
Chemistry (OER)
2nd Edition
ISBN: 9781947172616
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 41E
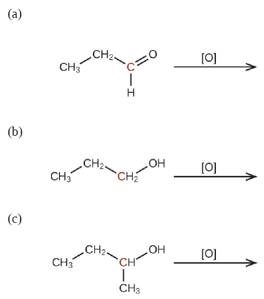
Predict the products of oxidizing the molecules shown in this problem. In each case, identify the product that will result from the minimal increase in oxidation state for the highlighted carbon atom:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
NGLISH
b)
Identify the bonds present in the molecule drawn (s) above.
(break)
State the function of the following equipments found in laboratory.
Omka)
a) Gas mask
b) Fire extinguisher
c) Safety glasses
4.
60cm³ of oxygen gas diffused through a porous hole in 50 seconds. How long w
80cm³ of sulphur(IV) oxide to diffuse through the same hole under the same conditions
(S-32.0.0-16.0)
(3 m
5.
In an experiment, a piece of magnesium ribbon was cleaned with steel w
clean magnesium ribbon was placed in a crucible and completely burnt in oxy
cooling the
product weighed 4.0g
a)
Explain why it is necessary to clean magnesium ribbon.
Masterclass Holiday assignmen
PB 2
Hi!!
Please provide a solution that is handwritten. Ensure all figures, reaction mechanisms (with arrows and lone pairs please!!), and structures are clearly drawn to illustrate the synthesis of the product as per the standards of a third year organic chemistry course. ****the solution must include all steps, mechanisms, and intermediate structures as required.
Please hand-draw the mechanisms and structures to support your explanation. Don’t give me AI-generated diagrams or text-based explanations, no wordy explanations on how to draw the structures I need help with the exact mechanism hand drawn by you!!! I am reposting this—ensure all parts of the question are straightforward and clear or please let another expert handle it thanks!!
In three dimensions, explain the concept of the velocity distribution function of particles within the kinetic theory of gases.
Chapter 20 Solutions
Chemistry (OER)
Ch. 20 - Write the chemical formula and Lewis structure of...Ch. 20 - What is the difference between the hybridization...Ch. 20 - On a microscopic level, how does the reaction of...Ch. 20 - On a microscopic level, how does the reaction of...Ch. 20 - Explain why unbranched alkenes can form geometric...Ch. 20 - Explain why these two molecules are not isomers:Ch. 20 - Explain why these two molecules are not isomers:Ch. 20 - How does the carbon-atom hybridization change when...Ch. 20 - Write the Lewis structure and molecular formula...Ch. 20 - Write the chemical formula, condensed formula, and...
Ch. 20 - Give the complete IUPAC name for each of the...Ch. 20 - Give the complete IUPAC name for each of the...Ch. 20 - Butane is used as a fuel in disposable lighters....Ch. 20 - Write Lewis structures and name the five...Ch. 20 - Write Lewis structures for the Cis -trans isomers...Ch. 20 - Write structures for the three isomers of the...Ch. 20 - Isooctane is the common name of the isomer of...Ch. 20 - Write Lewis structures and IUPAC names for the...Ch. 20 - Write Lewis structures and IUPAC names for all...Ch. 20 - Name and write the structures of all isomers of...Ch. 20 - Write the structures for all the isomers of the...Ch. 20 - Write Lewis structures and describe the molecular...Ch. 20 - Benzene is one of the compounds used as an octane...Ch. 20 - Teflon is prepared by the polymerization of...Ch. 20 - Write two complete, balanced equations for each of...Ch. 20 - Write two complete, balanced equations for each of...Ch. 20 - What mass of 2-bromopropane could be prepared from...Ch. 20 - Acetylene is a very weak acid; however, it will...Ch. 20 - Ethylene can be produced by the pyrolysis of...Ch. 20 - Why do the compounds hexane, hexanol, and hexane...Ch. 20 - Write condensed formulas and provide IUPAC names...Ch. 20 - Give the complete IUPAC name for each of the...Ch. 20 - Give the complete IUPAC name and the common name...Ch. 20 - Write the condensed structures of both isomers...Ch. 20 - Write the condensed structures of all isomers with...Ch. 20 - Draw the condensed formulas for each of the...Ch. 20 - MTBE, Methyl tert -butyl ether, CH3OC(CH3)3, is...Ch. 20 - Write two complete balanced equations for each of...Ch. 20 - Write two complete balanced equations for each of...Ch. 20 - Order the following molecules from least to most...Ch. 20 - Predict the products of oxidizing the molecules...Ch. 20 - Predict the products of reducing the following...Ch. 20 - Explain why it is not possible to possible a...Ch. 20 - How does hybridization of the substituted carbon...Ch. 20 - Fatty acids are carboxylic acids that have long...Ch. 20 - Write a condensed structural formula, such as...Ch. 20 - Write a condensed structural formula, such as...Ch. 20 - The foul odor of rancid butter is caused by...Ch. 20 - Write the two-resonance structures for the acetate...Ch. 20 - Write two complete, balanced equations for each of...Ch. 20 - Write two complete balanced equations for each of...Ch. 20 - Yields in organic reactions are sometimes low....Ch. 20 - Alcohols A, B and C all have the composition C4H...Ch. 20 - Write the Lewis structures of both isomers with...Ch. 20 - What is the molecular structure about the nitrogen...Ch. 20 - Write the two resonance structures for the...Ch. 20 - Draw Lewis structures for pyridine and its...Ch. 20 - Write the Lewis structures of all isomers with the...Ch. 20 - Write two complete balanced equations for the...Ch. 20 - Write two complete, balanced equations for each of...Ch. 20 - Identify any carbon atoms that change...Ch. 20 - Identify any carbon atoms that change...Ch. 20 - Identify any carbon atoms that change...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. Why is the quantum-mechanical model of the atom important for understanding chemistry?
Chemistry: Structure and Properties (2nd Edition)
18. SCIENTIFIC THINKING By measuring the fossil remains of Homo floresiensis, scientists have estimated its wei...
Campbell Biology: Concepts & Connections (9th Edition)
With respect to angiosperms, which of the following is incorrectly paired with its chromosome count? (A) eggn (...
Campbell Biology (11th Edition)
Consider the balanced equation: 2N2H4(g)+N2O4(g)3N2(g)+4H2O(g) Complete the table with the appropriate number o...
Introductory Chemistry (6th Edition)
Using the pKa values listed in Table 15.1, predict the products of the following reactions:
Organic Chemistry (8th Edition)
1. Define and distinguish incomplete penetrance and variable expressivity.
Genetic Analysis: An Integrated Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In the kinetic theory of gases, explain the concept of the velocity distribution function of particles in space.arrow_forwardIn the kinetic theory of gases, explain the concept of the velocity distribution function of particles.arrow_forwardHi!! Please provide a solution that is handwritten. this is an inorganic chemistry question please answer accordindly!! its just one question with parts JUST ONE QUESTION with its parts spread out till part (g), please answer EACH part till the end and dont just provide wordy explanations wherever asked for structures, please DRAW DRAW them on a paper and post clearly!! answer the full question with all calculations step by step EACH PART CLEARLY please thanks!! im reposting this please solve all parts and drawit not just word explanations!!arrow_forward
- Hi!! Please provide a solution that is handwritten. this is an inorganic chemistry question please answer accordindly!! its just one question with parts JUST ONE QUESTION, please answer EACH part PART A AND PART B!!!!! till the end and dont just provide wordy explanations wherever asked for structures, please DRAW DRAW them on a paper and post clearly!! answer the full question with all details EACH PART CLEARLY please thanks!! im reposting this please solve all parts and drawit not just word explanations!!arrow_forwardHi!! Please provide a solution that is handwritten. this is an inorganic chemistry question please answer accordindly!! its just one question with parts JUST ONE QUESTION, please answer EACH part till the end and dont just provide wordy explanations wherever asked for structures, please DRAW DRAW them on a paper and post clearly!! answer the full question with all details EACH PART CLEARLY please thanks!! im reposting this please solve all parts and drawit not just word explanations!!arrow_forward8b. Explain, using key intermediates, why the above two products are formed instead of the 1,2-and 1,4- products shown in the reaction below. CIarrow_forward
- (5pts) Provide the complete arrow pushing mechanism for the chemical transformation depicted below Use proper curved arrow notation that explicitly illustrates all bonds being broken, and all bonds formed in the transformation. Also, be sure to include all lone pairs and formal charges on all atoms involved in the flow of electrons. CH3O H I I CH3O-H H I ① Harrow_forward6. Draw the products) formed from the following reactions. (a) HIarrow_forwardDon't used Ai solutionarrow_forward
- Please correct answer and don't used hand raitingarrow_forward1. For each of the following, predict the products of the reaction by writing a balance net ionic equation for each. If no reaction is expected, then write NO REACTION. (a) AgNO3 (aq) is mixed with Na2CO3 (aq). (b) An aqueous solution of ammonium sulfate is added to an aqueous solution of calcium chloride. (c) RbI (aq) is added to Pb(NO3)2 (aq). (d) NaCl (s) is added to AgNO3 (aq).arrow_forward4. Determine the amount in grams of AgCl (s) formed when 2.580 g AgNO3(s) is added to 45.00 mL of a 0.1250 M CrCl3 (aq) (The other product is aqueous chromium (III) nitrate) 5. Determine the amount (in grams) of Cobalt (II) phosphate formed when an aqueous solution of 30.0 ml of 0.450 M Sodium Phosphate is mixed with 20.0 mL of 0.500 M aqueous solution of cobalt (II) nitrate. (The other product is aqueous sodium nitrate)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENTChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENTChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning