
(a)
Interpretation:
A synthesis of the given compound by using acetyl chloride is to be determined.
Concept introduction:
Sodium benzoate is the salt of conjugate base or sodium salt of benzoic acid. When acyl halide reacts with sodium benzoate, it gives the ester. Sodium benzoate acts as the nucleophile. Nucleophile is the negatively charged ion attacks the carbonyl carbon and expels the good leaving group.
Answer to Problem 20.65P
The way of synthesis of the given compound by using acetyl chloride is as follows:
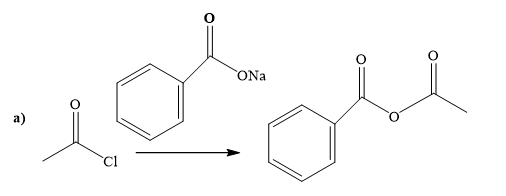
Explanation of Solution
The way of synthesis of the given compound by using acetyl chloride is as follows:

Sodium benzoate is a sodium salt of benzoic acid. When acyl halide reacts with sodium benzoate, it gives the ester. Sodium benzoate, as s nucleophile, is used to undergo nucleophilic addition- elimination reaction with acid chloride. Benzoate’s negatively charged ion attacks the carbonyl carbon of acetyl chloride and expels the good leaving group chlorine.
From the structure of the given reactant and product, structure of the reagent is determined.
(b)
Interpretation:
A synthesis of the given compound by using the acetyl chloride is to be determined.
Concept introduction:
Sodium phenoxide is produced by sodium hydroxide and Phenol. Ester can be prepared by treating acetyl chloride with phenoxide. Sodium phenoxide is a moderately strong base.
Answer to Problem 20.65P
The way of synthesis of the given compound by using acetyl chloride is as follows:
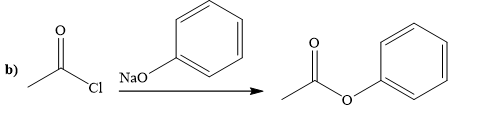
Explanation of Solution
The way of synthesis of the given compound by using acetyl chloride is as follows:

Ester can be prepared by treating acetyl chloride with sodium phenoxide. Sodium phenoxide, as a nucleophile, is used to undergo nucleophilic addition- elimination reaction with acid chloride. Phenoxide, the negatively charged ion attacks the carbonyl carbon of acetyl chloride and expels the good leaving group chlorine and forms ester.
From the structure of given reactant and product, structure of reagent is determined.
(c)
Interpretation:
A synthesis of the given compound by using acetyl chloride is to be determined.
Concept introduction:
It converts the acid chloride or anhydrides to primary alcohol. It’s also used for reduction of carbonyl compound to corresponding alcohol, and after removing the hydrogen from alcohol, it gives alkoxide. Alkoxide ion attacks the carbonyl carbon of the acid chloride and expels the good leaving group and forms the ester as product.
Answer to Problem 20.65P
The way of synthesis of the given compound by using acetyl chloride is as follows:
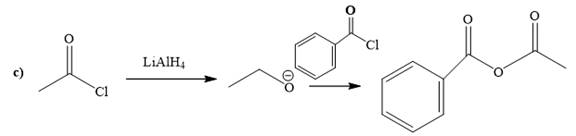
Explanation of Solution
The way of synthesis of the given compound by using acetyl chloride is as follows:

Acetyl halide is converted to alkoxide by treating it with
From the structure of the given reactant and product, structure of reagent is determined.
(d)
Interpretation:
A synthesis of the given compound by using acetyl chloride is to be determined.
Concept introduction:
It is also used for reduction of amides to corresponding
Answer to Problem 20.65P
The way of synthesis of given compound by using acetyl chloride is as follows:
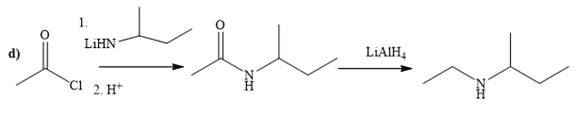
Explanation of Solution
The way of synthesis of given compound by using acetyl chloride is as follows:

In the first step, acid chloride converts to amide in the presence of organolithium amine reagent. Amide is reduced with
From the structure of given reactant and product, structure of reagent is determined.
(e)
Interpretation:
A synthesis of given compound by using the acetyl chloride is to be determined.
Concept introduction:
It converts the acid chloride or anhydrides to primary alcohol. It’s also used for reduction of carbonyl a compound to corresponding alcohol. And after remove the hydrogen from alcohol to get alkoxide. Alkoxide ion attacks the carbonyl carbon of the acid chloride and expels the good leaving group and form the ester as product. Alcohol is reacting with strong acid at required temperature to get the ester.
Answer to Problem 20.65P
The way of synthesis of given compound by using acetyl chloride is as follows:
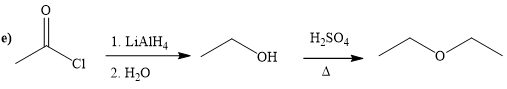
Explanation of Solution
The way of synthesis of given compound by using acetyl chloride is as follows:

In the first step, acetyl chloride is converted to
From the structure of given reactant and product, structure of reagent is determined.
(f)
Interpretation:
A synthesis of given compound by using acetyl chloride is to be determined.
Concept introduction:
The reagent
Answer to Problem 20.65P
The way of synthesis of given compound by using acetyl chloride is as follows:
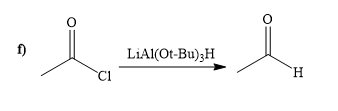
Explanation of Solution
The way of synthesis of given compound by using acetyl chloride is as follows:

When acyl chloride react with
From the structure of given reactant and product, structure of reagent is determined.
(g)
Interpretation:
A synthesis of given compound by using acetyl chloride is to be determined.
Concept introduction:
Acid chloride reacts with Gilmann reagent to give the
Answer to Problem 20.65P
The way of synthesis of given compound by using acetyl chloride is as follows:
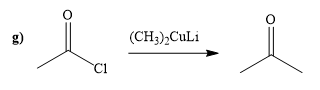
Explanation of Solution
The way of synthesis of given compound by using acetyl chloride is as follows:
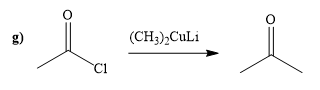
Acetyl chloride is converted to ketone in the presence lithium dimethylcuprate. Ketone does not undergo nucleophilic attack by Gilmann reagent as the reagent is a soft nucleophile.
From the structure of given reactant and product, structure of reagent is determined.
(h)
Interpretation:
A synthesis of given compound by using the acetyl chloride is to be determined.
Concept introduction:
Acid chloride is converted to ester in presence of sodium alkoxide. Alkoxide ion attacks the carbonyl carbon of acid chloride and expels the good leaving group forming ester as the product.
Answer to Problem 20.65P
The way of synthesis of given compound by using acetyl chloride is as follows:
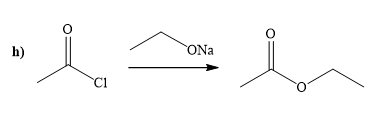
Explanation of Solution
The way of synthesis of given compound by using acetyl chloride is as follows:

Acetyl chloride is converted to ester in presence of sodium ethoxide. Ethoxide ion attacks the carbonyl carbon of acid chloride and expels the good leaving group, forming the ester as a product.
From the structure of given reactant and product, structure of reagent is determined.
Want to see more full solutions like this?
Chapter 20 Solutions
EBK ORGANIC CHEMISTRY: PRINCIPLES AND M
- Draw the stepwise mechanism for the reactionsarrow_forwardPart I. a) Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone b) Pinacol (2,3-dimethyl, 1-3-butanediol) on treatment w/ acid gives a mixture of pina colone (3,3-dimethyl-2-butanone) and 2, 3-dimethyl - 1,3-butadiene. Give reasonable mechanism the formation of the products Forarrow_forward3. The explosive decomposition of 2 mole of TNT (2,4,6-trinitrotoluene) is shown below: Assume the C(s) is soot-basically atomic carbon (although it isn't actually atomic carbon in real life). 2 CH3 H NO2 NO2 3N2 (g)+7CO (g) + 5H₂O (g) + 7C (s) H a. Use bond dissociation energies to calculate how much AU is for this reaction in kJ/mol.arrow_forward
- Part I. Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff: Pinacol (2,3-dimethyl, 1-3-butanediol) on treatment w/ acid gives a mixture of pina colone and (3,3-dimethyl-2-butanone) 2,3-dimethyl-1,3-butadiene. Give reasonable mechanism the formation of the products Forarrow_forwardShow the mechanism for these reactionsarrow_forwardDraw the stepwise mechanismarrow_forward
- Draw a structural formula of the principal product formed when benzonitrile is treated with each reagent. (a) H₂O (one equivalent), H₂SO₄, heat (b) H₂O (excess), H₂SO₄, heat (c) NaOH, H₂O, heat (d) LiAlH4, then H₂Oarrow_forwardDraw the stepwise mechanism for the reactionsarrow_forwardDraw stepwise mechanismarrow_forward
- Part I. Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff: a) Give the major reason for the exposure of benzophenone al isopropyl alcohol (w/acid) to direct sunlight of pina colone Mechanism For b) Pinacol (2,3-dimethy 1, 1-3-butanediol) on treatment w/ acid gives a mixture (3,3-dimethyl-2-butanone) and 2, 3-dimethyl-1,3-butadiene. Give reasonable the formation of the productsarrow_forwardwhat are the Iupac names for each structurearrow_forwardWhat are the IUPAC Names of all the compounds in the picture?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





