
Pearson eText Organic Chemistry -- Instant Access (Pearson+)
8th Edition
ISBN: 9780135213711
Author: Paula Bruice
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 64P
For each compound, indicate the atom that is most apt to be protonated.
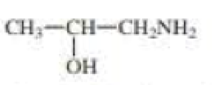
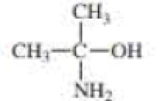
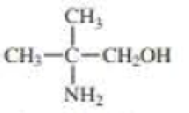
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
If a high molecular weight linear polyethylene is chlorinated by inducing the substitution of chlorine atoms by hydrogen, if 5% of all hydrogen atoms are replaced, what approximate percentage of chlorine by weight would the product have?
O Macmillan Learning
Chemistry: Fundamentals and Principles
Davidson
presented by Macmillan Learning
Poly(ethylene terephthalate), known as PET or industrially as Dacron, is a polyester synthesized through a condensation reaction
between two bifunctional monomers. The monomers, ethylene glycol and terepthalic acid, are given.
Add bonds and remove atoms as necessary to show the structure of a two repeat unit portion of a longer polymer chain
of PET. You may need to zoom out to see the complete structure of all four monomer units.
Select
Draw
/ || |
C
H
0
3
©
Templates
More
°
°
°
||
C
CC - OH
HO
OH
HOC - C
Erase
CC OH
HO C
C
〃
C
H₂
Q2Q
c)
+
H₂O
Chapter 2 Solutions
Pearson eText Organic Chemistry -- Instant Access (Pearson+)
Ch. 2.1 - Which of the following are not acids? CH3COOH CO2...Ch. 2.1 - Consider the following reaction: a. What is the...Ch. 2.1 - Draw the products of the addbase renc1 ion when a....Ch. 2.1 - a. What is the conjugate acid of each or the...Ch. 2.2 - a. Which is a stronger acid: one with a pKa of 5.2...Ch. 2.2 - An acid has a Ka of 4.53 106 in water. What is...Ch. 2.2 - Prob. 7PCh. 2.2 - Antacids are compounds that neutralize stomach...Ch. 2.2 - Are the following body fluids acidic or basic? a....Ch. 2.3 - Draw the conjugate acid of each of the following:...
Ch. 2.3 - a. Write an equation showing CH3OH reacting as an...Ch. 2.3 - Estimate the pKa values of the following...Ch. 2.3 - a. Which is a stronger base: CH3COO or HCOO? (The...Ch. 2.3 - Using the pKa values in Section 2.3, rank the...Ch. 2.4 - Prob. 15PCh. 2.5 - a. For each of the acid-base reactions in Section...Ch. 2.5 - Ethyne has a pKa value of 25, water has a pKa...Ch. 2.5 - Which of the following bases can remove a proton...Ch. 2.5 - Calculate the equilibrium constant for the...Ch. 2.6 - Rank the ions (CH3, NH2, HO, and F) from most...Ch. 2.6 - Rank the carbanions shown in the margin from most...Ch. 2.6 - Which is the stronger acid?Ch. 2.6 - Prob. 23PCh. 2.6 - What reaction in Problem 23 has the smallest...Ch. 2.6 - Rank the halide ions (F, Cl, Br, and l) from...Ch. 2.6 - a. Which is more electronegative, oxygen or...Ch. 2.6 - Which is a stronger acid? a. HCl or HBr b....Ch. 2.6 - a. Which of the halide ions (F, Cl, Br, and l) is...Ch. 2.6 - Which is a stronger base? (The potential maps in...Ch. 2.7 - What is a stronger acid? a. CH3OCH2CH2OH or...Ch. 2.7 - Rank the following compounds from strongest add to...Ch. 2.7 - What is a stronger base?Ch. 2.8 - For each of the following compounds, indicate the...Ch. 2.8 - Prob. 35PCh. 2.8 - Which is a stronger acid? Why?Ch. 2.8 - Fosamax (shown on the previous page) has six...Ch. 2.9 - Using the table of pKa values given in Appendix I,...Ch. 2.10 - For each of the following compounds (here shown in...Ch. 2.10 - As long as the pH is not less than _______, at...Ch. 2.10 - a. Indicate whether a protonated amine (RN+H3)...Ch. 2.10 - A naturally occurring amino acid such as alanine...Ch. 2.10 - a. At what pH is the concentration of a compound,...Ch. 2.10 - For each of the following compounds, indicate the...Ch. 2.10 - Given the data in Problem 47: a. What pH would you...Ch. 2.11 - Write the equation that shows how a buffer made by...Ch. 2.12 - Draw the products of the following react ions. Use...Ch. 2.12 - What product are formed when each of the following...Ch. 2 - Which is a stronger base? a. HS or HO b. CH3O or...Ch. 2 - According to the explanations by Lewis, if a...Ch. 2 - a. Rank the following alcohols from strongest to...Ch. 2 - a. Rank the following carboxylic acids from...Ch. 2 - Prob. 57PCh. 2 - For the following compound. a. draw its conjugate...Ch. 2 - Rank the following compounds from strongest to...Ch. 2 - Prob. 60PCh. 2 - Prob. 61PCh. 2 - a. Rank the following alcohols from strongest to...Ch. 2 - A single bond between two carbons with different...Ch. 2 - For each compound, indicate the atom that is most...Ch. 2 - a. Given the Ka values, estimate the pKa value of...Ch. 2 - Tenormin, a member of the group of drugs known as...Ch. 2 - From which of the following compounds can HO...Ch. 2 - a. For each of the following pairs of reactions,...Ch. 2 - Prob. 69PCh. 2 - Which is a stronger acid? a. b. c. d.Ch. 2 - Prob. 71PCh. 2 - Prob. 72PCh. 2 - Given that pH+ pOH = 14 and that the concentration...Ch. 2 - How could you separate a mixture of the following...Ch. 2 - Prob. 75PCh. 2 - a. If an add with a pKa of 5.3 is in an aqueous...Ch. 2 - Calculate the pH values of the following...Ch. 2 - Prob. 1PCh. 2 - Prob. 2PCh. 2 - Prob. 3PCh. 2 - Which of the reactions in Problem 3 favor...Ch. 2 - Prob. 5PCh. 2 - Prob. 6PCh. 2 - Prob. 7PCh. 2 - Which is the stronger acid? a. ClCH2CH2OH or...Ch. 2 - Prob. 9PCh. 2 - Prob. 10PCh. 2 - Which is a more stable base? Remembering that the...Ch. 2 - Which is the Stronger acid?Ch. 2 - Prob. 13PCh. 2 - a. Draw the structure of (CH3COOH (pKa = 4.7) at...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 으 b) + BF. 3 H2Oarrow_forwardQ4: Draw the product of each Lewis acid-bas reaction. Label the electrophile and nucleophile. b) S + AICI 3 + BF 3arrow_forwardQ1 - What type(s) of bonding would be expected for each of the following materials: solid xenon, calcium fluoride (CaF2), bronze, cadmium telluride (CdTe), rubber, and tungsten? Material solid xenon CaF2 bronze CdTe rubber tungsten Type(s) of bonding Q2- If the atomic radius of lead is 0.175 nm, calculate the volume of its unit cell in cubic meters.arrow_forward
- Determine the atomic packing factor of quartz, knowing that the number of Si atoms per cm3 is 2.66·1022 and that the atomic radii of silicon and oxygen are 0.038 and 0.117 nm.arrow_forwardUse the following data for an unknown gas at 300 K to determine the molecular mass of the gas.arrow_forward2. Provide a complete retrosynthetic analysis and a complete forward synthetic scheme to make the following target molecule from the given starting material. You may use any other reagents necessary. Brarrow_forward
- 146. Use the following data for NH3(g) at 273 K to determine B2p (T) at 273 K. P (bar) 0.10 0.20 0.30 0.40 0.50 0.60 (Z -1)/10-4 1.519 3.038 4.557 6.071 7.583 9.002 0.70 10.551arrow_forward110. Compare the pressures given by (a) the ideal gas law, (b) the van der Waals equation, and (c) the Redlic-Kwong equation for propane at 400 K and p = 10.62 mol dm³. The van der Waals parameters for propane are a = 9.3919 dm6 bar mol-2 and b = 0.090494 dm³ mol−1. The Redlich-Kwong parameters are A = 183.02 dm bar mol-2 and B = 0.062723 dm³ mol-1. The experimental value is 400 bar.arrow_forwardResearch in surface science is carried out using stainless steel ultra-high vacuum chambers with pressures as low as 10-12 torr. How many molecules are there in a 1.00 cm3 volume at this pressure and at a temperature of 300 K? For comparison, calculate the number of molecules in a 1.00 cm3 volume at atmospheric pressure and room temperature. In outer space the pressure is approximately 1.3 x 10-11 Pa and the temperature is approximately 2.7 K (determined using the blackbody radiation of the universe). How many molecules would you expect find in 1.00 cm3 of outer space?arrow_forward
- Draw the predominant form of arginine at pH = 7.9. The pKa of the side chain is 12.5. Include proper stereochemistry. H2N OH NH H₂N 'N' છ H pH = 7.9 Select to Drawarrow_forwardPlease correct answer and don't used hand raitingarrow_forward142. A mixture of H2(g) and N2(g) has a density of 0.216 g/liter at 300 K and 500 torr. What is the mole fraction composition of the mixture?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning



Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY