
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
5th Edition
ISBN: 9781260170405
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.61P
Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia.
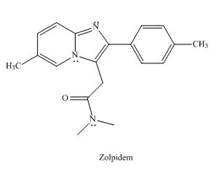
a In what type of orbital does the lone pair on each N atom in the heterocycle reside?
b. Explain why the bicyclic ring system that contains both N atoms is
c. Draw all reasonable resonance structures for the bicyclic ring system.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used hand raiting and don't used Ai solution
IX) By writing the appropriate electron configurations and orbital box diagrams briefly EXPLAIN in your own words
each one of the following questions:
a) The bond length of the Br2 molecule is 2.28 Å, while the bond length of the compound KBr is 3.34 Å. The radius of
K✶ is 1.52 Å. Determine the atomic radius in Å of the bromine atom and of the bromide ion.
Br =
Br
b) Explain why there is a large difference in the atomic sizes or radius of the two (Br and Br).
T
When 15.00 mL of 3.00 M NaOH was mixed in a calorimeter with 12.80 mL of 3.00 M HCl, both initially at room temperature (22.00 C), the temperature increased to 29.30 C. The resultant salt solution had a mass of 27.80 g and a specific heat capacity of 3.74 J/Kg. What is heat capacity of the calorimeter (in J/C)? Note: The molar enthalpy of neutralization per mole of HCl is -55.84 kJ/mol.
Chapter 17 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Ch. 17 - Prob. 17.1PCh. 17 - Problem 17.2 What orbitals are used to form the...Ch. 17 - Problem-17.3. Give the IUPAC name for each...Ch. 17 - Prob. 17.4PCh. 17 - Problem-17.5 What is the structure of propofol,...Ch. 17 - Problem 17.6 What is the structure of a compound...Ch. 17 - Problem 17.7 How many NMR signals does each...Ch. 17 - Prob. 17.8PCh. 17 - Prob. 17.9PCh. 17 - Prob. 17.10P
Ch. 17 - Prob. 17.11PCh. 17 - Prob. 17.12PCh. 17 - Prob. 17.13PCh. 17 - Problem 17.14 Januvia, the trade name for...Ch. 17 - Prob. 17.15PCh. 17 - Problem 17.16 Rank the following compounds in...Ch. 17 - Problem 17.17 Draw the seven resonance structures...Ch. 17 - Prob. 17.18PCh. 17 - Prob. 17.19PCh. 17 - Prob. 17.20PCh. 17 - Prob. 17.21PCh. 17 - Problem 17.22 How many NMR signals does ...Ch. 17 - 17.23 Name each compound and state how many lines...Ch. 17 - Prob. 17.24PCh. 17 - Prob. 17.25PCh. 17 - Prob. 17.26PCh. 17 - 17.27 Give the IUPAC name for each compounds.
a....Ch. 17 - 17.28 Draw a structure corresponding to each...Ch. 17 - 17.29 a. Draw the 14 constitutional isomers of...Ch. 17 - Prob. 17.30PCh. 17 - Prob. 17.31PCh. 17 - Prob. 17.32PCh. 17 - 17.33 Label each compound as aromatic,...Ch. 17 - Prob. 17.34PCh. 17 - 17.35 Pentalene, azulene, and heptalene are...Ch. 17 - 17.36 The purine heterocycle occurs commonly in...Ch. 17 - Prob. 17.37PCh. 17 - 17.38
How many electrons does C contain?
How...Ch. 17 - Prob. 17.39PCh. 17 - 17.40 Explain the observed rate of reactivity of...Ch. 17 - 17.41 Draw a stepwise mechanism for the following...Ch. 17 - Prob. 17.42PCh. 17 - 17.43 Draw additional resonance structures for...Ch. 17 - Prob. 17.44PCh. 17 - Prob. 17.45PCh. 17 - 17.46 Which compound in each pair is the stronger...Ch. 17 - 17.47 Treatment of indene with forms its...Ch. 17 - Prob. 17.48PCh. 17 - 17.49 Draw the conjugate bases of pyrrole and...Ch. 17 - 17.50 a. Explain why protonation of pyrrole occurs...Ch. 17 - Prob. 17.51PCh. 17 - Prob. 17.52PCh. 17 - 17.53 How many signals does each compound...Ch. 17 - 17.54 Which of the diethylbenzene isomers (ortho,...Ch. 17 - 17.55 Propose a structure consistent with each...Ch. 17 - 17.56 Propose a structure consistent with each...Ch. 17 - 17.57 Thymol (molecular formula ) is the major...Ch. 17 - 17.58 You have a sample of a compound of molecular...Ch. 17 - 17.59 Explain why tetrahydrofuran has a higher...Ch. 17 - 17.60 Rizatriptan (trade name Maxalt) is a...Ch. 17 - 17.61 Zolpidem (trade name Ambien) promotes the...Ch. 17 - 17.62 Answer the following questions about...Ch. 17 - 17.63 Stanozolol is an anabolic steroid that...Ch. 17 - Prob. 17.64PCh. 17 - 17.65 Use the observed data to decide whether C...Ch. 17 - Prob. 17.66PCh. 17 - Prob. 17.67PCh. 17 - Prob. 17.68PCh. 17 - 17.69 Although benzene itself absorbs at in its ...