
Concept explainers
The fatty acid composition of three triacylglycerols (A, B, and C) is reported below. Predict which one has the highest melting point. Which one do you expect to be liquid (oil) at room temperature? Explain.
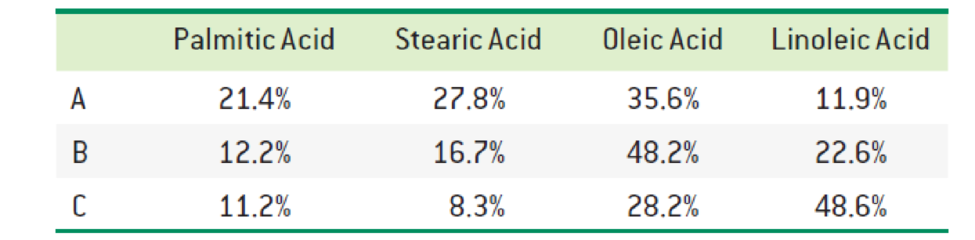
Interpretation:
The triacylglycerols with highest melting point and fatty acid expected to be liquid at room temperature has to be identified.
Concept Introduction:
Triacylglycerol:
It is also known as triglyceride, an ester composed of glycerol and long chains of acyl fatty acids.
Explanation of Solution
The fatty acid composition A has the highest melting point and B and C are expected to be at low melting point.
Since the composition A contains
The triacylglycerols with highest melting point and fatty acid expected to be liquid at room temperature was identified.
Want to see more full solutions like this?
Chapter 23 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- 14. What is the IUPAC name of this compound? A) 6-hydroxy-4-oxohexanenitrile B) 5-cyano-3-oxo-1-pentanol C) 5-cyano-1-hydroxy-3-pentanone D) 1-cyano-5-hydroxy-3-pentanone E) 5-hydroxy-3-oxopentanenitrile HO. CNarrow_forward13. What is the IUPAC name of this compound? A) 5-hydroxy-3,3-dimethylpentanoic acid B) 3,3-dimethylpentanoic acid C) 3,3-dimethyl-1-oxo-1,5-pentanediol D) 1,5-dihydroxy-3,3-dimethylpentanal E) 4-hydroxy-2,2-dimethylbutanoic acid HO OHarrow_forwardHelp me understand how carbon disulfide leads to toxicity in the brain, using terms like distal axonopathy, neurofilaments, covalent cross-linking, adducts, etc.,...please intuitively explain what is happening and where and the effects of it. For example, I know that CS2 reacts with amide and sulfhydryl groups on proteins, but what proteins exactly and where are they located?arrow_forward
- What is the standard free energy change (in kJ/mole) of the spontaneous reaction between Oxygen and NADH to form H2O2 and NAD+?arrow_forwardRedox Chemistry: Give standard free energy changes expected for the following reactions:-Succinate -> fumarate (using FAD/FADH2)-Oxaloacetate -> Malate (using NAD/NADH)-NADH --> NAD+ (using FMN/FMNH2)-CoQ --> CoQH2 (using Cytochrome C)arrow_forwardGive examples of balanced redox reactions that match the following:-Catabolic-Anabolic-Oxidative-Reductivearrow_forward
- If there are 20uM of a GLUT2 transporter on the surface of a cell, each able to move 8 per second, and 50mM glucose outside of the cell, what is the flux into the cell in mM/sec?arrow_forwardA transporter is responsible for antiporting calcium and glucose. The transporter brings glucose into the cell and sends calcium out of the cell. If blood [calcium] = 2.55mM and intracellular [calcium] = 7uM, blood [glucose] = 5.2mM, and intracellular [glucose] = 40uM, what is the free energy of transport? Assume a membrane potential of 62mV (negative inside).arrow_forwardAn ATP-coupled transporter is used to import 1 phosphate from the extracellular environment. Intracellular phosphate exists at 65mM, while it is 2mM outside.Assume a free energy change of ATP hydrolysis of -42.7 kJ/mol. What is the net free energy change of the coupled reaction? Assume a membrane potential of 70mV.arrow_forward
- Another transporter brings 3 chloride ions into the cell. Outside, chloride has a concentration of 107mM, and 4mM inside the cell. Assuming a membrane potential of 62mV (negative inside), what is the free energy of transport of these ions?arrow_forwardFor the Oxaloacetate -> Malate reaction, assume the normal ratio of NAD/NADH, what is the maximum ratio of Malate/Oxaloacetate that will allow reaction progress?arrow_forwardA particular particle is trying to cross a membrane by simple diffusion from a high concentration of 20mM to a low concentration of 20uM. If a membrane is 15uM in width, and the diffusion coefficient of the particle is 5 uM/sec, what is the influx in uM/sec?arrow_forward
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning





