
Concept explainers
(a)
To identify: The potential hydrogen bond donors and accepters present in Fig.1.
Concept introduction: Hydrogen bonds are formed between polar molecules. It is an intermolecular attraction that forms between partially positive hydrogen atoms of a polar molecule with a partially negative atom of another polar molecule. In general chemical formulation, a hydrogen bond is explained as D–H···A, where D–H is a hydrogen bond “donor group” and A is considered as a hydrogen bond accepter group or an atom.
(a)
Answer to Problem 1E
Correct answer: The potential hydrogen bond donor groups are HN1, H2N at the C2 position, and HN9. The potential hydrogen bond accepter groups are O at the C6 position, N3, and N7.
Explanation of Solution
Pictorial presentation: Fig. 1 shows structure of guanine, where the potential hydrogen bond donors and accepters are identified.
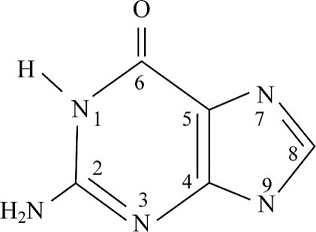
Fig.1: Guanine
The given formula of Fig.1 is identified as Guanine. Here, at the HN1 position, the hydrogen bond is donated and nitrogen acts as a hydrogen bond donor when it is paired with a hydrogen atom. When nitrogen is not paired with a hydrogen atom, it acts as a hydrogen accepter. Therefore, at HN1, H2N at the C2 position, and HN9 portion, the hydrogen bond acts like a donor; O at the C6 position, N3, and N7 portion, the hydrogen bond acts like an acceptor.
(b)
To identify: The potential hydrogen bond donors and acceptors present in Fig.2.
Concept introduction: Hydrogen bonds are formed between polar molecules. It is an intermolecular attraction that forms between partially positive hydrogen atoms of a polar molecule with a partially negative atom of another polar molecule. In general chemical formulation, a hydrogen bond is explained as D–H···A, where D–H is a hydrogen bond “donor group” and A is considered as a hydrogen bond accepter group or atom.
(b)
Answer to Problem 1E
Correct answer: The potential hydrogen bond donor groups are HN1 and H2N at the C4 position. The potential hydrogen bond accepter groups are O at the C2 position, N3.
Explanation of Solution
Pictorial presentation: Fig.2 shows structure of cytosine, where the potential hydrogen bond donors and accepters are identified.
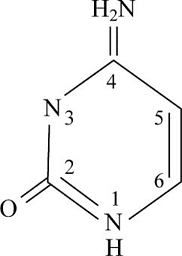
Fig.1: Cytosine
The given formula of Fig.2 is identified as cytosine. Here, at HN1 position, the hydrogen bond is donated as nitrogen acts as the hydrogen bond donor when it is paired with a hydrogen atom. When nitrogen is not paired with a hydrogen atom, it acts as a hydrogen acceptor. Therefore, at HN1 and H2N at the C4 position, the hydrogen bond acts like a donor and O at the C2 position and N3 portion, the hydrogen bond acts like an acceptor.
(c)
To identify: The potential hydrogen bond donors and accepters present in Fig.3.
Concept introduction: Hydrogen bonds are formed between the polar molecules. It is an intermolecular attraction that forms between partially positive hydrogen atoms of a polar molecule with a partially negative atom of another polar molecule. In general chemical formulation, a hydrogen bond is explained as D–H···A, where D–H is a hydrogen bond “donor group” and A is considered as a hydrogen bond accepter group or atom.
(c)
Answer to Problem 1E
Correct answer: The potential hydrogen bond donor groups are H3N+ group and OH group. The potential hydrogen bond accepter groups are COO and OH.
Explanation of Solution
Pictorial presentation: Fig.3 shows structure of serine, where the potential hydrogen bond donors and acceptors are identified.
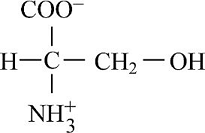
Fig.3: Serine
The given formula of Fig.3 is identified as serine. Here, at H3N+ position, the hydrogen bond is donated as nitrogen acts as a hydrogen bond donor when it is paired with a hydrogen atom. When nitrogen is not paired with a hydrogen atom, it acts as a hydrogen acceptor. Therefore, at H3N+ group and OH group, the hydrogen bond acts like a donor and COO and OH position, the hydrogen bond acts like an acceptor.
Want to see more full solutions like this?
Chapter 2 Solutions
Fundamentals of Biochemistry: Life at the Molecular Level
- 9. Which one of the compounds below is a major final product of the reaction sequence shown at the right? A) para-bromonitrobenzene C) meta-bromoaniline B) meta-bromonitrobenzene D) para-bromoaniline 1. HNO3, H2SO4 2. Br₂, FeBr3 3. H₂/Ni (3 atm) E) ortho-bromoanilinearrow_forward10. This reaction sequence includes an intramolecular Friedel-Crafts reaction. Which of the compounds below is expected to be the major product? PhCH2CH2CH2COOH 4-phenylbutanoic acid SOCI₂ AICI 3 A B C D Earrow_forward5. Which one is the major organic product obtained from the following reaction sequence? A B C OH i 1. NaBH4 CI 2. H₂O, H+ AICI 3 D OH Earrow_forward
- 1. Which one is the major organic product obtained from the reaction of toluene and cyclopentanol in the presence of H3PO4, as shown here? CH3 CH3 CH3 CH3 CH3 H3PO4 A B с D E OHarrow_forwardAscorbic acid is a diprotic, with ionizations of: pKa1 = 4.10; pKa2 =11.80. You need to make 350 mL of an ascorbate buffer that is pH 5.05, andyou have 1.5 mM stock solutions of :ascorbic acidmonosodium ascorbatedipotassium ascorbateHow much 1.5 mM monosodium ascorbate do you use to make yoursolution? Answer in mL and report your value to three signicant gures.Please type only the number.arrow_forward1. What is the abbreviated form of the name for the molecule below. Punctuate it correctly ( image attached) 2. How much ATP is formed by the complete oxidation of lignocerate? Show stepsarrow_forward
- fill in the blank and circle the active site for each molecule. urgent!arrow_forwardfill in the table and circle the active sitearrow_forwardThe two half reactions for beginning and end of the electron transport chain are given below in standard form. Calculate & for the overall process. Using the Nernst equation (AG° = -n Fo, F= 96.485 kJ/volt mol), calculate AG°. Explain the need for a stepwise process in the electron transport chain. NAD* + H+ + 2 e- = NADH ½ 0г + 2H+ + 2е- = H20 = -0.32v E = +0.82Varrow_forward
- answer the questions and the example steps should be from carbohydrates glycolysis and citric acid cycle. Please put down reactions and structuresarrow_forwardidentify the general type of reaction catalyzed and an example step from glycolisis structure for each of the following enzymes/ co factor Kinase, isomerase, mutase, dehydrogenase, NAD+ , FADarrow_forwardfill in the blanks with the missing structures and give namesarrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