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- When 15.00 mL of 3.00 M NaOH was mixed in a calorimeter with 12.80 mL of 3.00 M HCl, both initially at room temperature (22.00 C), the temperature increased to 29.30 C. The resultant salt solution had a mass of 27.80 g and a specific heat capacity of 3.74 J/Kg. What is heat capacity of the calorimeter (in J/C)? Note: The molar enthalpy of neutralization per mole of HCl is -55.84 kJ/mol. Which experimental number must be initialled by the Lab TA for the first run of Part 1 of the experiment? a) the heat capacity of the calorimeter b) Mass of sample c) Ti d) The molarity of the HCl e) Tfarrow_forwardPredict products for the Following organic rxn/s by writing the structurels of the correct products. Write above the line provided" your answer D2 ①CH3(CH2) 5 CH3 + D₂ (adequate)" + 2 mited) 19 Spark Spark por every item. 4 CH 3 11 3 CH 3 (CH2) 4 C-H + CH3OH CH2 CH3 + CH3 CH2OH 0 CH3 fou + KMnDy→ C43 + 2 KMn Dy→→ C-OH ") 0 C-OH 1110 (4.) 9+3 =C CH3 + HNO 3 0 + Heat> + CH3 C-OH + Heat CH2CH3 - 3 2 + D Heat H 3 CH 3 CH₂ CH₂ C = CH + 2 H₂ → 2 2arrow_forwardWhen 15.00 mL of 3.00 M NaOH was mixed in a calorimeter with 12.80 mL of 3.00 M HCl, both initially at room temperature (22.00 C), the temperature increased to 29.30 C. The resultant salt solution had a mass of 27.80 g and a specific heat capacity of 3.74 J/Kg. What is heat capacity of the calorimeter (in J/C)? Note: The molar enthalpy of neutralization per mole of HCl is -55.84 kJ/mol.arrow_forward
- Q6: Using acetic acid as the acid, write the balanced chemical equation for the protonation of the two bases shown (on the -NH2). Include curved arrows to show the mechanism. O₂N- O₂N. -NH2 -NH2 a) Which of the two Bronsted bases above is the stronger base? Why? b) Identify the conjugate acids and conjugate bases for the reactants. c) Identify the Lewis acids and bases in the reactions.arrow_forwardQ5: For the two reactions below: a) Use curved electron-pushing arrows to show the mechanism for the reaction in the forward direction. Redraw the compounds to explicitly illustrate all bonds that are broken and all bonds that are formed. b) Label Bronsted acids and bases in the left side of the reactions. c) For reaction A, which anionic species is the weakest base? Which neutral compound is the stronger acid? Is the forward or reverse reaction favored? d) Label Lewis acids and bases, nucleophiles and electrophiles in the left side of the reactions. A. 용 CH3OH я хон CH3O OH B. HBr CH3ONa NaBr CH3OHarrow_forwardpotential energy Br b) Translate the Newman projection below to its wedge-and-dash drawing. F H. OH CH3 CI c) Isopentane (2-methylbutane) is a compound containing a branched carbon chain. Draw a Newman projection of six conformations about the C2-C3 bond of isopentane. On the curve of potential energy versus angle of internal rotation for isopentane, label each energy maximum and minimum with one of the conformations. 0° 。 F A B D C angle of internal rotation E F 360° (=0°) JDownlarrow_forward
- Q7: Identify the functional groups in these molecules a) CH 3 b) Aspirin: HO 'N' Capsaicin HO O CH3 CH 3arrow_forwardQ2: Name the following alkanesarrow_forward1. Complete the following table in your laboratory notebook. Substance Formula Methanol CH3OH Ethanol C2H5OH 1-Propanol C3H7OH 1-Butanol C4H9OH Pentane C5H12 Hexane C6H14 Water H₂O Acetone C3H60 Structural Formula Molecular Weight (g/mol) Hydrogen Bond (Yes or No)arrow_forward
- Q1: Compare the relative acidity in each pair of compounds. Briefly explain. (a) CH3OH vs NH 3 (b) HF vs CH3COOH (c) NH3 vs CH4 (d) HCI vs HI (e) CH3COOH vs CH3SH (f) H₂C=CH2 vs CH3 CH3 (g) compare the acidity of the two bolded hydrogens O. H N- (h) compare the acidity of the two bolded hydrogens, draw resonance structures to explain H H Harrow_forwardQ3: Rank the following molecules in order of decreasing boiling point: (a) 3-methylheptane; (b) octane; (c) 2,4-dimethylhexane; (d) 2,2,4-trimethylpentane.arrow_forwardQ5: Conformations of Alkanes a) Draw a Newman Projection of the compound below about the C2-C3 bond. H3C Cli... H IIIH Br CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY