
(a)
Interpretation: To draw the lewis dot structure and molecular structure of
Concept introduction:
A lewis dot structure is a diagram that show bonding between the atoms. A lewis dot structure is used to predict the geometry of the molecule.
A lewis dot structure is a two dimensional representation whereas a molecular structure is a three dimensional structure and many of the properties both chemical and physical are dictated by molecular structure.
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone pair of electrons.
According to VSEPR theory, the geometry is predicted by the minimizing the repulsions between electron-pairs in the bonds and lone-pairs of electrons. The VSEPR theory is summarized in the given table as,
(a)
Answer to Problem 78GQ
The lewis dot structure of
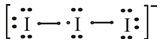
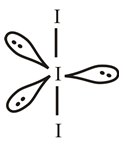
The molecular structure of
Explanation of Solution
The iodine atom belongs to group
The lewis dot structure of
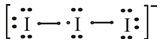
The molecular structure of
The molecular structure of

(b)
Interpretation: To draw the lewis dot structure and molecular structure of
Concept introduction:
A lewis dot structure is a diagram that show bonding between the atoms. A lewis dot structure is used to predict the geometry of the molecule.
A lewis dot structure is a two dimensional representation whereas a molecular structure is a three dimensional structure and many of the properties both chemical and physical are dictated by molecular structure.
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone pair of electrons.
According to VSEPR theory, the geometry is predicted by the minimizing the repulsions between electron-pairs in the bonds and lone-pairs of electrons. The VSEPR theory is summarized in the given table as,
(b)
Answer to Problem 78GQ
The lewis dot structure of
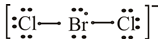
The molecular structure of
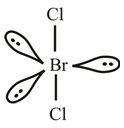
Explanation of Solution
The bromine atom belongs to group
The lewis dot structure of
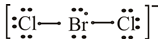
The molecular structure of
The molecular structure of

(c)
Interpretation: To draw the lewis dot structure and molecular structure of
Concept introduction:
A lewis dot structure is a diagram that show bonding between the atoms. A lewis dot structure is used to predict the geometry of the molecule.
A lewis dot structure is a two dimensional representation whereas a molecular structure is a three dimensional structure and many of the properties both chemical and physical are dictated by molecular structure.
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone pair of electrons.
According to VSEPR theory, the geometry is predicted by the minimizing the repulsions between electron-pairs in the bonds and lone-pairs of electrons. The VSEPR theory is summarized in the given table as,
(c)
Answer to Problem 78GQ
The lewis dot structure of
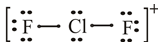
The molecular structure of
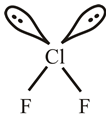
Explanation of Solution
The flourine atom belongs to group
The lewis dot structure of
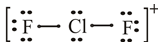
The molecular structure of
The molecular geometry of

(d)
Interpretation: To draw the lewis dot structure and molecular structure of
Concept introduction:
A lewis dot structure is a diagram that show bonding between the atoms. A lewis dot structure is used to predict the geometry of the molecule.
A lewis dot structure is a two dimensional representation whereas a molecular structure is a three dimensional structure and many of the properties both chemical and physical are dictated by molecular structure.
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone pair of electrons.
According to VSEPR theory, the geometry is predicted by the minimizing the repulsions between electron-pairs in the bonds and lone-pairs of electrons. The VSEPR theory is summarized in the given table as,
(d)
Answer to Problem 78GQ
The lewis dot structure of
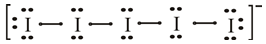
The molecular structure of
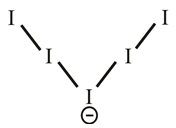
Explanation of Solution
The iodine atom belongs to group
The lewis dot structure of
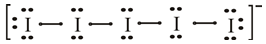
The molecular structure of

Want to see more full solutions like this?
Chapter 21 Solutions
Bundle: Chemistry & Chemical Reactivity, Loose-Leaf Version, 9th + OWLv2, 4 terms (24 Months) Printed Access Card
- Help with annotating the labeled peaks in the 'H NMR (solvent CDCls) spectra and 'H NMR (solvent Acetone-D6) spectra Also help with Calculating the keto-enol tautomerization Ka constant for the product in both solvents.Two solvents and two different Kaarrow_forwardDraw a Haworth projection of a common cyclic form of this monosaccharide CH₂OH HO H HO H H OH CH₂OHarrow_forwardCan you explain how I get these here and show the steps plz?arrow_forward
- Give the IUPAC name for this compound Hydrocarbon Condensed Formulas Hint C2H5 CH2CH3 expand that in all the formula Part A: (CH3)2CHCH(C2H5)CH2CH2CH3 Give the IUPAC name for this compound. Part B: CH2=C(C2H5)CH2CH2CH3 Give the IUPAC name for this compound. Part C: (CH3)2C=CHC(C2H5)=CH2 Give the IUPAC name for this compound. Part D: CH3C=CCH(C2H5)2 Give the IUPAC name for this compound. Part E: (CH3)3CC=CCH2CH=C(CH3)2arrow_forwardSelect/ Match the correct letter from the image below for the IUPAC names given below: A B C D 3 E F G H K L Part 1. 4-methylheptane For example.mmmm Answer Letter H _for part 1 Part 2. 2,4-dimethylhexane Part 3. 2,3-dimethylpentane Part 4. 2,2-dimethylhexane Part 5. 2-ethyl-1,1,3,3-tetramethylcyclopentane Part 6. 3-ethyl-2-methylpentanearrow_forwardCan u show the process as to how to get these?arrow_forward
- Sketch the expected 'H NMR spectra for the following compound. Label all of the H's in the structure and the corresponding signal for the spectra you sketch. Make sure you include the integration value and the splitting pattern for each signal Indicate how many signals you would expect in the 13C NMRarrow_forwardUse IUPAC naming rules to name the following hydrocarbon compounds: CH2-CH3 | a) CH-CH-CH2-CH-CH-CH3 b) | CH2 CH3 | CH3 CH3 \ / C=C H 1 H CH2-CH3 c) d) CH=C-CH3 e) CH3-CH2-CH2-CH=CH-CH3 f) CH2=CH-CH2-CH=CH-CH3 g) CH3-CH2-C = C-CH2-CH3 h)arrow_forwardQ5 Name the following : a. b. C. d. e.arrow_forward
- 25. Predict the major product of the following reaction. 1 equivalent of each of the starting materials was used. H₂C CH3 CH3 H3C H3C H3C. CH2 + H3C. heat CH3 CH H.C. CH3 H.C H.C CH3 CH CH3 CH3 A B C Earrow_forwardFind chemical structures based on the below information. a) Chemical formula C6H8O Compound is aromatic plus has two 1H NMR peaks that integrated for 3 each that are singlets (it could have more peaks in the 1H NMR b) Chemical Formula: C6H100 Compounds is conjugated 'H NMR has a signal that integrates for 6 and is a doublet IR spectra has a signal at 1730 cm-1arrow_forwardJaslev Propose a synthesis of the following starting from benzene and any other reagents and chemicals. No mechanisms are required. Indicate the condition for each step plus the major product for each step. More than two steps are required. Step 1 Step 2 مہد Brarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax




