that it is formed by the reaction between the ethylacetoacetate (nucleophilic donor) and methyl vinyl ketone (electrophilic acceptor).
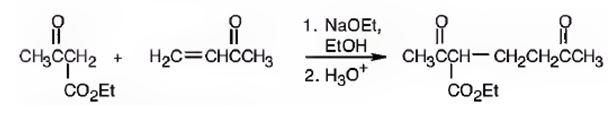
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

c)
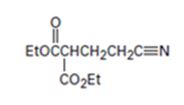
Interpretation:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are to be given.
Concept introduction:
Michael reaction involves the conjugate addition of a stable enolate ion derived from a β-ketoesters or β-diketones or β-ketonitriles or malonic esters (donors) to an unhindered α,β-unsaturated ketones or aldehydes or esters or thioesters or nitriles or amides or nitro compounds (acceptors). The enolate ion from the donor attacks the double bond in acceptor. A new bond is formed between the α-carbon of the donor and the β-carbon of the unsaturated ester.
To give:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown.
Answer to Problem 62AP
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

Explanation of Solution
An analysis of the structure of the compound indicates that it is formed by the reaction between the ethylacetoacetate (nucleophilic donor) and vinyl nitrile (electrophilic acceptor).
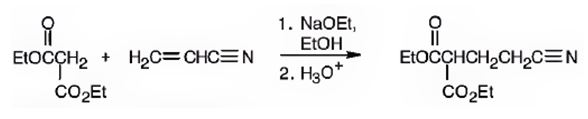
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

d)
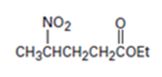
Interpretation:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are to be given.
Concept introduction:
Michael reaction involves the conjugate addition of a stable enolate ion derived from a β-ketoesters or β-diketones or β-ketonitriles or malonic esters (donors) to an unhindered α,β-unsaturated ketones or aldehydes or esters or thioesters or nitriles or amides or nitro compounds (acceptors). The enolate ion from the donor attacks the double bond in acceptor. A new bond is formed between the α-carbon of the donor and the β-carbon of the unsaturated ester.
To give:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown.
Answer to Problem 62AP
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

Explanation of Solution
An analysis of the structure of the compound indicates that it is formed by the reaction between nitro ethane (nucleophilic donor) and ethylacrylate (electrophilic acceptor).
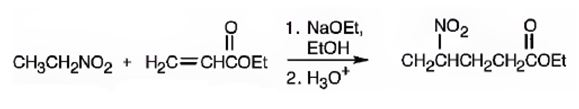
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are
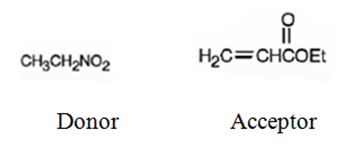
e)
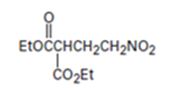
Interpretation:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound are to be given.
Concept introduction:
Michael reaction involves the conjugate addition of a stable enolate ion derived from a β-ketoesters or β-diketones or β-ketonitriles or malonic esters (donors) to an unhindered α,β-unsaturated ketones or aldehydes or esters or thioesters or nitriles or amides or nitro compounds (acceptors). The enolate ion from the donor attacks the double bond in acceptor. A new bond is formed between the α-carbon of the donor and the β-carbon of the unsaturated ester.
To give:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown.
Answer to Problem 62AP
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

Explanation of Solution
An analysis of the structure of the compound indicates that it is formed by the reaction between the ethylsuccinate (nucleophilic donor) and nitro ethene (electrophilic acceptor).
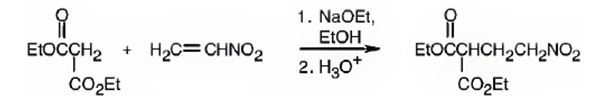
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are

f)
Interpretation:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are to be given.
Concept introduction:
Michael reaction involves the conjugate addition of a stable enolate ion derived from a β-ketoesters or β-diketones or β-ketonitriles or malonic esters (donors) to an unhindered α,β-unsaturated ketones or aldehydes or esters or thioesters or nitriles or amides or nitro compounds (acceptors). The enolate ion from the donor attacks the double bond in acceptor. A new bond is formed between the α-carbon of the donor and the β-carbon of the unsaturated ester.
To give:
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown.
Answer to Problem 62AP
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are
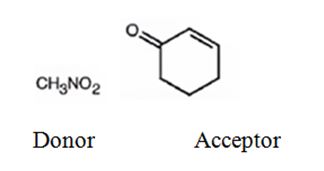
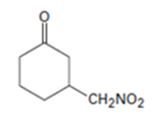
Explanation of Solution
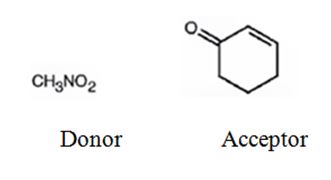
An analysis of the structure of the compound indicates that it is formed by the reaction between nitro methane (nucleophilic donor) and 2-cyclopentenone (electrophilic acceptor).
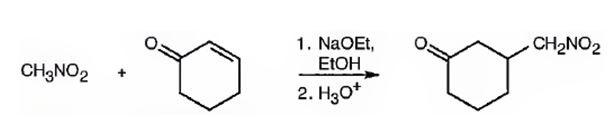
The nucleophilic donor and electrophilic acceptor that react in a Michael reaction to yield the compound shown are
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry - With Access (Custom)
- Draw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M HCl is titrated with 37.75 mL of NaOH. What is the molarity of the NaOH?arrow_forward
- Calculate the pH of 0.450 M KOH.arrow_forwardWhich does NOT describe a mole? A. a unit used to count particles directly, B. Avogadro’s number of molecules of a compound, C. the number of atoms in exactly 12 g of pure C-12, D. the SI unit for the amount of a substancearrow_forward5 What would the complete ionic reaction be if aqueous solutions of potassium sulfate and barium acetate were mixed? ed of Select one: O a 2 K SO4 + Ba2 +2 C₂H3O21 K+SO4 + Ba2+ + 2 C2H3O21 K+SO42 + Ba2 +2 C2H3O2 BaSO4 +2 K+ + 2 C2H3O estion Ob. O c. Od. 2 K SO4 +Ba2 +2 C₂H₂O₂ BaSO4 + K+ + 2 C2H3O BaSO4 + K + 2 C2H301 →Ba² +SO42 +2 KC2H3O s pagearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


