
(a)
Interpretation:
The preparation method of benzyl methyl ether from toluene is to be stated.
Concept introduction:
Cyclic
Answer to Problem 17.28AP
The preparation method of benzyl methyl ether from toluene is shown below.
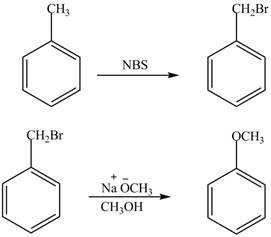
Explanation of Solution
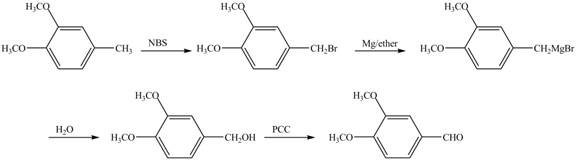
For preparing benzyl methyl ether from toluene, toluene is first brominated.
For bromination of toluene,
The corresponding reaction sequences are shown below.
Figure 1
The preparation method of benzyl methyl ether from toluene is shown in Figure 1.
(b)
Interpretation:
The preparation method of
Concept introduction:
Cyclic alkenes on reaction with N-Bromosuccinimide (NBS) forms allyl or benzyl bromide, that is, bromine is substituted at the allylic or benzylic position. NBS is a rich source of free radical of
Answer to Problem 17.28AP
The preparation method of
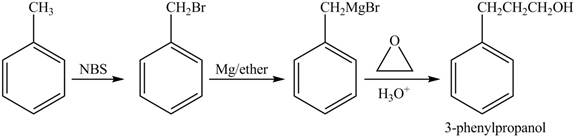
Explanation of Solution
In given reaction, toluene is first brominated with NBS (N-bromosuccinamide). Brominated benzyl reacts with Grignard reagent and
The corresponding reaction sequences to obtain the desired product are shown below.

Figure 2
The preparation method of
(c)
Interpretation:
The preparation method of
Concept introduction:
The Lindlar’s catalyst is composed of
Answer to Problem 17.28AP
The preparation method of
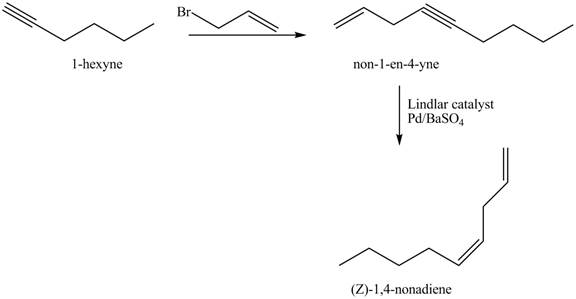
Explanation of Solution
The addition of
The corresponding reaction sequences to obtain the desired product are shown below.
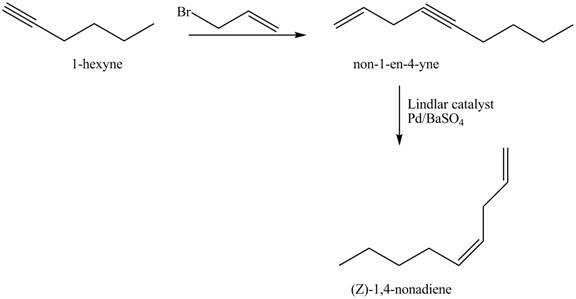
Figure 3
The preparation method of
(d)
Interpretation:
The preparation method of given compound from cyclopentene is to be stated.
Concept introduction:
Cyclic alkenes on reaction with N-Bromosuccinimide (NBS) forms allyl or benzyl bromide, that is, bromine is substituted at the allylic or benzylic position. NBS is a rich source of free radical of
Answer to Problem 17.28AP
The preparation method of given compound from cyclopentene is shown below.
