
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
5th Edition
ISBN: 9780133887518
Author: H. Scott Fogler
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 18.16P
Consider the following system in Figure P18-16C used to model a real reactor:
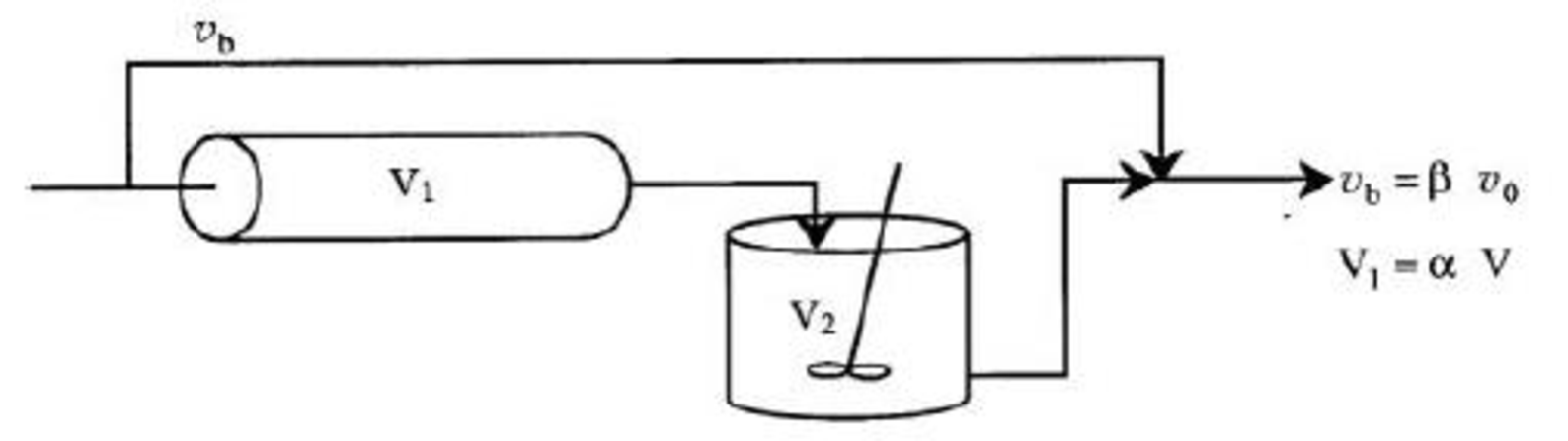
Figure P18-16C Model system.
Describe how you would evaluate the parameters α and β.
- (a) Draw the F- and E-curves for this system of ideal reactors used to model a real reactor using β = 0.2 and α = 0.4. Identify the numerical values of the points on the F-curve (e.g., t1) as they relate to τ.
- (b) If the reaction A → B is second order with kCA0 = 0.5 min–1, what is the conversion assuming the space time for the real reactor is 2 min?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
give me solution math not explin
give me solution math not explin
give me solution math not explin
Chapter 18 Solutions
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Ch. 18 - The second-order liquid-phase reaction AB+C is to...Ch. 18 - The elementary liquid-phase reaction...Ch. 18 - Prob. 18.5PCh. 18 - Use the data in Example 16-2 to make the following...Ch. 18 - A tubular reactor has been sized to obtain 98%...Ch. 18 - Prob. 18.11PCh. 18 - Prob. 18.13PCh. 18 - Prob. 18.14PCh. 18 - Prob. 18.15PCh. 18 - Consider the following system in Figure P18-16C...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Example (6): An evaporator is concentrating F kg/h at 311K of a 20wt% solution of NaOH to 50wt %. The saturated steam used for heating is at 399.3K. The pressure in the vapor space of the evaporator is 13.3 KPa abs. The 5:48 O Transcribed Image Text: Example (7): Determine thearrow_forward14.9. A forward feed double-effect vertical evaporator, with equal heating areas in each effect, is fed with 5 kg/s of a liquor of specific heat capacity of 4.18 kJ/kg K. and with no boiling point rise, so that 50 per cent of the feed liquor is evaporated. The overall heat transfer coefficient in the second effect is 75 per cent of that in the first effect. Steam is fed at 395 K and the boiling point in the second effect is 373 K. The feed is heated by an external heater to the boiling point in the first effect. It is decided to bleed off 0.25 kg/s of vapour from the vapour line to the second effect for use in another process. If the feed is still heated to the boiling point of the first effect by external means, what will be the change in steam consumption of the evaporator unit? For the purpose of calculation, the latent heat of the vapours and of the steam may both be taken as 2230 kJ/kgarrow_forwardExample(3): It is desired to design a double effect evaporator for concentrating a certain caustic soda solution from 12.5wt% to 40wt%. The feed at 50°C enters the first evaporator at a rate of 2500kg/h. Steam at atmospheric pressure is being used for the said purpose. The second effect is operated under 600mmHg vacuum. If the overall heat transfer coefficients of the two stages are 1952 and 1220kcal/ m2.h.°C. respectively, determine the heat transfer area of each effect. The BPR will be considered and present for the both effect 5:49arrow_forward
- العنوان ose only Q Example (7): Determine the heating surface area 개 required for the production of 2.5kg/s of 50wt% NaOH solution from 15 wt% NaOH feed solution which entering at 100 oC to a single effect evaporator. The steam is available as saturated at 451.5K and the boiling point rise (boiling point evaluation) of 50wt% solution is 35K. the overall heat transfer coefficient is 2000 w/m²K. The pressure in the vapor space of the evaporator at atmospheric pressure. The solution has a specific heat of 4.18kJ/ kg.K. The enthalpy of vaporization under these condition is 2257kJ/kg Example (6): 5:48 An evaporator is concentrating F kg/h at 311K of a 20wt% solution of NaOH to 50wt %. The saturated steam used for heating is at 399.3K. The pressure in the vapor space of the evaporator is 13.3 KPa abs. The 5:48 1 J ۲/۱ ostrarrow_forwardExample 8: 900 Kg dry solid per hour is dried in a counter current continues dryer from 0.4 to 0.04 Kg H20/Kg wet solid moisture content. The wet solid enters the dryer at 25 °C and leaves at 55 °C. Fresh air at 25 °C and 0.01Kg vapor/Kg dry air is mixed with a part of the moist air leaving the dryer and heated to a temperature of 130 °C in a finned air heater and enters the dryer with 0.025 Kg/Kg alry air. Air leaving the dryer at 85 °C and have a humidity 0.055 Kg vaper/Kg dry air. At equilibrium the wet solid weight is 908 Kg solid per hour. *=0.0088 Calculate:- Heat loss from the dryer and the rate of fresh air. Take the specific heat of the solid and moisture are 980 and 4.18J/Kg.K respectively, A. =2500 KJ/Kg. Humid heat at 0.01 Kg vap/Kg dry=1.0238 KJ/Kg. "C. Humid heat at 0.055 Kg/Kg 1.1084 KJ/Kg. "C 5:42 Oarrow_forwardQ1: From the Figure below for (=0.2 find the following 1. Rise Time 2. Time of oscillation 3. Overshoot value 4. Maximum value 5. When 1.2 which case will be? 1.6 1.4 1.2 12 1.0 |=0.8- 0.6 0.4 0.8 0.2- 0.6 0.4 0.2 1.2 = 1.0 0 2 4 6 8 10 10 t/Tarrow_forward
- A system, in a closed container, consists of an unknown number of components and three phases. You are told that the system is fully defined by giving you only one mole fraction! What is the number components that is present? 3 1 2 The question is ill-posed.arrow_forwardA mixture of 2 components in 2 phases are present. You are given the temperature and mole fraction. How many additional variables can be specified before the system is completely determined? none 2 the system is overspecified 1 3arrow_forwardAt a Pressure of 600 mm Hg, match the substance with the boiling temperature. 54.69°C 1. n-Pentane 49.34°C 2. n-Hexane 3. Acetone 29.32°C く 61.40°C 4. Chloroformarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The

Introduction to Chemical Engineering Thermodynami...
Chemical Engineering
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind...
Chemical Engineering
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY

Elements of Chemical Reaction Engineering (5th Ed...
Chemical Engineering
ISBN:9780133887518
Author:H. Scott Fogler
Publisher:Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:9781285061238
Author:Lokensgard, Erik
Publisher:Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:9780072848236
Author:Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:McGraw-Hill Companies, The
General Mole Balance Equation // Reactor Engineering - Class 4; Author: Chemical Engineering Guy;https://www.youtube.com/watch?v=OYBhLfX0DYE;License: Standard YouTube License, CC-BY
Reactor Engineering 03 - Mole Balances; Author: Randhir Rawatlal;https://www.youtube.com/watch?v=KzmMTJHgjgM;License: Standard Youtube License