
Physics for Scientists and Engineers with Modern Physics, Technology Update
9th Edition
ISBN: 9781305401969
Author: SERWAY, Raymond A.; Jewett, John W.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22.7, Problem 22.5QQ
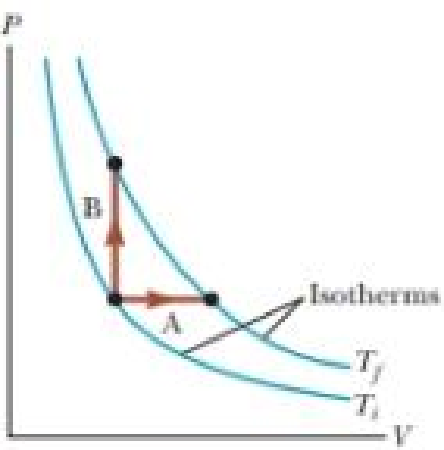
An ideal gas is taken from an initial temperature Ti to a higher final temperature Tf along two different reversible paths as shown in Figure 21.15. Path A is at constant pressure, and path B is at constant volume. What is the relation between the entropy changes of the gas for these paths? (a) ΔSA > ΔSB (b) ΔSA = ΔSB (c) ΔSA < ΔSB
Figure 21.15 (Quick Quiz 21.5) An ideal gas is taken from temperature Ti to Tf via two different paths.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
PROBLEM 3
Cables A and B are Supporting a 185-lb wooden crate.
What is the magnitude of the tension force in each
cable?
A
20°
35°
185 lbs
No chatgpt pls will upvote
No chatgpt pls will upvote
Chapter 22 Solutions
Physics for Scientists and Engineers with Modern Physics, Technology Update
Ch. 22.1 - The energy input to an engine is 4.00 times...Ch. 22.2 - The energy entering an electric heater by...Ch. 22.4 - Three engines operate between reservoirs separated...Ch. 22.6 - (a) Suppose you select four cards at random from a...Ch. 22.7 - An ideal gas is taken from an initial temperature...Ch. 22.7 - True or False: The entropy change in an adiabatic...Ch. 22 - Prob. 1OQCh. 22 - Prob. 2OQCh. 22 - Prob. 3OQCh. 22 - Of the following, which is not a statement of the...
Ch. 22 - Prob. 5OQCh. 22 - Prob. 6OQCh. 22 - Prob. 7OQCh. 22 - Prob. 8OQCh. 22 - Prob. 9OQCh. 22 - Prob. 10OQCh. 22 - The arrow OA in the PV diagram shown in Figure...Ch. 22 - The energy exhaust from a certain coal-fired...Ch. 22 - Discuss three different common examples of natural...Ch. 22 - Prob. 3CQCh. 22 - The first law of thermodynamics says you cant...Ch. 22 - Energy is the mistress of the Universe, and...Ch. 22 - (a) Give an example of an irreversible process...Ch. 22 - The device shown in Figure CQ22.7, called a...Ch. 22 - A steam-driven turbine is one major component of...Ch. 22 - Discuss the change in entropy of a gas that...Ch. 22 - Prob. 10CQCh. 22 - Prob. 11CQCh. 22 - (a) If you shake a jar full of jelly beans of...Ch. 22 - What are some factors that affect the efficiency...Ch. 22 - A particular heat engine has a mechanical power...Ch. 22 - The work done by an engine equals one-fourth the...Ch. 22 - A heat engine takes in 360 J of energy from a hot...Ch. 22 - A gun is a heat engine. In particular, it is an...Ch. 22 - Prob. 5PCh. 22 - Prob. 6PCh. 22 - Suppose a heat engine is connected to two energy...Ch. 22 - Prob. 8PCh. 22 - During each cycle, a refrigerator ejects 625 kJ of...Ch. 22 - Prob. 10PCh. 22 - Prob. 11PCh. 22 - Prob. 12PCh. 22 - A freezer has a coefficient of performance of...Ch. 22 - Prob. 14PCh. 22 - One of the most efficient heat engines ever built...Ch. 22 - Prob. 16PCh. 22 - Prob. 17PCh. 22 - Prob. 18PCh. 22 - Prob. 19PCh. 22 - Prob. 20PCh. 22 - Prob. 21PCh. 22 - How much work does an ideal Carnot refrigerator...Ch. 22 - Prob. 23PCh. 22 - A power plant operates at a 32.0% efficiency...Ch. 22 - Prob. 25PCh. 22 - Prob. 26PCh. 22 - Prob. 27PCh. 22 - Prob. 28PCh. 22 - A heat engine operates in a Carnot cycle between...Ch. 22 - Suppose you build a two-engine device with the...Ch. 22 - Prob. 31PCh. 22 - Prob. 32PCh. 22 - Prob. 33PCh. 22 - Prob. 34PCh. 22 - Prob. 35PCh. 22 - Prob. 36PCh. 22 - Prob. 37PCh. 22 - Prob. 38PCh. 22 - Prob. 39PCh. 22 - Prob. 40PCh. 22 - Prob. 41PCh. 22 - Prob. 42PCh. 22 - A Styrofoam cup holding 125 g of hot water at 100C...Ch. 22 - Prob. 44PCh. 22 - A 1 500-kg car is moving at 20.0 m/s. The driver...Ch. 22 - Prob. 46PCh. 22 - Prob. 47PCh. 22 - Prob. 48PCh. 22 - Prob. 49PCh. 22 - What change in entropy occurs when a 27.9-g ice...Ch. 22 - Calculate the change in entropy of 250 g of water...Ch. 22 - Prob. 52PCh. 22 - Prob. 53PCh. 22 - Prob. 54PCh. 22 - Prob. 55PCh. 22 - Prob. 56APCh. 22 - Prob. 57APCh. 22 - A steam engine is operated in a cold climate where...Ch. 22 - Prob. 59APCh. 22 - Prob. 60APCh. 22 - Prob. 61APCh. 22 - In 1993, the U.S. government instituted a...Ch. 22 - Prob. 63APCh. 22 - Prob. 64APCh. 22 - Prob. 65APCh. 22 - Prob. 66APCh. 22 - In 1816, Robert Stirling, a Scottish clergyman,...Ch. 22 - Prob. 68APCh. 22 - Prob. 69APCh. 22 - Prob. 70APCh. 22 - Prob. 71APCh. 22 - Prob. 72APCh. 22 - Prob. 73APCh. 22 - A system consisting of n moles of an ideal gas...Ch. 22 - A heat engine operates between two reservoirs at...Ch. 22 - Prob. 76APCh. 22 - Prob. 77APCh. 22 - Prob. 78APCh. 22 - A sample of an ideal gas expands isothermally,...Ch. 22 - Prob. 80APCh. 22 - Prob. 81CP
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Rub your hands together vigorously. What happens? Discuss the energy transfers and transformations that take...
College Physics: A Strategic Approach (3rd Edition)
Gregor Mendel never saw a gene, yet he concluded that some inherited factors were responsible for the patterns ...
Campbell Essential Biology (7th Edition)
To test your knowledge, discuss the following topics with a study partner or in writing ideally from memory. Th...
HUMAN ANATOMY
Choose the best answer to each of the following. Explain your reasoning. If Earth were twice as far as it actua...
Cosmic Perspective Fundamentals
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, and Biological Chemistry - 4th edition
Why do scientists think that all forms of life on earth have a common origin?
Genetics: From Genes to Genomes
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Which of the following best describes how to calculate the average acceleration of any object? Average acceleration is always halfway between the initial acceleration of an object and its final acceleration. Average acceleration is always equal to the change in velocity of an object divided by the time interval. Average acceleration is always equal to the displacement of an object divided by the time interval. Average acceleration is always equal to the change in speed of an object divided by the time interval.arrow_forwardThe figure shows the velocity versus time graph for a car driving on a straight road. Which of the following best describes the acceleration of the car? v (m/s) t(s) The acceleration of the car is negative and decreasing. The acceleration of the car is constant. The acceleration of the car is positive and increasing. The acceleration of the car is positive and decreasing. The acceleration of the car is negative and increasing.arrow_forwardWhich figure could represent the velocity versus time graph of a motorcycle whose speed is increasing? v (m/s) v (m/s) t(s) t(s)arrow_forward
- Unlike speed, velocity is a the statement? Poisition. Direction. Vector. Scalar. quantity. Which one of the following completesarrow_forwardNo chatgpt pls will upvote Already got wrong chatgpt answerarrow_forward3.63 • Leaping the River II. A physics professor did daredevil stunts in his spare time. His last stunt was an attempt to jump across a river on a motorcycle (Fig. P3.63). The takeoff ramp was inclined at 53.0°, the river was 40.0 m wide, and the far bank was 15.0 m lower than the top of the ramp. The river itself was 100 m below the ramp. Ignore air resistance. (a) What should his speed have been at the top of the ramp to have just made it to the edge of the far bank? (b) If his speed was only half the value found in part (a), where did he land? Figure P3.63 53.0° 100 m 40.0 m→ 15.0 marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning


College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
The Second Law of Thermodynamics: Heat Flow, Entropy, and Microstates; Author: Professor Dave Explains;https://www.youtube.com/watch?v=MrwW4w2nAMc;License: Standard YouTube License, CC-BY