
The pV diagram in Figure 16.19 shows a
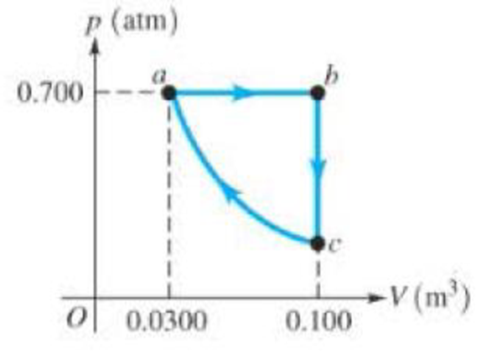
Figure 16.19
Problem 43.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
College Physics (10th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers with Modern Physics
Tutorials in Introductory Physics
Conceptual Integrated Science
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
Physics (5th Edition)
University Physics with Modern Physics (14th Edition)
- The pV diagram in (Figure 1) shows a process abc involving 0.550 mol of an ideal gas. For related problem-solving tips and strategies, you may want to view a Video Tutor Solution of A cyclic process. Figure 8.0 6.0 4.0 p(Pax 105) 2.0 O a 0.020 0.040 0.060 b 0.080 1 of 1 V(m³) ▶ Part A What was the temperature of this gas at points a, b, and c? Enter your answers separated by commas. Ta, Tb, Tc= 535,9355,14968 V Submit Previous Answers Request Answer Part B X Incorrect; Try Again; 5 attempts remaining Part C ΑΣΦ ΑΣΦ Q = 3.5104 How much heat had to be put in during the process to increase the internal energy of the gas by 1.00×104 J ? Express your answer in joules. Provide Feedback Submit Previous Answers Request Answer 國」? ? A p V diagram of a two-step process. X Incorrect; Try Again; 4 attempts remaining K Next >arrow_forwardCan you please answer number 8 and show all of the stepsarrow_forwardA gas sample undergoes the cyclic process ABCA shown in the figure (Figure 1), where AB lies on an adiabat (a specific-heat ratio of y = 1.4). The pressure at point A is 56 kPa. Figure Pressure, p 0 B 2 3 4 Volume, V (L) A 5 1 of 1 Part A Find the pressure at B. Express your answer in kilopascals. PB 530 kPa Submit Part B W Correct Find the net work done on the gas. Express your answer in joules. = Previous Answers Submit VE ΑΣΦ Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Return to Assignment Provide Feedback ? Jarrow_forward
- please helparrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 212 J when the gas goes from I to F along the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V (liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B (2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I (2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F (4,1). (a) What is the change in internal energy of the gas? Use the relations between various features of the graph and the work done on the gas to find the energy added by work and then use your result to find the change in internal energy of the gas. J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in…arrow_forwardPlease answer the following and show the complete solution: ANSWERS: a. 3000J b. 1000Jarrow_forward
- A gas follows the pV trajectory shown in Figure 16.2. How much work is done per cycle by the gas if po= 6.3 atm? Figure 16.2 P (atm) 3Po 2Po Po V (cm) 600 0- 0 400 200 D) 260 J C) 510 J B) 1300 J A) 1000 Jarrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 302 J when the gas goes from I to Falong the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V(liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B(2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I(2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F(4,1). (a) What is the change in internal energy of the gas?J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in internal energy?Jarrow_forwardCompressed air can be pumped underground into huge caverns as a form of energy storage. The volume of a cavern is 6.3 x 105 m³, 5 and the pressure of the air in it is 7.4 × 106 Pa. Assume that air is a diatomic ideal gas whose internal energy U is given by U = nRT. If one home uses 30.0 kWh of energy per day, how many homes could this internal energy serve for one day?arrow_forward
- 0.0037 mol of gas undergoes the process shown in (Figure 1). For the steps and strategies involved in solving a similar problem, you may view a Video Tutor Solution. Figure p (atm) 3 2 1 0 0 f 100 200 300 V (cm³) < 1 of 1 Part A What type of process is this? isothermal O isobaric constant-volume Submit Part B T₁ = What is the initial temperature? Express your answer in degrees Celsius. Submit Part C Tf = Request Answer Submit VE ΑΣΦ What is the final temperature? Express your answer in degrees Celsius. Request Answer ΑΣΦ Request Answer www. ? ? Oarrow_forwardPlease look at the picture... 0.05 mole of helium is expanded from 1 L to 2 L while pressure is kept constant at 1 atm, as demonstrated in the PV diagram in the picture. Question A: What is the pressure of the gas in this process? Please answer in N/m^2 Question B: What is the initial temperature of the gas? Please answer in kelvin Question C: What is the final temperature of the gas? Please answer in kelvinarrow_forwardThermodynamics, put units in the solutionarrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

