
(a)
Interpretation:
The complete, detailed mechanism and the overall product for the given reaction is to be drawn.
Concept introduction:
The ester is hydrolyzed to the
Answer to Problem 21.14P
The mechanism and product for the given reaction is:
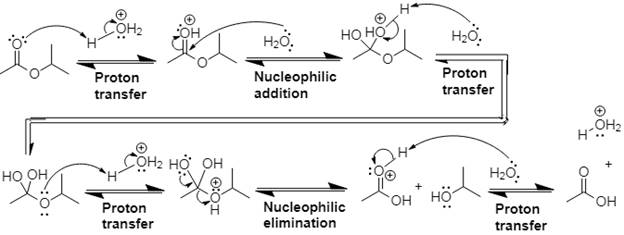
Explanation of Solution
In Step 1 of the mechanism, the acid protonates the ester’s carbonyl group, and in Step 2, water attacks the carbonyl
The complete, detailed mechanism and the overall product for the given reaction is drawn by acid-catalyzed ester hydrolysis.
(b)
Interpretation:
The complete, detailed mechanism and the overall product for the given reaction is to be drawn.
Concept introduction:
This is an acid-catalyzed transesterification, and a new ester is formed.
Answer to Problem 21.14P
The mechanism and product for the given reaction is:
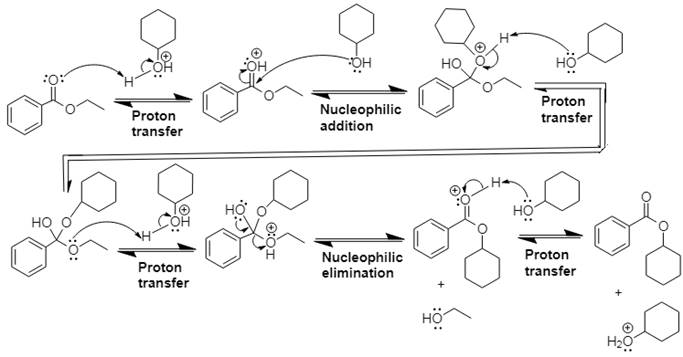
Explanation of Solution
In Step 1 of the mechanism, the acid protonates the ester’s carbonyl group, and in Step 2,
The complete, detailed mechanism and the overall product for the given reaction is drawn by acid-catalyzed transesterification.
(c)
Interpretation:
The complete, detailed mechanism and the overall product for the given reaction is to be drawn.
Concept introduction:
This is an acid catalyzed amide hydrolysis which produces a carboxylic acid. Water acts as the nucleophile in this acid-catalyzed nucleophilic addition–elimination mechanism, and the leaving group is a molecule of
Answer to Problem 21.14P
The mechanism and product for the given reaction is:
Explanation of Solution
In Step 1 of the mechanism, the acid protonates the ester’s carbonyl group, and in Step 2, water attacks the carbonyl
The complete, detailed mechanism and the overall product for the given reaction is drawn by acid-catalyzed amide hydrolysis.
(d)
Interpretation:
The complete, detailed mechanism and the overall product for the given reaction is to be drawn.
Concept introduction:
This is an acid catalyzed ester hydrolysis which produces a carboxylic acid. Water acts as the nucleophile in this acid-catalyzed nucleophilic addition–elimination mechanism, and the leaving group is a molecule of alcohol. Proton transfer steps are incorporated to avoid the appearance of strong bases.
Answer to Problem 21.14P
The mechanism and product for the given reaction is:
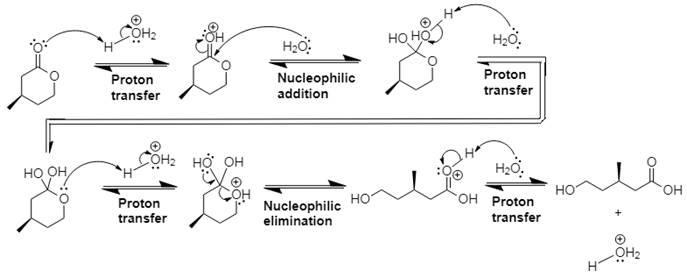
Explanation of Solution
In Step 1 of the mechanism, the acid protonates the ester’s carbonyl group, and in Step 2, water attacks the carbonyl
The complete, detailed mechanism and the overall product for the given reaction is drawn by acid-catalyzed ester hydrolysis.
(e)
Interpretation:
The complete, detailed mechanism and the overall product for the given reaction is to be drawn.
Concept introduction:
This is Fischer esterification reaction.
Answer to Problem 21.14P
The mechanism and product for the given reaction is:
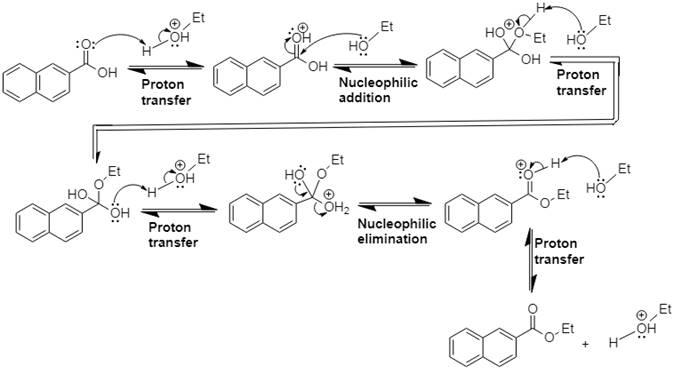
Explanation of Solution
In Step 1 of the mechanism, the acid protonates the acid’s carbonyl group, and in Step 2,
The complete, detailed mechanism and the overall product for the given reaction is drawn by Fischer esterification reaction.
(f)
Interpretation:
The complete, detailed mechanism and the overall product for the given reaction is to be drawn.
Concept introduction:
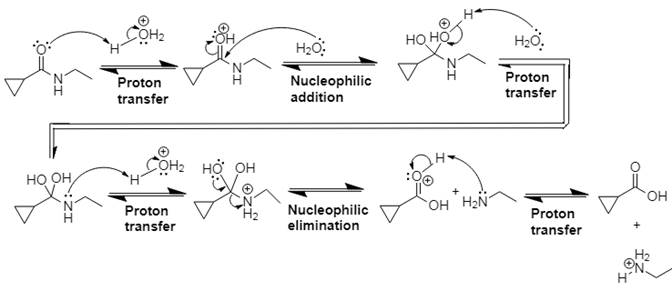
This is an acid catalyzed amide hydrolysis which produces a carboxylic acid. Water acts as the nucleophile in this acid-catalyzed nucleophilic addition–elimination mechanism, and the leaving group is a molecule of amine. Proton transfer steps are incorporated to avoid the appearance of strong bases.
Answer to Problem 21.14P
The mechanism and product for the given reaction is:
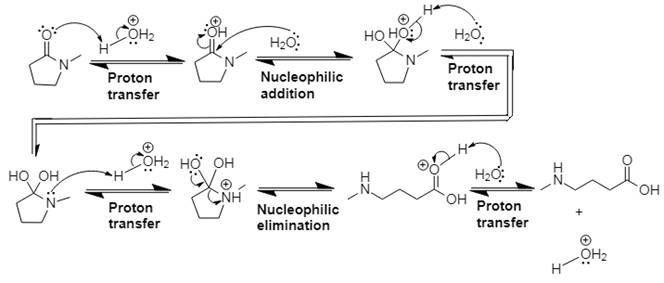
Explanation of Solution
In Step 1 of the mechanism, the acid protonates the ester’s carbonyl group, and in Step 2, water attacks the carbonyl
The complete, detailed mechanism and the overall product for the given reaction is drawn by acid-catalyzed amide hydrolysis.
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry: Principles And Mechanisms
- Draw a tetramer of this alternating copolymer.arrow_forwardH I T H HH H -H C. H- Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above. H- H CIH H H H HHHH H H 0 ·H H– 冊 CH CHI HH C- H- H H- H H A. H H C H H- -H HH H B. H- -H D. H H H H • H -H E. -H H H HICH T HHH F. H-arrow_forwardPolylactic acid (shown below) is a biodegradable polymer used for food packaging. Identify the monomer(s) used in the production of this polymer using a condensation process.arrow_forward
- Draw the product of the reaction shown below. Ignore small byproducts that would evaporate pleasearrow_forwardPoly(ethylene adipate) is a biodegradable polyester (shown below). Identify the type of polymerization process used in the production of this polymer.arrow_forwardPolymers may be composed of thousands of monomers. draw two repeat units(dimer) of the polymer formed in this reaction. assume there are hydrogen atoms on the two ends of the dimer. ignore inorganic byproducts pleasearrow_forward
- Draw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, Ignore inorganic byproductsarrow_forwardDraw the product of this reaction please. Ignore inorganic byproductsarrow_forwardOne of the pi molecular orbitals of 1,3-butadiene (CH2=CHCH=CH2) is shown below. Please identify the number of nodal planes perpendicular to the bonding axisarrow_forward
- Draw the monomers required to synthesize this condensation polymer please.arrow_forwardProvide the correct systematic name for the compound shown here. Please take into account the keyboard options belowarrow_forwardcurved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
