
Concept explainers
a)
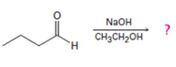
Interpretation:
The dehydration product formed in the addition reaction of butanal along with the mechanism of its formation is to be predicted.
Concept introduction:
The β- hydroxyl aldehyde or ketone obtained loses a molecule of water upon heating to give an α, β- unsaturated aldehyde or ketone.
To predict:
The dehydration product formed in the addition reaction of butanal along with the mechanism of its formation.
Answer to Problem 28MP
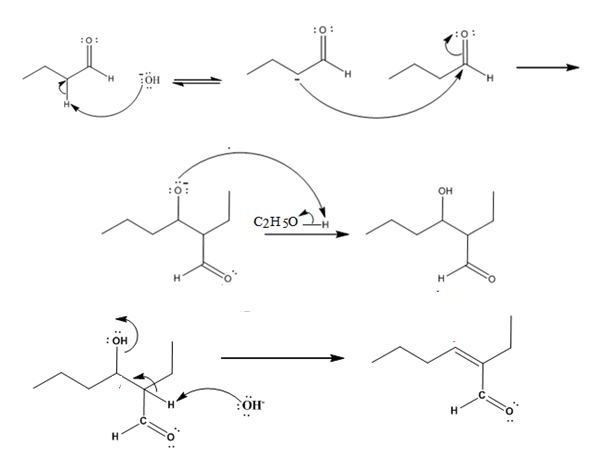
The dehydration product formed in the addition reaction of butanal is 2-ethyl-2-hexenal.
The mechanism of its formation is
Explanation of Solution
In the first step the base picks up an acidic proton from the diketone to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbon of the other carbonyl group in the same molecule to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield the β- hydroxyl aldehyde. Removal of a proton by the base from the hydroxy aldehyde leads to the formation of α, β- unsaturated aldehyde.
The dehydration product formed in the addition reaction of butanal is 2-ethyl-2-hexenal.
The mechanism of its formation is

b)
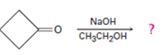
Interpretation:
The dehydration product formed in the addition reaction given along with the mechanism of its formation is to be predicted.
Concept introduction:
Aldehydes and ketones that have α- hydrogen atom undergo aldol condensation to yield a β- hydroxyl aldehyde or ketone as the product. The reaction occurs in three steps i) Abstraction of α- hydrogen by a base to yield an enolate anion ii) Attack of the anion on the carbonyl carbon of another molecule iii) Protonation of the alkoxide intermediate.
The β- hydroxyl aldehyde or ketone obtained on heating loses a water molecule to yield an α, β- unsaturated aldehyde or ketone.
To identify:
The dehydration product formed in the addition reaction of cyclobutanone along with the mechanism of its formation.
Answer to Problem 28MP
The dehydration product formed in the addition reaction of cyclobutanone is
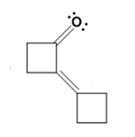
The mechanism of its formation is
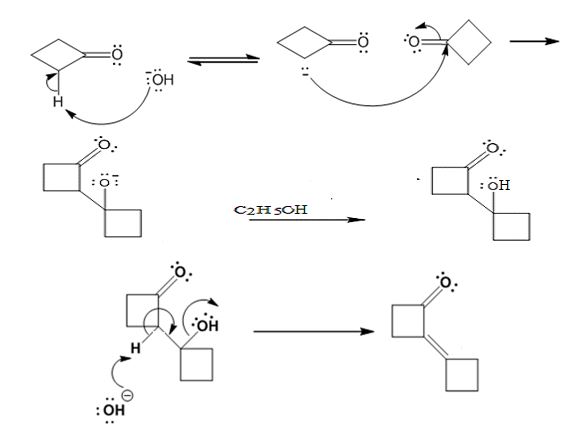
Explanation of Solution
In the first step the base picks up a proton from the α-carbon of one cyclobutanone molecule to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbonyl carbon of another cyclobutanoe molecule to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield a β- hydroxyketone. In the final step the base picks up a proton from the hydroxyl group that leads to the formation of α, β- unsaturated ketone.
The dehydration product formed in the addition reaction of cyclobutanone is

The mechanism of its formation is

c)
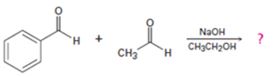
Interpretation:
The dehydration product formed in the addition reaction given along with the mechanism of its formation is to be predicted.
Concept introduction:
The reaction given is a mixed aldol reaction. Aldehydes and ketones that have α- hydrogen atom undergo aldol condensation to yield a β- hydroxyl aldehyde or ketone as the product. The reaction occurs in three steps i) Abstraction of α- hydrogen by a base to yield an enolate anion ii) Attack of the anion on the carbonyl carbon of another molecule iii) Protonation of the alkoxide intermediate.
The β- hydroxyl aldehyde or ketone loses a molecule of water when heated in the presence of a base to yield α, β- unsaturated aldehyde or ketone.
To identify:
The dehydration product formed in the addition reaction given along with the mechanism of its formation.
Answer to Problem 28MP
The dehydration product formed in the addition reaction given is
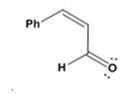
The mechanism of its formation is
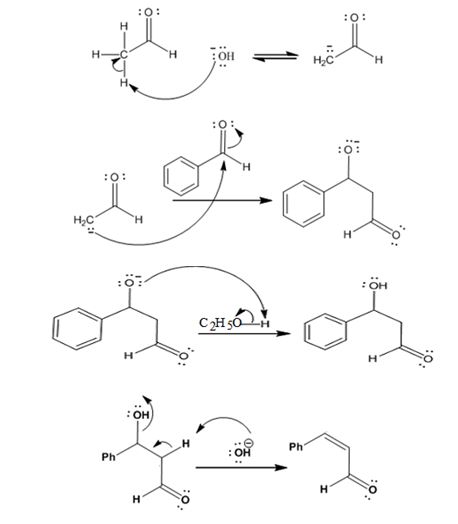
Explanation of Solution
In the first step the base picks up a proton from the α-carbon of acetaldehyde as it has α- hydrogen atoms to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbonyl carbon of benzaldehyde to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield a β- hydroxyaldehyde. In the final step the base picks up a proton from the hydroxyl group that leads to the formation of α, β- unsaturated aldehyde.
The dehydration product formed in the addition reaction given is

The mechanism of its formation is

d)
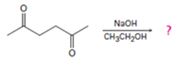
Interpretation:
The product formed in the addition reaction given along with the mechanism of its formation is to be predicted.
Concept introduction:
In intramolecular aldol reactions dicarbonyl compounds such as diketones react with a base to yield a cyclic enone as the products. The reaction occurs in four steps i) Abstraction of α-hydrogen by a base to yield an enolate anion ii) Attack of the anion on the carbonyl carbon in another molecule iii) Protonation of the alkoxide intermediate. iv) Loss of water from the keto alcohol upon heating.
To identify:
The product formed in the addition reaction given along with the mechanism of its formation.
Answer to Problem 28MP
The product formed in the addition reaction given is
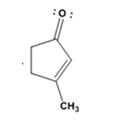
The mechanism of its formation is
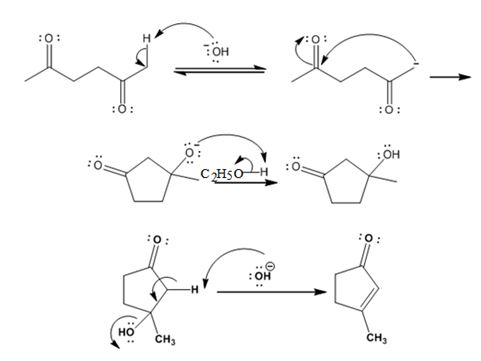
Explanation of Solution
In the first step the base picks up an acidic proton from the diketone to produce the enolate anion. In the next step the nucleophilic enolate anion attacks the electrophilic carbon of the other carbonyl group in the same molecule to give an alkoxide. In the next step the alkoxide intermediate is protonated to yield a hydroxyl ketone. In the final step the base picks up a proton from the hydroxyl group that leads to the formation of α, β- unsaturated aldehyde.
The product formed in the addition reaction given is

The mechanism of its formation is

Want to see more full solutions like this?
Chapter 23 Solutions
EBK ORGANIC CHEMISTRY
- Draw a Haworth projection of a common cyclic form of this monosaccharide CH₂OH HO H HO H H OH CH₂OHarrow_forwardCan you explain how I get these here and show the steps plz?arrow_forwardGive the IUPAC name for this compound Hydrocarbon Condensed Formulas Hint C2H5 CH2CH3 expand that in all the formula Part A: (CH3)2CHCH(C2H5)CH2CH2CH3 Give the IUPAC name for this compound. Part B: CH2=C(C2H5)CH2CH2CH3 Give the IUPAC name for this compound. Part C: (CH3)2C=CHC(C2H5)=CH2 Give the IUPAC name for this compound. Part D: CH3C=CCH(C2H5)2 Give the IUPAC name for this compound. Part E: (CH3)3CC=CCH2CH=C(CH3)2arrow_forward
- Select/ Match the correct letter from the image below for the IUPAC names given below: A B C D 3 E F G H K L Part 1. 4-methylheptane For example.mmmm Answer Letter H _for part 1 Part 2. 2,4-dimethylhexane Part 3. 2,3-dimethylpentane Part 4. 2,2-dimethylhexane Part 5. 2-ethyl-1,1,3,3-tetramethylcyclopentane Part 6. 3-ethyl-2-methylpentanearrow_forwardCan u show the process as to how to get these?arrow_forwardSketch the expected 'H NMR spectra for the following compound. Label all of the H's in the structure and the corresponding signal for the spectra you sketch. Make sure you include the integration value and the splitting pattern for each signal Indicate how many signals you would expect in the 13C NMRarrow_forward
- Use IUPAC naming rules to name the following hydrocarbon compounds: CH2-CH3 | a) CH-CH-CH2-CH-CH-CH3 b) | CH2 CH3 | CH3 CH3 \ / C=C H 1 H CH2-CH3 c) d) CH=C-CH3 e) CH3-CH2-CH2-CH=CH-CH3 f) CH2=CH-CH2-CH=CH-CH3 g) CH3-CH2-C = C-CH2-CH3 h)arrow_forwardQ5 Name the following : a. b. C. d. e.arrow_forward25. Predict the major product of the following reaction. 1 equivalent of each of the starting materials was used. H₂C CH3 CH3 H3C H3C H3C. CH2 + H3C. heat CH3 CH H.C. CH3 H.C H.C CH3 CH CH3 CH3 A B C Earrow_forward
- Find chemical structures based on the below information. a) Chemical formula C6H8O Compound is aromatic plus has two 1H NMR peaks that integrated for 3 each that are singlets (it could have more peaks in the 1H NMR b) Chemical Formula: C6H100 Compounds is conjugated 'H NMR has a signal that integrates for 6 and is a doublet IR spectra has a signal at 1730 cm-1arrow_forwardJaslev Propose a synthesis of the following starting from benzene and any other reagents and chemicals. No mechanisms are required. Indicate the condition for each step plus the major product for each step. More than two steps are required. Step 1 Step 2 مہد Brarrow_forwardPart C: The line formula for another branched alkane is shown below. i. In the IUPAC system what is the root or base name of this compound? ii. How many alkyl substituents are attached to the longest chain? iii. Give the IUPAC name for this compound.arrow_forward