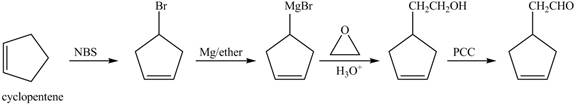
Explanation of Solution
Cyclopentene is first brominated and then on treatment with Grignard reagent and epoxide it gives corresponding alkyl alcohol. This alkyl alcohol is oxidized with mild reagent PCC to convert it into
The corresponding reaction sequences to obtain the desired product are shown below.
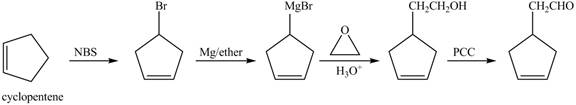
Figure 4
The preparation method of given compound from cyclopentene is shown in Figure 4.
(e)
Interpretation:
The preparation method of
Concept introduction:
The
Answer to Problem 17.28AP
The preparation method of
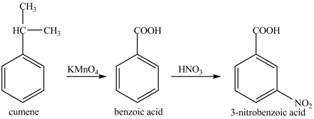
Explanation of Solution
Cumene has to be oxidized first and then its nitration is done to get the required product
Alkyl group is oxidized to gives benzoic acid. This benzoic acid is nitrated with
The corresponding reaction sequences to obtain the desired product are shown below.
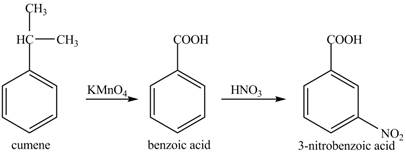
Figure 5
The preparation method of
(f)
Interpretation:
The preparation method of para-nitro benzoic acid from cumene (isopropyl benzene) is to be stated.
Concept introduction:
The chemical reaction in which an electrophile group is replaced by another functional group is known as the electrophilic substitution reaction. When the electrophilic substitution happens on an aromatic ring such as benzene then the reaction is known as electrophilic aromatic substitution.
Answer to Problem 17.28AP
The preparation method of para-nitro benzoic acid from cumene (isopropyl benzene) is shown below.
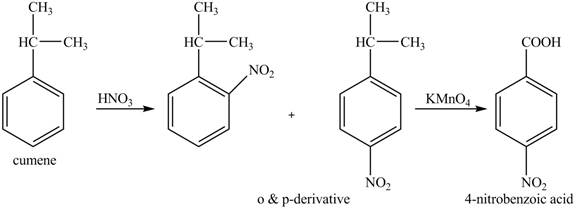
Explanation of Solution
As alkyl group is ortho and para directing, so, both the derivatives are formed. The reaction of cumene with nitric acid results in the formation of ortho and para nitro-cumene. The para nitro cumene is further oxidized to p-nitro benzoic acid with potassium permangnate.
The corresponding reaction sequences to obtain the desired product are shown below.
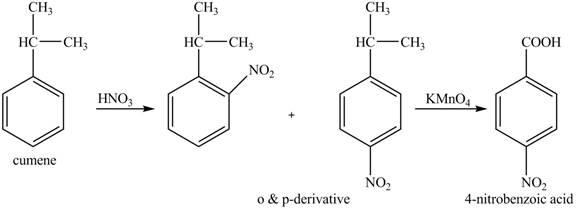
Figure 6
The preparation method of para-nitro benzoic acid from cumene (isopropyl benzene) is shown in Figure 6.
(g)
Interpretation:
The preparation method of
Concept introduction:
Cyclic alkenes on reaction with N-Bromosuccinimide (NBS) forms allyl or benzyl bromide, that is, bromine is substituted at the allylic or benzylic position. NBS is a rich source of free radical of
Answer to Problem 17.28AP
The preparation method of
Explanation of Solution
In the given reaction, at first, the substrate,
The corresponding reaction sequences to obtain the desired product are shown below.
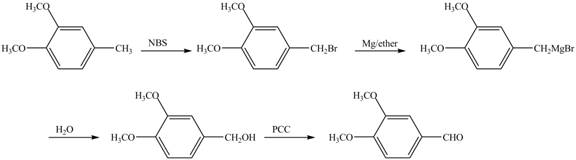
Figure 7
The preparation method of
(h)
Interpretation:
The preparation of given compound from cyclopentenol is to be given.
Concept introduction:
The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as a substitution reaction. In a nucleophilic substitution reaction, nucleophile takes the position of leaving the group by attacking the electron-deficient carbon atom.
Answer to Problem 17.28AP
The preparation of given compound from cyclopentenol is shown below.
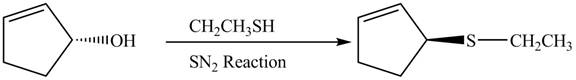
Explanation of Solution
The given reaction is a nucleophilic substitution reaction. The nucleophile attacks to replace the
This is a single step substitution nucleophile reaction as shown below.
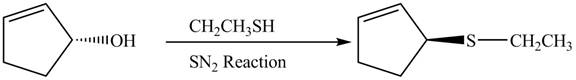
Figure 8
In this reaction, the stereochemistry of molecule changes as from one side the group is leaving and upon other side of the molecule, nucleophile attacks.
The preparation of given compound from cyclopentenol is shown in Figure 8.
Want to see more full solutions like this?
Chapter 17 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- In this question, the product of the aldol condensation is shown. What would be the reactants for this product? Please provide a detailed explanation, as well as a drawing showing how the reactants will react to produce the product.arrow_forward7. Propene undergoes a hydration reaction with water in the presence of an acid. a. There are two possible products for this reaction, both with the formula C,H,O. Show their structural formulas and names. (A1, B2) SCH4UR Name: (answer for part a. here!) VER 3 2021-2022 b. Which of the two products do you predict will form. Explain your choice using details from your learning. (B3)arrow_forwardWhat are the major products of the following organic reaction? Please include a detailed explanation as well as a drawing as to how the reaction proceeds.arrow_forward
- What are the major products of the following reaction? Please provide a detailed explanation and a drawing to show how the reaction proceeds.arrow_forwardWhat are the major products of the following organic reaction? Please include a detailed explanation as well as a drawing as to how the reaction proceeds.arrow_forwardPredict the organic product that forms in the reaction below: H + гон OH H+ H+ ☑ O Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the product. In the drawing area below, draw the skeletal ("line") structure of the missing organic product X. Explanation Check Click and drag to start drawing a structure. S 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centearrow_forward
- In the analysis of Mg content in a 25 mL sample, a titration volume of 5 mL was obtained using 0.01 M EDTA. Calculate the Mg content in the sample if the Ca content is 20 ppmarrow_forwardPredict the organic products that form in the reaction below: H. H+ + OH H+ Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Explanation Check Click and drag to start drawing a structure. G X C © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Access +arrow_forward111 Carbonyl Chem Choosing reagants for a Wittig reaction What would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 3 1 2 2. n-BuLi • Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure. × ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Usearrow_forward
- A student proposes the transformation below in one step of an organic synthesis. There may be one or more reactants missing from the left-hand side, but there are no products missing from the right-hand side. There may also be catalysts, small inorganic reagents, and other important reaction conditions missing from the arrow. • Is the student's transformation possible? If not, check the box under the drawing area. . If the student's transformation is possible, then complete the reaction by adding any missing reactants to the left-hand side, and adding required catalysts, inorganic reagents, or other important reaction conditions above and below the arrow. • You do not need to balance the reaction, but be sure every important organic reactant or product is shown. + T X O O лет-ле HO OH HO OH This transformation can't be done in one step.arrow_forwardDetermine the structures of the missing organic molecules in the following reaction: X+H₂O H* H+ Y OH OH Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. X Sarrow_forwardPredict the major products of this organic reaction. If there aren't any products, because nothing will happen, check the box under the drawing area instead. No reaction. HO. O :☐ + G Na O.H Click and drag to start drawing a structure. XS xs H₂Oarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





