
Sketch and label a galvanic cell that makes use of the following spontaneous
Write the half-reactions for the anode and cathode. Give the standard cell notation. (Hint: Determine which half-reaction represents oxidation and which represents reduction.)
Interpretation:
For the given spontaneous redox reaction, a galvanic cell is to be labeled and sketched, the half- reactions for anode and cathode are to be written, and a standard cell notation is to be given.
Concept Introduction:
The electrode at which reduction occurs is known as cathode and the electrode at which oxidation occurs is known as anode.
The experimental apparatus that creates electric current from the spontaneous redox reaction is known as galvanic cell.
In the standard cell notation, the single vertical line represents the phase boundary and the double vertical line represents the salt bridge, the anode half-cell is described to the left of the double line and the cathode half-cell is described to the right of the double line.
Answer to Problem 1PE
Solution:
Cathode:
Anode:
Cell notation:
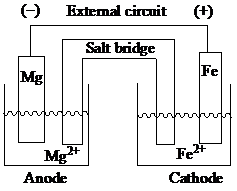
Explanation of Solution
The given spontaneous redox reaction is as:
At anode, oxidation takes place, whereas, at cathode, reduction takes place. In this galvanic cell, magnesium acts as the anode and iron acts as the cathode. In order to balance the redox reaction, iron gains two electrons while magnesium loses two electrons. Salt bridge enables the flow of ions which neutralize the charge imbalance. Within the salt bridge, the negative charge flows towards the anode to neutralize the accumulation of positive charge. Similarly, the positive charge flows towards the cathode to neutralize the accumulation of negative charge.
The half-cell reactions that take place at cathode and anode are as:
Half-cell reaction at cathode:
Half-cell reaction at anode:
The sketch of a galvanic cell is as:

In the standard cell notation, the magnesium anode half-cell is placed on the left and the cathode half-cell is placed on the right separated by the salt bridge. The standard cell notation for the given spontaneous redox reaction is represented as:
The half-cell reaction for anode and cathode and the standard notation cell notation have been written as:
Cathode:
Anode:
Cell notation:
Want to see more full solutions like this?
Chapter 19 Solutions
CHEMISTRY:MOLECULAR NATURE (LL)W/ACCESS
Additional Science Textbook Solutions
Chemistry: The Central Science (14th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Applications and Investigations in Earth Science (9th Edition)
Living By Chemistry: First Edition Textbook
Campbell Essential Biology (7th Edition)
- A standard galvanic cell is constructed so that the overall cell reaction is 2A13++(aq)+3M(s)3M2+(aq)+2A1(s) Where M is an unknown metal. If G = 411 kJ for the overall cell reaction, identify the metal used to construct the standard cell.arrow_forwardA voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO3.The other half cell consists of an inert platinum wire in an aqueous solution containing Fe2+(aq) and Fe3+(aq). (a) Calculate the cell potential, assuming standard conditions. (b) Write the net ionic equation for the reaction occurring in the cell. (c) Which electrode is the anode and which is the cathode? (d) If [Ag+] is 0.10 M, and [Fe2+] and [Fe3+] are both 1.0 M, what is the cell potential? Is the net cell reaction still that used in part (a)? If not, what is the net reaction under the new conditions?arrow_forwardThe mass of three different metal electrodes, each from a different galvanic cell, were determined before and after the current generated by the oxidation-reduction reaction in each cell was allowed to flow for a few minutes. The first metal electrode, given the label A, was found to have increased in mass; the second metal electrode, given the label B, did not change in mass; and the third metal electrode, given the label C, was found to have lost mass. Make an educated guess as to which electrodes were active and which were inert electrodes, and which were anode(s) and which were the cathode(s).arrow_forward
- Use the data from the table of standard reduction potentials in Appendix H to calculate the standard potential of the cell based on each of the following reactions. In each case, state whether the reaction proceeds spontaneously as written or spontaneously in the reverse direction under standard-state conditions. (a) H2(g)+Cl2(g)2H+(aq)+2Cl(aq) (b) Al3+(aq)+3Cr2+(aq)Al(s)+3Cr3+(aq) (c) Fe2+(aq)+Ag+(aq)Fe3+(aq)+Ag(s)arrow_forwardAn aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.39 hours. The electroplating is carried out with an efficiency of 93.0%, resulting in a deposit of 21.221 g of gold. a How many faradays are required to deposit the gold? b What is the charge on the gold ions (based on your calculations)?arrow_forwardA voltaic cell is constructed using the reaction of chromium metal and iron(II) ions. 2 Cr(s) + 3 Fe2+(aq) 2 Cr3+(aq) + 3 Fe(s) Complete the following sentences: Electrons in the external circuit flow from the ________ electrode to the ______ electrode. Negative ions move in the salt bridge from the ________ half-cell to the ______ half-cell. The half-reaction at the anode is _______ and that at the cathode is ________.arrow_forward
- Consider the following cell running under standard conditions: Fe(s)Fe2+(aq)Al3+(aq)Al(s) a Is this a voltaic cell? b Which species is being reduced during the chemical reaction? c Which species is the oxidizing agent? d What happens to the concentration of Fe3+(aq) as the reaction proceeds? e How does the mass of Al(s) change as the reaction proceeds?arrow_forwardAn electrolysis experiment is performed to determine the value of the Faraday constant (number of coulombs per mole of electrons). In this experiment, 28.8 g of gold is plated out from a AuCN solution by running an electrolytic cell for two hours with a current of 2.00 A. What is the experimental value obtained for the Faraday Constant?arrow_forwardGive the notation for a voltaic cell whose overall cell reaction is Mg(s)+2Ag+(aq)Mg2+(aq)+2Ag(s) What are the half-cell reactions? Label them as anode or cathode reactions. What is the standard cell potential of this cell?arrow_forward
- In principle, a battery could be made from aluminum metal and chlorine gas. (a) Write a balanced equation for the reaction thatwould occur in a battery using Al3+(aq) | Al(s) andCl2(g) | Cl(aq) half-cells. (b) Identify the half-reaction at the anode and at the cathode. Do electrons flow from the Al electrode when thecell does work? Explain. (c) Calculate the standard potential, Ecell, for the battery.arrow_forwardConsider the following galvanic cell: Calculate the concentrations of Ag+(aq) and Ni2+(aq) once the cell is dead.arrow_forwardYou have 1.0 M solutions of Al(NO3)3 and AgNO3 along with Al and Ag electrodes to construct a voltaic cell. The salt bridge contains a saturated solution of KCl. Complete the picture associated with this problem by a writing the symbols of the elements and ions in the appropriate areas (both solutions and electrodes). b identifying the anode and cathode. c indicating the direction of electron flow through the external circuit. d indicating the cell potential (assume standard conditions, with no current flowing). e writing the appropriate half-reaction under each of the containers. f indicating the direction of ion flow in the salt bridge. g identifying the species undergoing oxidation and reduction. h writing the balanced overall reaction for the cell.arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





