
Concept explainers
a)
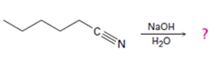
Interpretation:
The products of the reaction along with the mechanism of the reaction are to be given.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine.iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and hexanoic acid.
The mechanism of the reaction is given below.
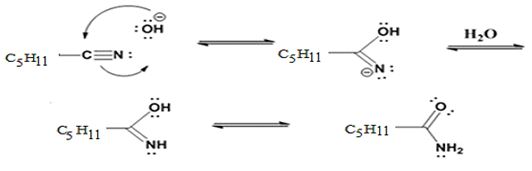
Formation of the amide:
Hydrolysis of the amide:
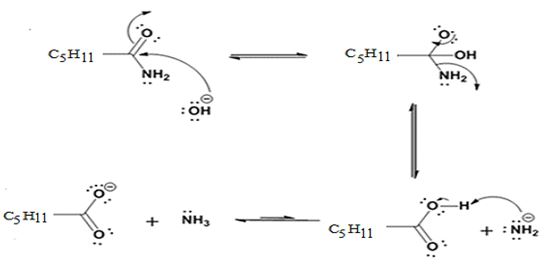
Where C5H11 = -CH2CH2CH2CH2CH3
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in hexanenitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give hexanamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields hexanoic acid. The deprotonation of the acid by the amide anion leads to the formation of carboxylate anion with the liberation of ammonia.
The products of the reaction are ammonia and hexanoic acid.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

Where C5H11 = -CH2CH2CH2CH2CH3
b)
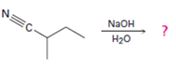
Interpretation:
The products of the reaction along with the mechanism of the reaction are to be given.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine.iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and cyclobutanecarboxylate anion.
The mechanism of the reaction is given below.
Formation of the amide:
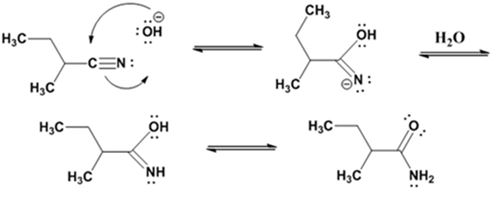
Hydrolysis of the amide:
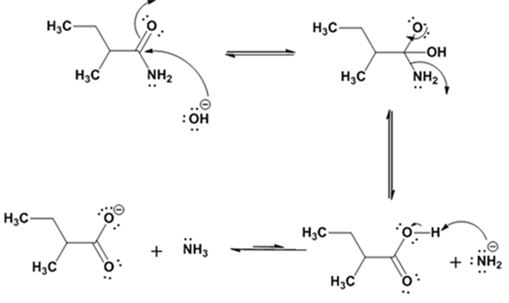
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in 2-methylbutanenitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give 2-methylbutanamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields butanoic acid. The deprotonation of the acid by the amide anion leads to the formation of carboxylate anion with the liberation of ammonia.
The products of the reaction are ammonia and cyclobutanecarboxylate anion.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

c)
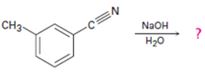
Interpretation:
The product of the reaction is to be given along with the mechanism of the reaction.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine. iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and m-methylbenzamide.
The mechanism of the reaction is given below.
Formation of the amide:
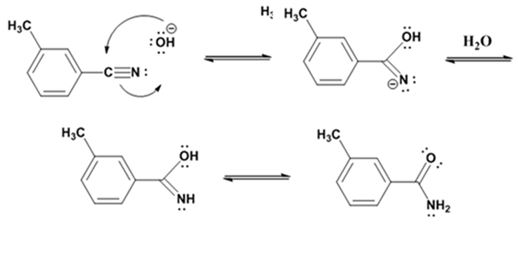
Hydrolysis of the amide:
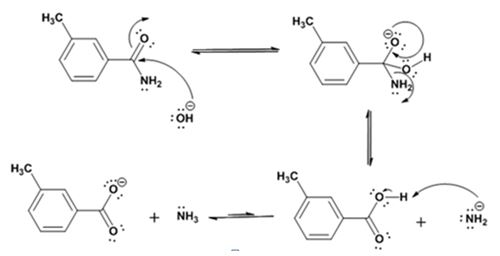
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in m-methylbenzonitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give 3-methylbenzamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields m-methylbenzoic acid. The deprotonation of the acid by the amide anion leads to the formation of the carboxylate anion with the liberation of ammonia.
The products of the reaction are ammonia and m-methylbenzamide.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

d)
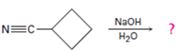
Interpretation:
The products of the reaction along with the mechanism of the reaction is to be given.
Concept introduction:
The base catalyzed nitrile hydrolysis into an amide which is then further hydrolyzed to a carboxylate anion and ammonia takes place through the following steps. i) Nucleophilic attack of the hydroxide ion to the cyanide triple bond to yield an imideanion. ii)Protonation of the imine anion by water to yield a hydroxylamine.iii) Tautomerization of the hydroxylimine to an amide iv) Hydrolysis of the amide to carboxylate anion and ammonia.
To give:
The products of the reaction along with the mechanism of the reaction.
Answer to Problem 23MP
The products of the reaction are ammonia and cyclobutanecarboxylate ion.
The mechanism of the reaction is given below.
Formation of the amide:
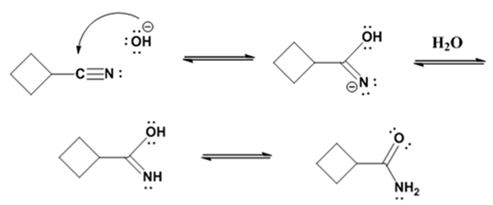
Hydrolysis of the amide:
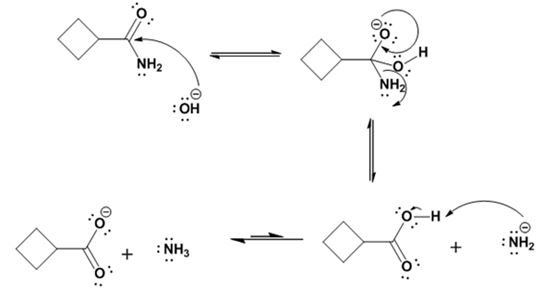
Explanation of Solution
The nucleophilic attack of the hydroxide ion the nitrile carbon in cyclobutanenitrile yields an imine anion as the product which in the next step gets protonated to produce a hydroxylimine. The hydroxylamine then tautomerizes to give cyclobutanecarboxylamide.
The nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide and subsequent loss an amide ion yields cyclobutane
The products of the reaction are ammonia and cyclobutanecarboxylate ion.
The mechanism of the reaction is given below.
Formation of the amide:

Hydrolysis of the amide:

Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY W/OWL
- Calculate the pH and the pOH of each of the following solutions at 25 °C for which the substances ionize completely: (a) 0.000259 M HClO4arrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardDetermine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. NaN₃arrow_forward
- A. Draw the structure of each of the following alcohols. Then draw and name the product you would expect to produce by the oxidation of each. a. 4-Methyl-2-heptanol b. 3,4-Dimethyl-1-pentanol c. 4-Ethyl-2-heptanol d. 5,7-Dichloro-3-heptanolarrow_forwardWhat is the pH of a 1.0 L buffer made with 0.300 mol of HF (Ka = 6.8 × 10⁻⁴) and 0.200 mol of NaF to which 0.160 mol of NaOH were added?arrow_forwardCan I please get help with this.arrow_forward
- Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction. N₂H₅ClO₄arrow_forwardPlease help me with identifying these.arrow_forwardCan I please get help with this?arrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


