
Concept explainers
a Draw a pH titration curve that represents the titration of 25.0 mL of 0.15 M propionic acid. CH3CH2COOH, by the addition of 0.15 M KOH from a buret. Label the axes and put a scale on each axis. Show where the equivalence point and the buffer region are on the titration curve. You should do calculations for the 0%, 50%, 60%, and 100% titration points. b Is the solution neutral, acidic, or basic at the equivalence point? Why?
(a)
Interpretation:
For titration of 25.0 mL of 0.15 M propionic acid,
A pH titration curve showing the equivalence point and buffer region has to be drawn
- (a) The pH of the titration points for the 0%, 50%, 60% and 100% has to be calculated
- (b) Whether the solution at the equivalence point is neutral, acidic or basic has to be explained
Concept Introduction:
Equivalence point:
The equivalence point in titration is the point where the amount of standard titrant solution (in moles) and the unknown concentration analyte solution (in moles) becomes equal.
In other words, the equivalence point is the point obtained in a titration once a stoichiometric amount of reactant has been added.
Relationship between pH and pOH:
Answer to Problem 16.120QP
A pH titration curve showing the equivalence point and buffer region is given in Figure 1 as follows,
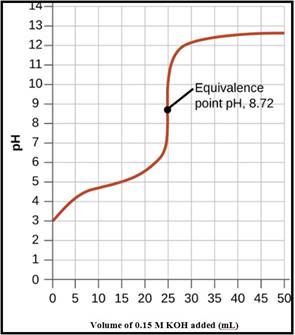
Figure 1
(a)
The pH at the 0% titration point is 2.85
The pH at the 50% titration point is 4.89
The pH at the 60% titration point is 5.06
The pH at the 100% titration point is 8.88
Explanation of Solution
To Draw: A pH titration curve showing the equivalence point and buffer region
The pH titration curve for the titration of 0.15 M propionic acid with 0.15 M
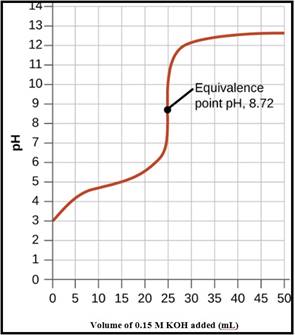
Figure 1
(a)
To Calculate: The pH of the titration points for the 0%, 50 %, 60% and 100%
Given data:
Titration of 0.15 M propionic acid with 0.15 M
pH at the 0% titration point:
Construct an equilibrium table with x as unknown concentration
Consider propionic acid as
|
|
|||
| Initial |
0.15
0.15-x |
0.00 | 0.00 |
| Change |
|
|
|
| Equilibrium |
x | x | |
Substitute equilibrium concentrations into the equilibrium-constant equation.
The
Assume x is negligible compared to 0.15 M
Therefore, the concentration of hydronium ion
In the end, pH is calculated as follows,
Therefore, the pH at the 0% titration point is 2.85
pH at the 50% titration point:
The pH is calculated as follows,
Therefore, the pH at the 50% titration point is 4.89
pH at the 60% titration point:
For convenience, express the concentrations as percents.
Substitute the concentrations into the equilibrium expression.
Therefore, the concentration of hydronium ion
In the end, pH is calculated as follows,
Therefore, the pH at the 60% titration point is 5.06
pH at the 100% titration point:
The salt that got produced has undergone a twofold dilution.
Therefore,
Construct an equilibrium table with x as unknown concentration
|
|
|||
| Initial |
0.0750
0.0750-x |
0.00 | 0.00 |
| Change |
|
|
|
| Equilibrium |
x | x | |
Now, calculate
Substitute into the equilibrium constant expression.
Here, x gives the concentration of hydroxide ion,
The pH is calculated as follows,
Therefore, the pH at the 100% titration point is 8.88
The pH at the 0% titration point was calculated as 2.85
The pH at the 50% titration point was calculated as 4.89
The pH at the 60% titration point was calculated as 5.06
The pH at the 100% titration point was calculated as 8.88
(b)
Interpretation:
For titration of 25.0 mL of 0.15 M propionic acid,
A pH titration curve showing the equivalence point and buffer region has to be drawn
- (a) The pH of the titration points for the 0%, 50%, 60% and 100% has to be calculated
- (b) Whether the solution at the equivalence point is neutral, acidic or basic has to be explained
Concept Introduction:
Equivalence point:
The equivalence point in titration is the point where the amount of standard titrant solution (in moles) and the unknown concentration analyte solution (in moles) becomes equal.
In other words, the equivalence point is the point obtained in a titration once a stoichiometric amount of reactant has been added.
Relationship between pH and pOH:
Answer to Problem 16.120QP
The solution at the equivalence point is basic
Explanation of Solution
To Explain: Whether the solution at the equivalence point is neutral, acidic or basic
As a result of titration Potassium propionate salt is produced.
Potassium propionate is the salt of a weak acid and a strong base.
Propionate ion reacts with water to produce hydroxide ions.
Therefore, the given solution is basic
The solution at the equivalence point was found as basic
Want to see more full solutions like this?
Chapter 16 Solutions
Student Solutions Manual for Ebbing/Gammon's General Chemistry, 11th
- Choose a number and match the atomic number to your element on the periodic table. For your element, write each of these features on a side of your figure. 1. Element Name and symbol 2. Family and group 3. What is it used for? 4. Sketch the Valence electron orbital 5. What ions formed. What is it's block on the periodic table. 6. Common compounds 7. Atomic number 8. Mass number 9. Number of neutrons- (show calculations) 10. Sketch the spectral display of the element 11.Properties 12. Electron configuration 13. Submit a video of a 3-meter toss in slow-moarrow_forward[In this question, there are multiple answers to type in a "fill-in-the-blank" fashion - in each case, type in a whole number.] Consider using Slater's Rules to calculate the shielding factor (S) for the last electron in silicon (Si). There will be electrons with a 0.35 S-multiplier, electrons with a 0.85 S-multiplier, and electrons with a 1.00 S-multiplier.arrow_forwardProvide the unknown for the given data.arrow_forward
- Draw the Lewis structures of two methanol (CH3OH) molecules and depict hydrogenbonding between them with dashed lines. Show all lone pairs. Provide a thorough analysis to apply concept idea into other problems.arrow_forwardSteps and explanation please.arrow_forwardHow could you distinguish between each pair of compounds below using IR? For each pair citeone bond and it’s frequency that you could use to distinguish between them. Please provide thorough analysis to apply into further problems.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





