
Concept explainers
(a)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
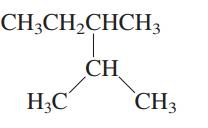
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“ and “neo-“ .For example isobutane is common name used popularly for
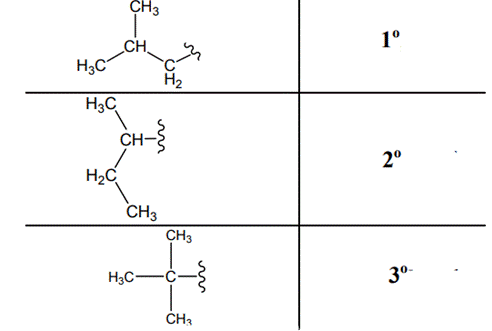
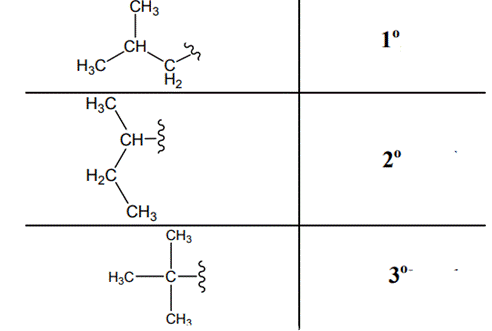
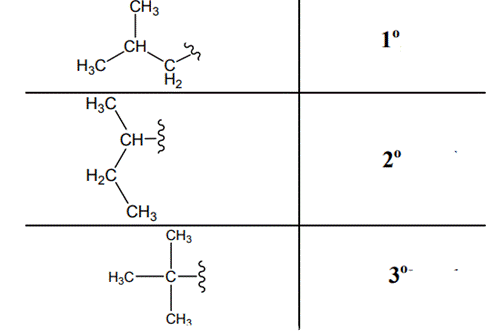
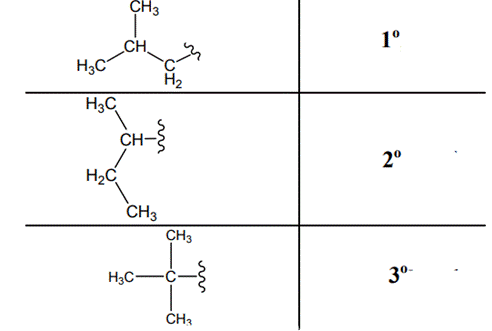
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(b)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
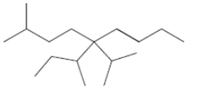
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“ and “neo-“ .For example isobutane is common name used popularly for
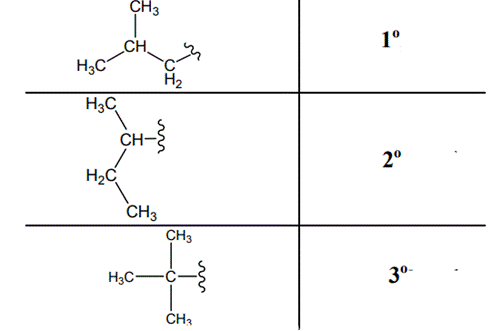
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(c)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
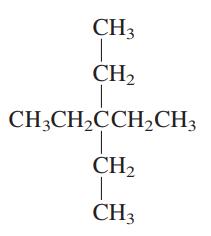
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“ and “neo-“ .For example isobutane is common name used popularly for
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(d)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
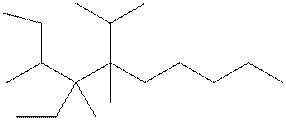
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
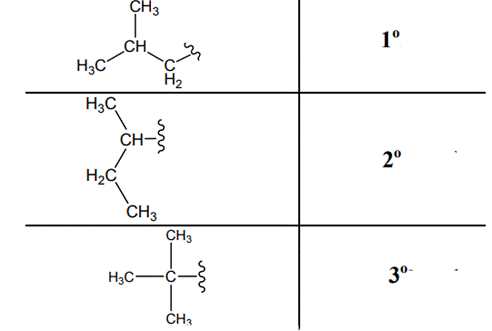
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(e)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
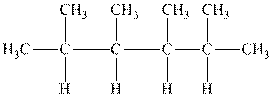
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(f)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
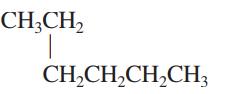
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
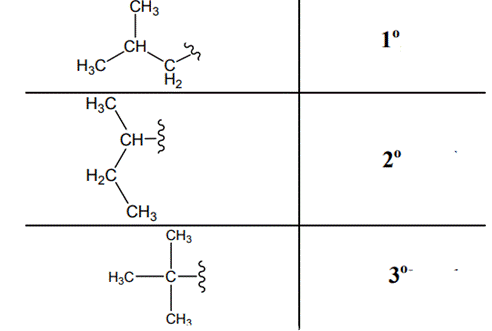
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(g)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
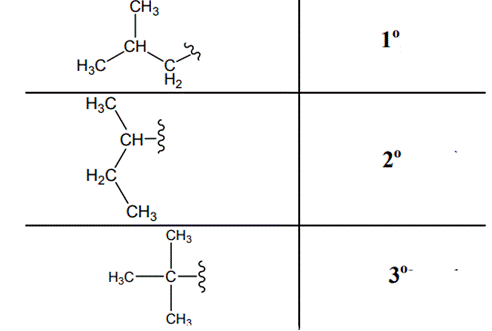
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(h)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
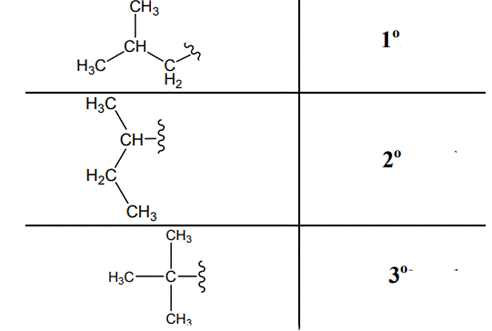
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(i)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
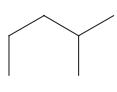
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated below:
(j)
Interpretation: The following molecule should be named according to the IUPAC system of nomenclature:
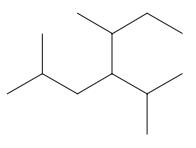
Concept introduction: In accordance with IUPAC convention longest chain can be found from either direction provided it is longest and digits indicate the position of carbon or the position of branched alkyl chain in cases of branched hydrocarbons. All the side chains are named in alphabetical order.
The IUPAC system for nomenclature of straight hydrocarbon makes use of table given as follows:
Beside the IUPAC names there are certain common names. The common prefixes used include prefixes” iso-“and “neo-“.For example isobutane is common name used popularly for
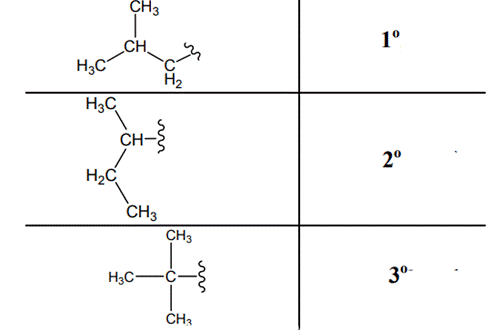
The carbon linked to one alkyl / carbon while other two
The carbon linked to two alkyl /carbons and one
The carbon linked to three alkyl groups/carbons and no
These are indicated as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
EBK STUDY GUIDE/SOLUTIONS MANUAL FOR OR
- Can I please get help with this?arrow_forward1. Draw structures corresponding to each of the following names [3 ONLY]: A. 2,2,2-trichloroethanal (chloral). B. trans-3-isopropylcyclohexanecarbaldehyde C. What is the correct structure for 2-hydroxyacetophenone? Circle the letter of your response. a C 0 OH OH OH HO b. H3C CH 0 H d OH D. Provide IUPAC names for each structure below. 0 H C-H 0 0 CH3 H NO₂ E. The substance formed on addition of water to an aldehyde or ketone is called a hydrate or a/an: a. vicinal diol b. geminal diol C. acetal d. ketalarrow_forwardAssign this spectrumarrow_forward
- Redraw the tripeptide with or without its acidic hydrogensto demonstrate where the total charge of -2 comes from: *see imagearrow_forward2. Consider the data below to answer the following questions. Cyanohydrins are important intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The nitrile functional group can be hydrolyzed by aqueous acid to yield a carboxylic acid. Nitriles can also be hydrolyzed to carboxylic acids using aqueous base. Unfortunately, when a cyanohydrin is treated with aqueous base the original carbonyl compound is isolated. OH CH-COOH 0 HO CN C H30* C. H H HC N NaOH H₂O C=O 0 cyanohydrin H + NaCN + H₂Oarrow_forwardAssign all integrated peaksarrow_forward
- - Consider the data in the Table below to answer the following questions: Acidities of Substituted Benzoic and Acetic Acids pk,s at 25C Y-CH COOH Y Y - CH₂COOH meta para H 4.75 4.19 4.19 2.47 3.64 3.55 3.57 4.09 4.46 CN OCH 3 A. Draw the structure of the strongest acid in the table above. B. Explain why cyanoacetic acid and methoxyacetic acid are more acidic than their correspondingly substituted benzoic acid counterparts.arrow_forwardDraw the curved arrow mechanism for this reaction starting with 2-propanol in sulfuric acid. Show all nonzero formal charges and all nonbonded electrons in each step. Species not involved in a particular step do not need to be included in that step, and resonance forms do not need to be shown. Note that the alcohol is in much higher concentration than H₂O in this reaction. Harrow_forwardProvide reactions showing the following conversions: * see imagearrow_forward
- . Draw structures corresponding to each of the following names or Provide IUPAC names for each of the ollowing structures [for 4 ONLY]. A. 2-propylpentanoic acid. B. m-chlorobenzoic acid. D. C. O O HOC(CH2)3COH glutaricadd OH OH H3C CH3 C=C H COOH salicylicadd tiglicadd CH₂C=N Joe Marrow_forward. Provide structure(s) for the starting material(s), reagent(s) or the major organic product(s) of each of the ollowing reactions or sequences of reactions. Show all relevant stereochemistry [five only] A. O B. OET CH3 1. LIAIH, ether 2 H₂O O (CH3)2CH-C-CI + 0 0 ether (CH3)2CH-C-O-C-CH3 CH3 C. 0 OH HO CH3 ° Clarrow_forwardHow would you prepare each of the following compounds using either an acetoacetic ester synthesis or a alonic ester synthesis? Show all intermediate structures and all reagents.[Three only] A. B. COOH OH C. D. 0 H2C CHCH2CH2CCH3arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning



