
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 34P
Give the IUPAC or common name for each compound.
a.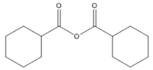 c.
c. 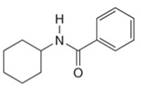 e.
e. 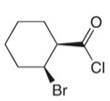
b.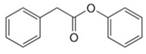 d.
d. 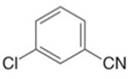 f.
f. 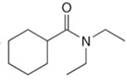
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Seee the attached ima
Please see the attached image.
Please see the attached image.
Chapter 20 Solutions
Organic Chemistry (6th Edition)
Ch. 20.1 - Prob. 1PCh. 20.2 - Draw the three possible resonance structures for...Ch. 20.2 - Prob. 3PCh. 20.3 - Give an IUPAC or common name for each compound. a....Ch. 20.3 - Problem 22.5 Draw the structure corresponding to...Ch. 20.4 - Problem 22.6 Explain why the boiling point of is...Ch. 20.7 - Prob. 11PCh. 20.7 - Prob. 12PCh. 20.8 - Prob. 13PCh. 20.9 - Problem 22.16 Draw the products of each reaction.
...
Ch. 20.9 - Prob. 17PCh. 20.9 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 20.9 - Prob. 19PCh. 20.10 - Problem 22.20 Fenofibrate is a...Ch. 20.10 - Problem 22.21 What product is formed when the...Ch. 20 - Prob. 33PCh. 20 - 22.40 Give the IUPAC or common name for each...Ch. 20 - 22.41 Give the structure corresponding to each...Ch. 20 - Prob. 36PCh. 20 - 22.43 Explain why is a stronger acid and a weaker...Ch. 20 - (a) Propose an explanation for the difference in...Ch. 20 - Draw the product formed when phenylacetic acid is...Ch. 20 - Prob. 42PCh. 20 - Prob. 43PCh. 20 - Prob. 44PCh. 20 - Prob. 45PCh. 20 - Prob. 46PCh. 20 - Prob. 47PCh. 20 - Prob. 48PCh. 20 - 22.64 What carboxylic acid and alcohol are needed...Ch. 20 - Problem 22.65 Devise a synthesis of each compound...Ch. 20 - 22.70 What polyester or poly amide can be prepared...Ch. 20 - 22.71 What two monomers are needed to prepare each...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- V Biological Macromolecules Drawing the Haworth projection of an aldose from its Fischer projection Draw a Haworth projection of a common cyclic form of this monosaccharide: H C=O HO H HO H H OH CH₂OH Explanation Check Click and drag to start drawing a structure. Xarrow_forwardComplete the mechanismarrow_forwardComplete the mechanismarrow_forward
- 8 00 6 = 10 10 Decide whether each of the molecules in the table below is stable, in the exact form in which it is drawn, at pH = 11. If you decide at least one molecule is not stable, then redraw one of the unstable molecules in its stable form below the table. (If more than unstable, you can pick any of them to redraw.) Check OH stable HO stable Ounstable unstable O OH stable unstable OH 80 F6 F5 stable Ounstable X Save For Later Sub 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C ཀྭ་ A F7 매 F8 F9 4 F10arrow_forwardJust try completing it and it should be straightforward according to the professor and TAs.arrow_forwardThe grading is not on correctness, so if you can just get to the correct answers without perfectionism that would be great. They care about the steps and reasoning and that you did something. I asked for an extension, but was denied the extension.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY