
a)

Interpretation:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how 2-bromo-4-nitrotoluene can be synthesized is to be stated.
Concept introduction:
In
Electrophilic substitution of di and trisubstituted benzenes follows three simple rules. (i) If the directing influence of both the substituents reinforce each other, a single product results. (ii) If the directing influences of both the substituent groups oppose each other, the most powerful activating group among them has the dominant influence but usually a mixture of products results. (iii) In meta disubstituted compounds, further substitution in between the groups occurs only rarely, due to steric reasons.
To state:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how 2-bromo-4-nitrotoluene can be synthesized.
b)

Interpretation:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how 1,3,5-trinitrobenzene can be synthesized is to be stated.
Concept introduction:
In aromatic electrophilic substitution reactions the nitro group is a meta directing group. Nitration of benzene gives nitrobenzene. Further nitration of nitrobenzene will introduce nitro groups into the two meta positions to yield the trinitro compound.
To state:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how 1,3,5-trinitrobenzene can be synthesized.
c)
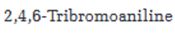
Interpretation:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how 2,4,6-tribromoaniline can be synthesized is to be stated.
Concept introduction:
In aromatic electrophilic substitution reactions, the amino group is an ortho and para directing group. Nitration of benzene gives nitrobenzene. Nitrobenzene can be reduced to aminobenzene (aniline). Aniline upon bromoination will yield 2,4,6-tribromoaniline.
To state:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how 2,4,6-tribromoaniline can be synthesized.
d)
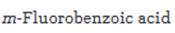
Interpretation:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how m-fluorobenzoic acid can be synthesized is to be stated.
Concept introduction:
In aromatic electrophilic substitution reactions the
To state:
Starting from benzene or toluene and assuming that the ortho and para isomers can be separated, how m-fluorobenzoic acid can be synthesized.
Trending nowThis is a popular solution!

Chapter 16 Solutions
EP ORGANIC CHEMISTRY,24 MONTH-OWLV2
- Part C A solution that is 0.040 M in HCIO4 and 0.046 M in HCI Express your answer numerically to two decimal places. ΜΕ ΑΣΦ ? pH = Submit Request Answer Part D A solution that is 1.08% HCl by mass (with a density of 1.01 g/mL) Express your answer numerically to three decimal places. ΜΕ ΑΣΦ -> 0 ? pH =arrow_forwardPredict the equilibrium arrows for the following reaction:*see imagearrow_forwardProvide the missing information for each of the two following reacitons: *see imagearrow_forward
- Draw an example of the following functional groups: *see imagearrow_forwardAldehydes and Ketones: Show the reaction conditions, and molecules, that connect the reactant to the product. A protecting group will be needed. *see imagearrow_forwardAldehydes and Ketones: Show the reaction conditions, and molecules, that connect the reactant to the product. *see imagearrow_forward
- Provide the missing information for each of the four reactions: *see imagearrow_forward6. Chlorine dioxide (CIO) is used as a disinfectant in municipal water-treatment plants. It decomposes in a first-order reaction with a rate constant of 14 s. How long would it take for an initial concentration of 0.06 M to decrease to 0.02 M? [6 pts]arrow_forwardIf possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ẞ carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. en HO OHarrow_forward
- Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the intermediate and product of this hydrohalogenation reaction. Include all lone pairs and charges as appropriate. Br Select to Draw 51°F Sunny esc F1 HBr Select to Draw 1,2-hydride shift Br Select to Draw Q Search F2 F3 F4 1 2 # # 3 DII L F5 F6 F tA $ % Λarrow_forwardplease help i cant find the article to even startarrow_forwardWhat are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





