
Organic Chemistry Study Guide and Solutions Manual, Books a la Carte Edition (8th Edition)
8th Edition
ISBN: 9780134649771
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23, Problem 33P
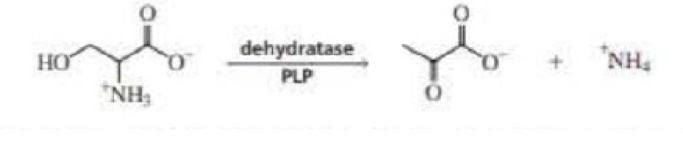
Dehydratase is a PLP-requiring enzyme that catalyzes an α,β-elimination reaction. Propose a mechanism for this reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Using the equation below, what is the rate of this reaction if the rate of disappearance of H2 is 0.44 M/sec?
H2 + Br2 → 2HBr
2Fe3+(aq) + Sn2+(aq) □ 2Fe²+(aq) + Sn 4+ (aq)
If the change in Sn²+ concentration is 0.0010M in 38.5 seconds, what is the rate of disappearance of
Sn²+?
For a neutral hydrogen atom with an electron in the n = 4 state, how many different energies
are possible when a photon is emitted?
4
3
2
1
There are infinite possibilities
Chapter 23 Solutions
Organic Chemistry Study Guide and Solutions Manual, Books a la Carte Edition (8th Edition)
Ch. 23.1 - Prob. 2PCh. 23.1 - Prob. 3PCh. 23.2 - How many conjugated double bonds are there in a....Ch. 23.2 - Instead of adding to the 4a position and...Ch. 23.2 - Prob. 7PCh. 23.3 - Prob. 8PCh. 23.3 - Acetolactate synthase is another TPP-requiring...Ch. 23.3 - Acetolactate synthase transfers the acyl group of...Ch. 23.3 - Prob. 12PCh. 23.5 - Which compound is more easily decarboxylated?
Ch. 23.5 - Prob. 14PCh. 23.5 - Explain why the ability of PLP to catalyze an...Ch. 23.5 - Explain why the ability of PLP to catalyze an...Ch. 23.5 - The enzyme that catalyzes the C C bond cleavage...Ch. 23.5 - Propose a mechanism for the ,-elimination reaction...Ch. 23.6 - Ethanolamine ammonia lyase, a coenzyme...Ch. 23.6 - Prob. 20PCh. 23.7 - How do the structure of tetrahydrofolate and...Ch. 23.7 - What is the source of the methyl group in...Ch. 23.8 - Thiols such as ethanethiol and propanethiol can be...Ch. 23 - How does the metal ion in carboxypeptidase A...Ch. 23 - Prob. 24PCh. 23 - Prob. 25PCh. 23 - For each of the following reactions, name both the...Ch. 23 - Prob. 27PCh. 23 - When transaminated, the three branched-chain amino...Ch. 23 - What acyl groups have we seen transferred by...Ch. 23 - Propose a mechanism for the following reaction:Ch. 23 - Draw the products of the following reaction, where...Ch. 23 - When UMP is dissolved in T2O, exchange of T for H...Ch. 23 - Dehydratase is a PLP-requiring enzyme that...Ch. 23 - In addition to the reaction mentioned in Section...Ch. 23 - PLP can catalyze both ,-elimination reactions...Ch. 23 - The glycine cleavage system is a group of four...Ch. 23 - Prob. 37PCh. 23 - FADH2 reduces , -unsaturated thioesters to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2 NO(g) + H2(g) → N2(g) +2 H2O(g) If NO has rate of disappearance of 0.025 M/min, what is the rate of this reaction?arrow_forward2Fe3+(aq) + Sn2+(aq) □ 2Fe²+(aq) + Sn 4+ (aq) If the change in Sn2+ concentration is 0.0010M in 38.5 seconds, what is the rate of appearance of Fe²+?arrow_forwardUsing the equation below, if the rate of disappearance of Cl2 is 0.26 M/min, what is the rate of this reaction? 2NO(g) + Cl2(g) → 2NOCI(g)arrow_forward
- A 45.0 mL solution containing a mixture of 0.0634 M KCN and 0.0634 M KCI is titrated with 0.107 M AgNO. From this mixture, which silver salt will precipitate first? A list of Ksp values can be found in the table of solubility constants. • AgCI • not enough information to determine AgCN What is the concentration of Ag* at the first equivalence point? [Ag*] = Will the second silver salt begin to precipitate at the first equivalence point before the first silver salt has completely precipitated? • not enough information to determine • yes • noarrow_forward[Review Topics] [References] Indicate whether the pair of structures shown represent stereoisomers, constitutional isomers, different conformations of the same compound, or the same conformation of a compound viewed from a different perspective. Note that cis, trans isomers are an example of stereoisomers. H₂N ✓ CI H₂N NH2 NH₂ CI Submit Answer Retry Entire Group 2 more group attempts remaining Previous Next>arrow_forwardDon't used Ai solutionarrow_forward
- Draw resonance structures for the following compounds. Please provide a thorough explanation that allows for undertanding of topic.arrow_forwardBF3 has a no dipole moment. a) Draw the Lewis structure for BF3, showing all nonbonding electrons. b) Indicate the polarity of every atom in the structure using δ+ and δ– notation, and explain why the molecule has no net dipole. Please provide a thorough explanation that allows for undertanding of topic.arrow_forwardFor each reaction shown below follow the curved arrows to complete each equation by showing the structure of the products. Identify the acid, the base, the conjugated acid and conjugated base. Consutl a pKa table and choose the direciton the equilibrium goes. Please provide a thorough explanation that allows for undertanding of topic.arrow_forward
- Need help understanding please help Let’s assume the initial volume of the gas is 4.80 LL , the initial temperature of the gas is 29.0 °C°C , and the system is in equilibrium with an external pressure of 1.2 bar (given by the sum of a 1 bar atmospheric pressure and a 0.2 bar pressure due to a brick that rests on top of the piston). What is the final pressure of the gas? What is the final volume of the gas? What happens with the piston after you finish heating the gas? Assume you do not need to worry about the gas cooling down again because the outside of the container is at a lower temperature. That is, you manage to keep the gas at a constant temperature that equals 54.2 °C°C What is the sign of w? What is the value of w? Be careful with units. How do you convert bar*L to J?arrow_forwardFor a neutral hydrogen atom with an electron in the n = 4 state, how many different energies are possible when a photon is emitted?arrow_forwardFor the following compound identify the lone pairs and indicate if each lone pair is localized or delocalized. Please provide a thorough explanation that allows for undertanding of topic.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY