
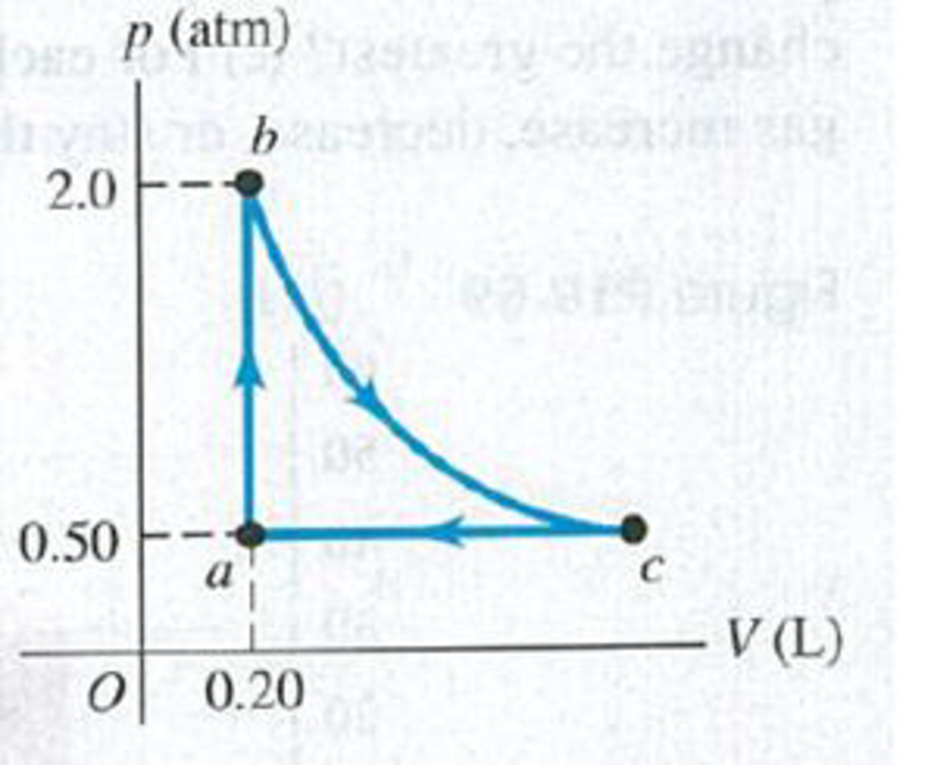
Figure P19.43 shows a pV-diagram for 0.0040 mol of ideal H2 gas. The temperature of the gas does not change during segment bc. (a) What volume does this gas occupy at point c? (b) Find the temperature of the gas at points a, b, and c. (c) How much heat went into or out of the gas during segments ab, ca, and bc? Indicate whether the heat has gone into or out of the gas. (d) Find the change in the internal energy of this hydrogen during segments ab, bc, and ca. Indicate whether the internal energy increased or decreased during each segment.
Figure P19.43
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
University Physics (14th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Physics for Scientists and Engineers with Modern Physics
Sears And Zemansky's University Physics With Modern Physics
An Introduction to Thermal Physics
University Physics Volume 2
Conceptual Physics (12th Edition)
- Case Study When a constant-volume thermometer is in thermal contact with a substance whose temperature is lower than the triple point of water, how does the right tube in Figure 19.22 need to be moved? Explain. FIGURE 19.22 1 Gas in the constant-volume gas thermometer is at Ti, and the mercury in the manometer is at height hi above the gasmercury boundary. 2 The thermometer is placed in thermal contact with an object, and its temperature increases. The increased temperature increases the gas volume. 3 By raising the right-hand tube of the mercury manometer, the gas volume is restored to its original size. The mercury is now at hi + h above the gasmercury boundary. This increase in height is a result of the increase in gas temperature and pressure.arrow_forwardIn Figure P19.22, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? Figure P19.22 (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D?arrow_forwardAn ideal gas is trapped inside a tube of uniform cross-sectional area sealed at one end as shown in Figure P19.49. A column of mercury separates the gas from the outside. The tube can be turned in a vertical plane. In Figure P19.49A, the column of air in the tube has length L1, whereas in Figure P19.49B, the column of air has length L2. Find an expression (in terms of the parameters given) for the length L3 of the column of air in Figure P19.49C, when the tube is inclined at an angle with respect to the vertical. FIGURE P19.49arrow_forward
- Consider a gas filling two connected chambers that are separated by a removable barrier (Fig. P20.68). The gas molecules on the left (red) are initially at a higher temperature than the ones on the right (blue). When the barrier between the two chambers is removed, the molecules begin to mix and move from one chamber to the other. a. Describe what happens to the temperature in the left chamber and in the right chamber as time goes on, once the barrier is open. Discuss in terms of the mixing of the molecules from each gas. b. Describe what happens to the most probable speed and average speed in the left chamber and in the right chamber as time goes on, once the barrier is open. Do they increase or decrease by the same factor? Explain. FIGURE P20.68 Problems 68 and 69.arrow_forwardIn Figure P17.32, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D? Figure P17.32arrow_forwardIn the text, it was shown that N/V=2.681025m3 for gas at STP. (a) Show that this quantity is equivalent to N/V=2.681019cm3, as stated. (b) About how many atoms are mere in one m3 (a cubic micrometer) at STP? (c) What does your answer to part (b) imply about the separation of Mama and molecules?arrow_forward
- Figure P21.37 shows a PV diagram for a gas that is compressed from Vi to Vf. Find the work done by the a. gas and b. environment during this process. Does energy enter the system or leave the system as a result of work? FIGURE P21.37arrow_forwardAn air bubble starts rising from the bottom of a lake. Its diameter is 3.60 mm at the bottom and 4.00 mm at the surface. The depth of the lake is 2.50 m, and the temperature at the surface is 40.0C. What is the temperature at the bottom of the lake? Consider the atmospheric pressure to be 1.01 105 Pa and the density of water to be 1.00 103 kg/m3. Model the air as an ideal gas. 53. Use the ideal gas law for the bottom (point 1) and the surface (point 2) of the lake. At the surface, the pressure is atmospheric pressure. However, at the bottom it is equal to to the sum of the atmospheric pressure and the pressure due to 2.50 m column of water. P2=1.01105PaP1=P2+WghWP1=1.01105Pa+(1.00103kg/m3)(9.81m/s2)(2.50m) Use the ideal gas law (Eq. 19.17). T1=P1V1P2V2T2 The volume ratio at the bottom and top of the lake can be calculated with the diameters given. V1V2=43r1343r23=(1.82.0)3 T1=P1P2(V1V2)T2T1=1.01105Pa+(1.00103kg/m3)(9.81m/s2)(2.50m)1.01105Pa(1.802.00)3(40.0+273.15K)T1=284Karrow_forwardA cylinder is closed at both ends and has insulating EZZ3 walls. It is divided into two compartments by an insulating piston that is perpendicular to the axis of the cylinder as shown in Figure P21.75a. Each compartment contains 1.00 mol of oxygen that behaves as an ideal gas with = 1.40. Initially, the two compartments haw equal volumes and their temperatures are 550 K and 250 K. The piston is then allowed to move slowly parallel to the axis of the cylinder until it comes to rest at an equilibrium position (Fig. P2l.75b). Find the final temperatures in the two compartments.arrow_forward
- Cylinder A contains oxygen (O2) gas, and cylinder B contains nitrogen (N2) gas. If the molecules in the two cylinders have the same rms speeds, which of the following statements is false? (a) The two gases haw different temperatures. (b) The temperature of cylinder B is less than the temperature of cylinder A. (c) The temperature of cylinder B is greater than the temperature of cylinder A. (d) The average kinetic energy of the nitrogen molecules is less than the average kinetic energy of the oxygen molecules.arrow_forwardIf a gas is compressed isothermally, which of the following statements is true? (a) Energy is transferred into the gas by heat. (b) No work is done on the gas. (c) The temperature of the gas increases. (d) The internal energy of the gas remains constant. (e) None of those statements is true.arrow_forwardOne mole of an ideal monatomic gas occupies a volume of 1.0102 m3 at a pressure of 2.0105 N/m2. (a) What is the temperature of the gas? (b) The gas undergoes a quasi-static adiabatic compression until its volume is decreased to 5.0103 m3. is the new gas temperature? (c) How much work is done on the gas during the compression? (d) What is the change in the internal energy of the gas?arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





