
Concept explainers
a)
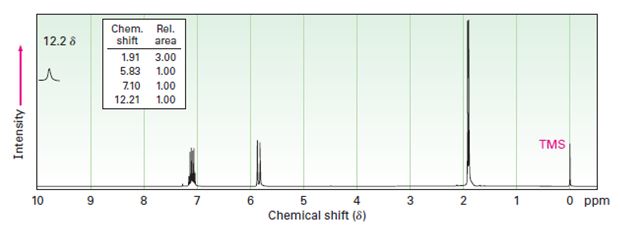
Interpretation:
Two 1HNMR spectra belonging to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) are given. Which corresponds to which is to be explained.
1HNMR: Spectrum (a): 1.91 δ (Rel.area: 3.00), 5.83 δ (Rel.area: 1.00), 7.10 δ (Rel.area: 1.00), 12.21 δ (Rel.area: 1.00).
Concept introduction:
Crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) can be easily distinguished by looking the signal for the methyl protons in these two compounds.
To explain:
Of the two 1HNMR spectra given, which belongs to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH).
b)
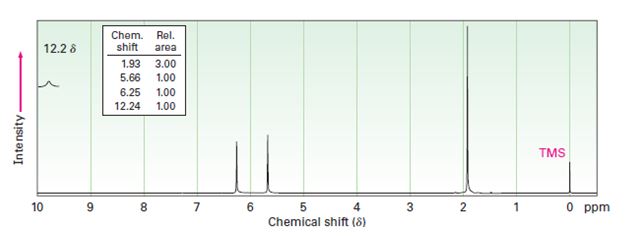
Interpretation:
Two 1HNMR spectra belonging to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) are given. Which corresponds to which is to be explained.
1HNMR: Spectrum (b): 1.93 δ (Rel.area: 3.00), 5.66 δ (Rel.area: 1.00), 6.25 δ (Rel.area: 1.00), 12.24 δ (Rel.area: 1.00).
Concept introduction:
Crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH) can be easily distinguished by looking the signal for the methyl protons in these two compounds.
To explain:
Of the two 1HNMR spectra given, which belongs to crotonic acid (trans-CH3CH=CHCOOH) and methacrylic acid (H2C=C (CH3) COOH).
Trending nowThis is a popular solution!

Chapter 20 Solutions
ORGANIC CHEMISTRY W/OWL
- What is the name of the following compound? SiMe3arrow_forwardK Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. PhCH2Br 4 57°F Sunny Q Searcharrow_forward7 Draw the starting alkyl bromide that would produce this alkyne under these conditions. F Drawing 1. NaNH2, A 2. H3O+ £ 4 Temps to rise Tomorrow Q Search H2arrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



