
a)
Interpretation:
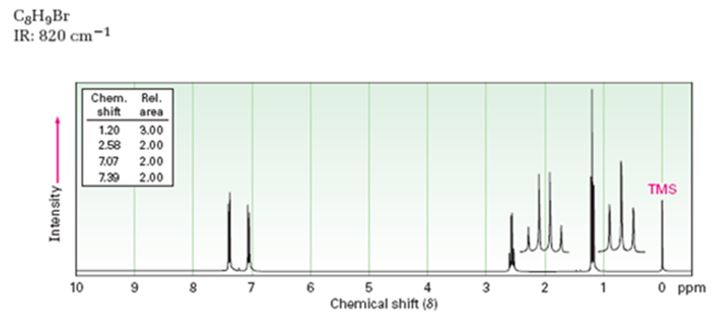
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C8H9Br I.R: 820 cm-1.
1HNMR spectrum: 7.07δ (2H, doublet); 7.39δ (2H, doublet); 2.58δ (2H, quartet); 1.20δ (3H, triplet).
Concept introduction:
In 1HNMR spectrum
In I.R, the o-disubstituted benzenes absorb around 735-770 cm-1, m-disubstituted benzenes absorb around 690-710 cm-1 and p-disubstituted benzenes absorb around 810-840 cm-1.
To propose:
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C8H9Br I.R: 820 cm-1.
1HNMR spectrum: 7.07δ (2H, doublet); 7.39δ (2H, doublet); 2.58δ (2H, quartet); 1.20δ (3H, triplet).
Answer to Problem 47AP
A structure for the compound with M.F = C8H9Br with spectral characteristics: I.R: 820 cm-1, 1HNMR spectrum: 7.07δ (2H, doublet); 7.39δ (2H, doublet); 2.58δ (2H, quartet); 1.20δ (3H, triplet) is
Explanation of Solution
The molecular formula of the compound is C8H9Br.
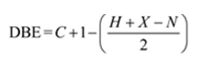
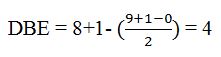
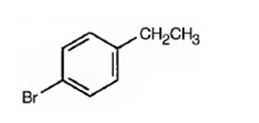
Thus the compound has four unsaturation units like double bonds and/or rings. The two doublets at 7.07δ and 7.39δ each corresponding to two protons indicate the compound is aromatic and each proton responsible for the doublet has a proton on the adjacent carbon. The two proton quartet at 2.58δ (benzylic) and three proton triplet at 1.20δ (primary) could be accounted for only if an ethyl substituent is present on the ring along with bromine. The I.R absorption at 820 cm-1 indicates that the ethyl group and bromine are arranged in para positions. Hence the compound is p-bromoethyl benzene.
A structure for the compound with M.F=C8H9Br with spectral characteristics: I.R: 820 cm-1, 1HNMR spectrum: 7.07δ (2H, doublet); 7.39δ (2H, doublet); 2.58δ (2H, quartet); 1.20δ (3H, triplet) is

b)
Interpretation:
A structure for the compound whose 1HNMR spectrum given is to be proposed.
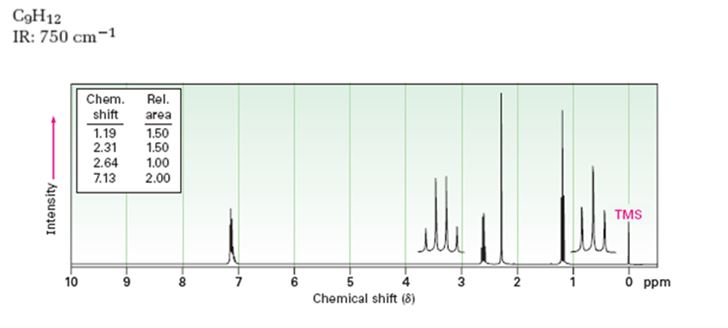
Given: M.F = C9H12 I.R: 750 cm-1
1HNMR spectrum: 7.13δ (4H, broad); 2.64δ (2H, quartet); 2.31δ (3H, singlet); 1.19δ (3H, triplet).
Concept introduction:
In 1HNMR spectrum aromatic protons give a broad peak in the range 6.5δ-8.0δ, the primary alkyl protons around 0.7δ-1.3δ, the secondary alkyl protons around 1.2δ-1.6δ, and a tertiary alkyl protons in between 1.4δ-1.8δ. The multiplicity of a signal gives an idea about the protons present in the adjacent carbons.
In I.R, the o-disubstituted benzenes absorb around 735-770 cm-1, m-disubstituted benzenes absorb around 690-710 cm-1and p-disubstituted benzenes absorb around 810-840 cm-1.
To propose:
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C9H12 I.R: 750 cm-1.
1HNMR spectrum: 7.13δ (4H, broad); 2.64δ (2H, quartet); 2.31δ (3H, singlet); 1.19δ (3H, triplet).
Answer to Problem 47AP
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C9H12, I.R: 750 cm-1, 1HNMR spectrum: 7.13δ (4H, broad); 2.64δ (2H, quartet); 2.31δ (3H, singlet); 1.19δ (3H, triplet) is
Explanation of Solution
The molecular formula of the compound is C9H12.
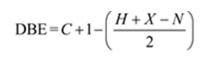
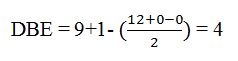
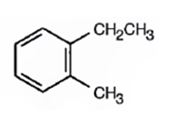
Thus the compound has four unsaturation units like double bonds and/or rings. The four proton broad band at 7.13δ indicates that the compound is aromatic with two substituent groups attached to the benzene ring. The two proton quartet at 2.64δ (benzylic) and three proton triplet at 1.19δ (primary alkyl) could be accounted for only if an ethyl group is present on the ring. The three proton singlet at 2.31δ (benzylic) can be attributed to a methyl group attached to the ring. Thus the two substituent groups attached to the benzene ring are ethyl and methyl. The I.R absorption at 750 cm-1 requires that the ethyl and methyl groups to be arranged in ortho positions. Hence the compound is o-ethyltoluene.
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C9H12, I.R: 750 cm-1, 1HNMR spectrum: 7.13δ (4H, broad); 2.64δ (2H, quartet); 2.31δ (3H, singlet); 1.19δ (3H, triplet) is

c)
Interpretation:
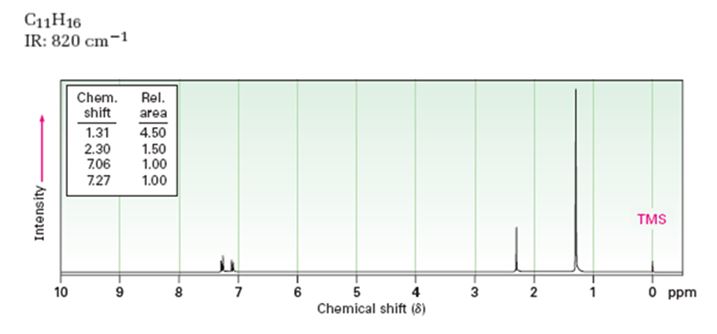
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C11H16 I.R: 820 cm-1.
1HNMR spectrum: 7.27δ (2H, doublet); 7.06δ (2H, doublet); 2.30δ (3H, singlet); 1.31δ (9H, singlet).
Concept introduction:
In 1HNMR spectrum aromatic protons give a broad peak in the range 6.5δ-8.0δ, the primary alkyl protons around 0.7δ-1.3 δ, the secondary alkyl protons around 1.2δ-1.6δ, and a tertiary alkyl protons in between 1.4δ-1.8δ. The multiplicity of a signal gives an idea about the protons present in the adjacent carbons.
In I.R, the o-disubstituted benzenes absorb around 735-770 cm-1, m-disubstituted benzenes absorb around 690-710 cm-1 and p-disubstituted benzenes absorb around 810-840 cm-1.
To propose:
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C11H16 I.R: 820 cm-1.
1HNMR spectrum: 7.27δ (2H, doublet); 7.06δ (2H, doublet); 2.30δ (3H, singlet); 1.31δ (9H, singlet).
Answer to Problem 47AP
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C11H16; I.R: 820 cm-1; 1HNMR spectrum: 7.27δ (2H, doublet); 7.06δ (2H, doublet); 2.30δ (3H, singlet); 1.31δ (9H, singlet).
Explanation of Solution
The molecular formula of the compound is C11H16.
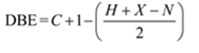
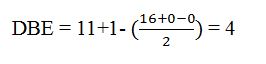
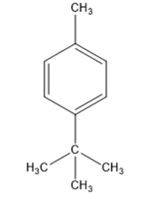
Thus the compound has four unsaturation units like double bonds and/or rings. The two doublets at 7.27δ and 7.06δ each corresponding to two protons indicate the compound is aromatic and each proton responsible for the doublet has a proton on the adjacent carbon. The three proton singlet at 2.58δ (benzylic) could be accounted for a methyl group attached to the benzene ring. The remaining four carbons and nine hydrogens indicate the presence of a tert-butyl group attached to the benzene ring which is confirmed by the nine proton singlet at 1.31δ (primary alkyl). Thus methyl and tert-butyl are the two substituent groups on the benzene ring. The I.R absorption at 820 cm-1 indicates that the two substituent groups are arranged in para positions. Hence the compound is p-t-butyltoluene.
A structure for the compound whose 1HNMR spectrum given is to be proposed. Given: M.F = C11H16; I.R: 820 cm-1; 1HNMR spectrum: 7.27δ (2H, doublet); 7.06δ (2H, doublet); 2.30δ (3H, singlet); 1.31δ (9H, singlet).

Want to see more full solutions like this?
Chapter 15 Solutions
ORGANIC CHEMISTRY W/OWL
- Can you explain how I get these here and show the steps plz?arrow_forwardGive the IUPAC name for this compound Hydrocarbon Condensed Formulas Hint C2H5 CH2CH3 expand that in all the formula Part A: (CH3)2CHCH(C2H5)CH2CH2CH3 Give the IUPAC name for this compound. Part B: CH2=C(C2H5)CH2CH2CH3 Give the IUPAC name for this compound. Part C: (CH3)2C=CHC(C2H5)=CH2 Give the IUPAC name for this compound. Part D: CH3C=CCH(C2H5)2 Give the IUPAC name for this compound. Part E: (CH3)3CC=CCH2CH=C(CH3)2arrow_forwardSelect/ Match the correct letter from the image below for the IUPAC names given below: A B C D 3 E F G H K L Part 1. 4-methylheptane For example.mmmm Answer Letter H _for part 1 Part 2. 2,4-dimethylhexane Part 3. 2,3-dimethylpentane Part 4. 2,2-dimethylhexane Part 5. 2-ethyl-1,1,3,3-tetramethylcyclopentane Part 6. 3-ethyl-2-methylpentanearrow_forward
- Can u show the process as to how to get these?arrow_forwardSketch the expected 'H NMR spectra for the following compound. Label all of the H's in the structure and the corresponding signal for the spectra you sketch. Make sure you include the integration value and the splitting pattern for each signal Indicate how many signals you would expect in the 13C NMRarrow_forwardUse IUPAC naming rules to name the following hydrocarbon compounds: CH2-CH3 | a) CH-CH-CH2-CH-CH-CH3 b) | CH2 CH3 | CH3 CH3 \ / C=C H 1 H CH2-CH3 c) d) CH=C-CH3 e) CH3-CH2-CH2-CH=CH-CH3 f) CH2=CH-CH2-CH=CH-CH3 g) CH3-CH2-C = C-CH2-CH3 h)arrow_forward
- Q5 Name the following : a. b. C. d. e.arrow_forward25. Predict the major product of the following reaction. 1 equivalent of each of the starting materials was used. H₂C CH3 CH3 H3C H3C H3C. CH2 + H3C. heat CH3 CH H.C. CH3 H.C H.C CH3 CH CH3 CH3 A B C Earrow_forwardFind chemical structures based on the below information. a) Chemical formula C6H8O Compound is aromatic plus has two 1H NMR peaks that integrated for 3 each that are singlets (it could have more peaks in the 1H NMR b) Chemical Formula: C6H100 Compounds is conjugated 'H NMR has a signal that integrates for 6 and is a doublet IR spectra has a signal at 1730 cm-1arrow_forward
- Jaslev Propose a synthesis of the following starting from benzene and any other reagents and chemicals. No mechanisms are required. Indicate the condition for each step plus the major product for each step. More than two steps are required. Step 1 Step 2 مہد Brarrow_forwardPart C: The line formula for another branched alkane is shown below. i. In the IUPAC system what is the root or base name of this compound? ii. How many alkyl substituents are attached to the longest chain? iii. Give the IUPAC name for this compound.arrow_forwardPart D: Draw the Structural Formula for 4-ethyl-2-methylhexane Part E. Draw the Structural Formula for 1-chloro-3,3-diethylpentane (Chloro = Cl)arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


