
Concept explainers
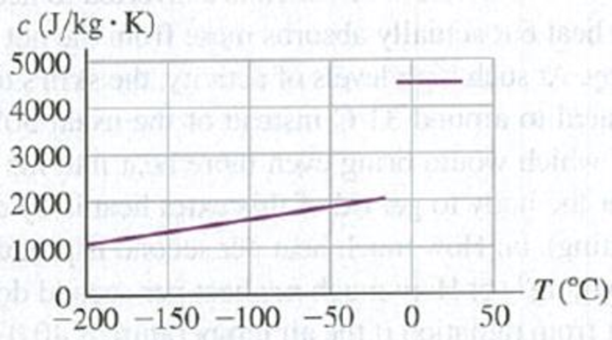
Careful measurements show that the specific heat of the solid phase depends on temperature (Fig. P17.117). How will the actual time needed for this cryoprotectant to come to equilibrium with the cold plate compare with the time predicted by using the values in the table? Assume that all values other than the specific heat (solid) are correct. The actual time (a) will be shorter; (b) will be longer; (c) will be the same; (d) depends on the density of the cryoprotectant.
Figure P17.117
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
University Physics with Modern Physics (14th Edition)
Additional Science Textbook Solutions
College Physics
Tutorials in Introductory Physics
Cosmic Perspective Fundamentals
Conceptual Physical Science (6th Edition)
Essential University Physics: Volume 1 (3rd Edition)
The Cosmic Perspective Fundamentals (2nd Edition)
- (a) What is the rate of heat conduction through the 3.00-cm-thick fur of a large animal having a I .40-m surface area? Assume that the animal's skin temperature is 32.0 , that the air temperature is 5.00 , and that has the same thermal conductivity as air. (b) What food intake will the animal need in one day to replace this heat transfer?arrow_forwardAn aluminum rod 0.500 m in length and with a cross sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 3(H) K. (a) If one-halt of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool, (b) If the circular surface of the upper end of the rod is maintained at 300 K. what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 YV/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forwardThe control system of the steam-jacketed kettle is shown in the figure. The system consists of a kettle through which water flows at a variable rate w (k9/s). The entering water is at temperature T; ("C), which may vary with time. The kettle water, which is well agitated, is heated by steam condensing in the jacket at temperature T, and pressure P, . The temperature of the water in the kettle is measured and transmitted to the controller. The output signal from the controller is used to change the stem position of the valve, which adjusts the flow of steam to the jacket w. |Computer/Controller The following assumptions are made for the kettle: • The hold-up volume of water in the kettle is constant. The thermal capacity of the kettle wall, which separates steam from water, is negligible compared with that of the Temperature-measuring element Water water in the kettle. The heat capacities of water cpw (K/kg°C) and the metal wall Cp,methal (K/kg°c) are constant. The density of water is…arrow_forward
- Help.... Its wintertime and you are in need of portable electrical heater for your room and you are comparing two different products. The first heater has metal grills made of aluminum and has dimensions of 0.75 m x 0.05 m x 0.01 m while the second heater has metal grills made of iron and has dimensions of 1.00 m x 0.03 m x 0.01 m. You wish to heat your room from 10 degrees Celsius to a nice toasty 28 degrees Celsius and your wall outlet provides 120 V. 1) What is the resistance of the aluminum and iron heater at 28 degrees Celsius? 2) What is the current for the aluminum and iron heater operating at 28 degrees Celsius? 3) If you wish to operate your heater for 4 hours and the cost of electricity is $0.12 per kilowatt-hour, which heater has cheaper operational costs?arrow_forwardAn experiment measures the temperature of a 200 g substance while steadily supplying heat to it. as shown the results of the experiment. What are (a) the specific heat of the liquid phase and (b) the heat of vaporization?arrow_forwardAir goes through a Brayton cycle gas turbine, entering the compressor at 11 °C and 92 kPa. When it reaches the turbine inlet, the air is at 807°C and 1.20 MPa. What is the net change in specific enthalpy of the working fluid after going through the compressor and heat addition processes? You may assume that the specific heat capacity G, is a constant 1.005 kJ-kg1-K1 throughout the whole cycle. Gjve your answer in kJ-kg to one decimal place.arrow_forward
- During an intense 30-minute fat-burning workout class, the instructor maintains an average power output of 2.0 x 10^2W, and she loses heat ot the environment at an average of rate of 2.5 x 10^2W. Ignoring heat loss due to perspiration, how much fat will the instrcutor lose during the workout? The enegry content of fat is 9.3 kcal/g.arrow_forwardNEED ANSWER ASAP At a temperature of 33.7 °C a 0.42-mm gap exists between the ends of the rods shown. At a later time when the temperature has reached 122.8°C, determine the change in length of the steel rod if L = 321.85 mm and M = 211 mm. Round off the final answer to four decimal places.arrow_forwardAn iron bar has original dimensions of 10 m x 2 cm x 2 cm. What is the amount of heat (Q) required to increase its length by 0.2 cm? Given: The density of copper is 7870 kg/m³, the specific heat is 449 J/kg K, and the linear expansion coefficient is 12 x 10-6 K-1 . Hint: Watch the assigned Clutch Prep video for PCQ-2! A. 17 kJ B. 31.5 kg C. 240 kJ D. 1.8 kJarrow_forward
- The basic principle of calorimetry states that: In an isolated system whose parts interact by heat exchange, the algebraic sum of the Q's for all parts of the system must be zero. Why is the algebraic sum of the Q's for all parts zero? In answering this question, assume the simplest case where the system is composed of just two parts A and B, that is, Qa + QB = 0.arrow_forwardA pronghorn antelope can run at a remarkable 18 m/sm/s for up to 10 minutes, almost triple the speed that an elite human runner can maintain. For a 32 kgkg pronghorn, this requires an astonishing 3.4 kWkW of metabolic power, which leads to a significant increase in body temperature. If the pronghorn had no way to exhaust heat to the environment, by how much would its body temperature increase during this run? (In fact, it will lose some heat, so the rise won't be this dramatic, but it will be quite noticeable, requiring adaptations that keep the pronghorn's brain cooler than its body in such circumstances.) Assume the efficiency of the pronghorn to be equal to that of human.arrow_forwardANSWER THE FF AND SHOW THE COMPLETE SOLUTION: a.) A Styrofoam cooler contains ice at 0 degrees Celsius. For the ice to melt into water at 0 degrees Celsius, it needs to take in 34 kJ of heat. How long would this take if the heat current through the cooler is 9.4 W? (answer for this is: 1hr) b.) A Styrofoam cooler contains ice at 0 degrees Celsius. The cooler is left outside where the temperature is 30 degrees Celsius. For the ice to melt into water at 0 degrees Celsius, it needs to take in 34 kJ of heat. If the cooler has a total wall area of 0.73 m^2, thickness 5.0 cm, and thermal conductivity 0.027 W/m·K, how long (in minutes) would it take for the ice to melt?arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning



