
Concept explainers
BIO Base pairing in DNA, I. The two sides of the DNA double helix are connected by pairs of bases (adenine, thymine, cytosine, and guanine). Because of the geometric shape of these molecules, adenine bonds with thymine and cytosine bonds with guanine. Figure 17.46 shows the thymine-adenine bond. Each charge shown is ±e. and the H—N distance is 0.110 nm. (a) Calculate the net force that thymine exerts on adenine. Is it attractive or repulsive? To keep the calculations fairly simple, yet reasonable, consider only the forces due to the O—H—N and the N —H—N combinations, assuming that these two combinations are parallel to each other. Remember, however, that in the O—H—N set. the O−exerts a force on both the H+ and the N−, and likewise along the N—H—N set. (b) Calculate the force on the electron in the hydrogen atom, which is 0.0529 nm from the proton. Then compare the strength of the bonding force of the electron in hydrogen with the bonding force of the adenine-thymine molecules.
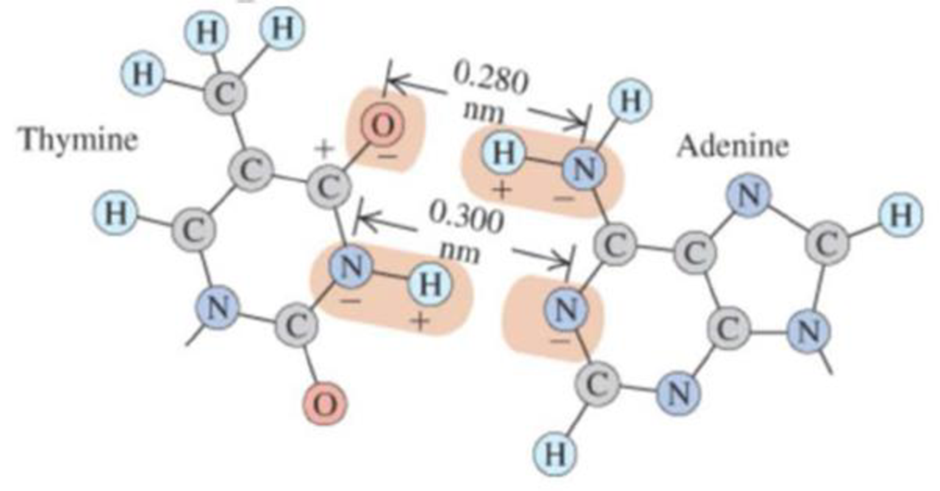
Figure 17.46
Problem 24.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
College Physics (10th Edition)
Additional Science Textbook Solutions
Cosmic Perspective Fundamentals
College Physics
College Physics: A Strategic Approach (4th Edition)
Sears And Zemansky's University Physics With Modern Physics
College Physics: A Strategic Approach (3rd Edition)
Conceptual Integrated Science
- (a) Find the total Coulomb force on a charge of 2.00 nC located at x = 4.00 cm in Figure 18.52 (b): given that q = 1,00C . (b) Find the x-position at which the electric field is zero in Figure 18.52 (b).arrow_forwardA water molecule consists of two hydrogen atoms bonded with one oxygen atom. The bond angle between the two hydrogen atoms is 104( (see below). Calculate the net dipole moment of a water molecule that is placed in a uniform, horizontal electric field of magnitude 2.3108N/C . (You are missing some information for solving this problem; you will need to determine what information you need, and look it up.)arrow_forward(a) What is the direction of the total Coulomb force on q in Figure 18.46 if q is negative, qa= qcand both are negative, and qb= qcand both are positive? (b) What is the direction of the electric field at the center of the square in this situation?arrow_forward
- Consider the following. 0.500 mm 60.0 (a) Red blood cells often become charged and can be treated as point charges. Healthy red blood cells are negatively charged, but unhealthy cells (due to the presence of a bacteria, for example) can become positively charged. In the figure, three red blood cells are oriented such that they are located on the corners of an equilateral triangle. The red blood cell charges are A = 1.80 pC, B = 7.10 pC, and C = -4.40 pC. Given these charges, what would the magnitude and direction of the electric field be at cell A? (1 pC = 1 x 10-12 C.) magnitude N/C direction ° counterclockwise from the +x-axis (b) If the charge of cell A were doubled, how would the electric field at cell A change? The magnitude of the field would be doubled. The magnitude of the field would be halved. Does the electric field at cell A include the electric field generated by cell A?The field would be unchanged. O The magnitude of the field would be quadrupled.arrow_forwardHow should I set up equations?arrow_forwardPart D Compare the strength of the bonding force of the electron in hydrogen with the bonding force of the adenine-thymine molecules. Essay answers are limited to about 500 words (3800 characters maximum, including spaces). 3800 Character(s) remaining Submit Request Answerarrow_forward
- I8arrow_forwardConsider the following. В 0.500 mm A 60.0° (a) Red blood cells often become charged and can be treated as point charges. Healthy red blood cells are negatively charged, but unhealthy cells (due to the presence of a bacteria, for example) can become positively charged. In the figure, three red blood cells are oriented such that they are located on the corners of an equilateral triangle. The red blood cell charges are А 3 1.90 рС, В 6.70 pC, and C = -3.90 pC. Given these charges, what would the magnitude and direction of the electric field be at cell A? (1 pC 1 x 10-12 C.) magnitude N/C direction ° counterclockwise from the +x-axis (b) If the charge of cell A were doubled, how would the electric field at cell A change? The magnitude of the field would be quadrupled. The magnitude of the field would be doubled. The field would be unchanged. O The magnitude of the field would be halved.arrow_forward60.0 0.500 mm (a) Red blood cells often become charged and can be treated as point charges. Healthy red blood cells are negatively charged, but unhealthy cells (due to the presence of a bacteria, for example) can become positively charged. In the figure, three red blood cells are oriented such that they are located on the corners of an equilateral triangle. The red blood cell charges are A = 1.90 pC, B = 6.50 PC, and C= -4.80 pC. Given these charges, what would the magnitude and direction of the electric field be at cell A? (1 pC= 1 x 10-12 C.) magnitude 210191.91 ✓N/C X 77.605 direction What are the components of the net electric field? Can you then use these to find the direction?" counterclockwise from the +x-axisarrow_forward
- Can you explain also. Thank you.arrow_forwardConsider the following. a) Red blood cells often become charged and can be treated as point charges. Healthy red blood cells are negatively charged, but unhealthy cells (due to the presence of a bacteria, for example) can become positively charged. In the figure, three red blood cells are oriented such that they are located on the corners of an equilateral triangle. The red blood cell charges are A = 2.30 pC, B = 6.90 pC, and C = −4.10 pC. Given these charges, what would the magnitude and direction of the electric field be at cell A? (1 pC = 1 ✕ 10−12 C.) Magnitude: (in N/C) Direction: ° counterclockwise from the +x-axisarrow_forwardA molecule of DNA (deoxyribonucleic acid) is 2.17 mm long. The ends of the molecule become singly ionized: negative on one end, positive on the other. The helical molecule acts like a spring and compresses 1.00% upon becoming charged. Determine the effective spring constant of the molecule.arrow_forward

 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning



