
a)
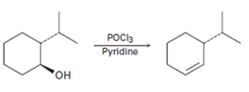
Interpretation:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed. The regiochemistry of the elimination is also to be explained.
Concept introduction:
The dehydration of alcohols to
To propose:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3 is to be proposed.
To explain:
The regiochemistry of the elimination reaction.
b)
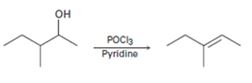
Interpretation:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed. The regiochemistry of the elimination is also to be explained.
Concept introduction:
The dehydration of alcohols to alkenes in the presence of POCl3 follows E2 mechanism. The –OH group is converted into a better leaving group dichlorophosphate, –OPOCl2, when treated with POCl3. Pyridine used as solvent is also a base and it removes a proton arranged anti to the leaving group from the β carbon (requirement for E2 mechanism). The removal of the proton and –OPOCl2 occurs simultaneously in a single step to yield the alkene as the product.
To propose:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed.
To explain:
The regiochemistry of the elimination reaction.
c)
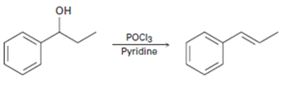
Interpretation:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed. The regiochemistry of the elimination is also to be explained.
Concept introduction:
The dehydration of alcohols to alkenes in the presence of POCl3 follows E2 mechanism. The –OH group is converted into a better leaving group dichlorophosphate, –OPOCl2, when treated with POCl3. Pyridine used as solvent is also a base and it removes a proton arranged anti to the leaving group from the β carbon (requirement for E2 mechanism). The removal of the proton and –OPOCl2 occurs simultaneously in a single step to yield the alkene as the product.
To propose:
A mechanism using curved arrows, for the conversion of alcohol given into the corresponding alkene by treating with POCl3, is to be proposed.
To explain:
The regiochemistry of the elimination reaction.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry - With Access (Custom)
- P A student claims the right-hand side of the reaction in the drawing area below shows the product of a Claisen condensation. • If the student is correct, complete the reaction by adding the necessary organic reactants to the left-hand side, and by adding any necessary reagents and reaction conditions above and below the arrow. • If the student is incorrect, because it's not possible to obtain this product from a Claisen condensation, check the box under the drawing area instead. those that will minimize any byproducts or competing • Note for advanced students: If you have a choice, use the most efficient reactants and reagents reactions. - ☐ ☐ : ☐ + I Х Click and drag to start drawing a structure.arrow_forwardidentify the relationship between the structures and H- OH HO H H- OH and HO H H -ОН HO H Br and Brarrow_forwardThe right-hand side of this reaction shows the product of an aldol condensation. What are the reactants missing from the left-hand side? Draw them below. ? NaOH Δ If there aren't any reactants that would lead to these products under the reaction conditions given, just check the box under the drawing area. Note for advanced students: don't worry if the reactants you propose might also make some other products under these reaction conditions. Just make sure the product above is one of the major products.arrow_forward
- Please help! I need to identify four labeled unknown bottles based off of their colors doing titration using phenlphtalein. I've included my answers, but I wanted to make sure they were correct and if not, what will be correct thank you in advance.arrow_forwardAn organic chemistry Teaching Assistant (TA) suggested in your last discussion section that there is only one major organic product of the following reaction and that this reaction builds a ring. If the TA is right, draw the product in the drawing area below. If the TA is wrong, just check the box below the drawing area. NaOH ?arrow_forwardA student suggests that the molecule on the right can be made from a single molecule that doesn't have a ring. If the student is correct, draw the starting material below, otherwise, check the box under the drawing area. Click and drag to start drawing a structure. : ☐ + NaOH टेarrow_forward
- Rate = k [I]1.7303[S2O82-]0.8502, Based on your rate, write down a mechanism consistent with your results and indicate which step is the rate determining step.arrow_forward36. Give the major product(s) of each of the following reactions. Aqueous work-up steps (when necessary) have been omitted. a. CH3CH=CHCH3 b. CH3CH2CH2CCH3 H,PO₂, H₂O, A (Hint: See Section 2-2.) 1. LIAIH. (CH,CH,),O 2. H', H₂O H NaBH, CH,CH₂OH d. Br LIAIH. (CH,CH,)₂O f. CH3 NaBH, CH,CH,OH (CH3)2CH H NaBH, CH,CH₂OH Harrow_forwardPredict the major products of this reaction: + H excess NaOH Δ ? Note that the second reactant is used in excess, that is, there is much more of the second reactant than the first. If there won't be any products, just check the box under the drawing area instead.arrow_forward
- An organic chemistry Teaching Assistant (TA) suggested in your last discussion section that there is only one major organic product of the following reaction and that this reaction builds a ring. If the TA is right, draw the product in the drawing area below. If the TA is wrong, just check the box below the drawing area. 1. NaOMe CH3O N. OCH3 ? 2. H3O+arrow_forwardComplete the reaction in the drawing area below by adding the major products to the right-hand side. If there won't be any products, because nothing will happen under these reaction conditions, check the box under the drawing area instead. Note: if the products contain one or more pairs of enantiomers, don't worry about drawing each enantiomer with dash and wedge bonds. Just draw one molecule to represent each pair of enantiomers, using line bonds at the chiral center. + More... ☐ ☐ : ☐ + G 1. NaOMe Click and drag to start drawing a structure. 2. H +arrow_forward6. Ammonia reacts with nitrogen monoxide and oxygen to form nitrogen and water vapor. If the rate of consumption of NO is 4.5 mollitermin) (a) Find the rate of reaction (b) Find the rate of formations of N; and HO (c) Find the rate of consumption of NH, and O 4NH: 4NO 0:4: +60arrow_forward

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

