
CHEMISTRY THE CENTRAL SCIENCE >EBOOK<
14th Edition
ISBN: 9780136873891
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 15, Problem 1E
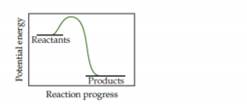
- Based on the following energy profile, predict whether kf > kr or kf < kr.
- Using Equation 15.5, predict whether the equilibrium constant for the process is greater than 1 or less than 1. [Section 15.1]
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
=Naming benzene derivatives
Name these organic compounds:
structure
C1
CH3
name
☐
CH3
ப
C1
×
☐
Blocking Group are use to put 2 large sterically repulsive group ortho. Show the correct sequence toconnect the reagent to product with the highest yield possible. * see image **NOTE: The compound on the left is the starting point, and the compound on the right is the final product. Please show the steps in between to get from start to final, please. These are not two different compounds that need to be worked.
I dont understand this.
Chapter 15 Solutions
CHEMISTRY THE CENTRAL SCIENCE >EBOOK<
Ch. 15.2 - Prob. 15.1.1PECh. 15.2 - Prob. 15.1.2PECh. 15.2 - Prob. 15.2.1PECh. 15.2 - Prob. 15.2.2PECh. 15.3 - Prob. 15.3.1PECh. 15.3 - Practice Exercise 2 For the reaction H2 (g) + I2...Ch. 15.3 - Prob. 15.4.1PECh. 15.3 - Prob. 15.4.2PECh. 15.4 - Prob. 15.5.1PECh. 15.4 - Prob. 15.5.2PE
Ch. 15.4 - Practice Exercise 1
If 8.0 g of NH4HS(s)...Ch. 15.4 - Prob. 15.6.2PECh. 15.5 - Practice Exercise 1
A mixture of gaseous sulfur...Ch. 15.5 - Prob. 15.7.2PECh. 15.5 - Practice Exercise 1 In Section 15.1, we discussed...Ch. 15.5 - Practice Exercise 2
The gaseous compound BrCl...Ch. 15.6 - Prob. 15.9.1PECh. 15.6 - Practice Exercise 2 At 1000 k, the value of Kp for...Ch. 15.6 - Prob. 15.10.1PECh. 15.6 - Prob. 15.10.2PECh. 15.6 - Practice Exercise 1 For the equilibrium Br2(g) +...Ch. 15.6 - Prob. 15.11.2PECh. 15.7 - Practice Exercise 1 For the reaction 4 NH3(g) + 5...Ch. 15.7 - Prob. 15.12.2PECh. 15 - Prob. 1DECh. 15 - Based on the following energy profile, predict...Ch. 15 - 15.2 The following diagrams represent a...Ch. 15 - Prob. 3ECh. 15 - Prob. 4ECh. 15 - Prob. 5ECh. 15 - 15.6 Ethene (C2H4) reacts with healogens (X2) by...Ch. 15 - When lead(IV) oxide is heated above 300 O C, it...Ch. 15 - Prob. 8ECh. 15 - The reactin A2(g) + B(g) + A(g) + AB(g) has an...Ch. 15 - Prob. 10ECh. 15 - Prob. 11ECh. 15 - The following graph represents the yield of the...Ch. 15 - Suppose that the gas-phase reactions A B and B A...Ch. 15 - Prob. 14ECh. 15 - Prob. 15ECh. 15 - Write the expression for KC for the following...Ch. 15 - When the following reaction come to equilibrium,...Ch. 15 - Prob. 18ECh. 15 - Prob. 19ECh. 15 - Prob. 20ECh. 15 - If Kc = 0.042 for PC13(g) + C12 (g) PC15 (g) at...Ch. 15 - Prob. 22ECh. 15 - 15.23 The equilibrium constant for the...Ch. 15 - Prob. 24ECh. 15 - Prob. 25ECh. 15 - Prob. 26ECh. 15 - The following equilibria were attained at 823 K:...Ch. 15 - Consider the equilibrium N2(g) + O2(g) + Br2(g) 2...Ch. 15 - Mercury(I) oxide decomposes into elemental mercury...Ch. 15 - Prob. 30ECh. 15 - Prob. 31ECh. 15 - Prob. 32ECh. 15 - Prob. 33ECh. 15 - Phosphorus trichloride gas and chlorine gas react...Ch. 15 - A mixture of 0.10 mol of NO, 0.050 mol of H2, and...Ch. 15 - Prob. 36ECh. 15 - A mixture of 0.2000 mol of CO2, 0.1000 mol of H2,...Ch. 15 - 15.38 A flask is charged with 1.500 atm of N2O4(g)...Ch. 15 - Prob. 39ECh. 15 - Prob. 40ECh. 15 - a. If QC KC, in which direction will a reaction...Ch. 15 - Prob. 42ECh. 15 - At 100 OC , the equilibrium constant for the...Ch. 15 - 15.44 As shown in Table 15.2, KP for the...Ch. 15 - At 100 C, K = 0.078 for the reaction SO2Cl2 (g) ...Ch. 15 - Prob. 46ECh. 15 - Prob. 47ECh. 15 - Prob. 48ECh. 15 - At 800 k, the equilibrium constant for I2 (g) ...Ch. 15 - Prob. 50ECh. 15 - At 2000 OC, the equilibrium constant for the...Ch. 15 - For the equilibrium Br2 (g) + Cl2 (g) 2BrCl(g) At...Ch. 15 - At 373 k, Kp = 0.416 for the equilibrium 2NOBr (g)...Ch. 15 - At 218 oC, KC= 1.2 X 10-4 for the equilibrium NH4...Ch. 15 - Prob. 55ECh. 15 - At 80 oC, K =1.87 X 10-3 for the reaction PH3 BCl3...Ch. 15 - Prob. 57ECh. 15 - Prob. 58ECh. 15 - Prob. 59ECh. 15 - Prob. 60ECh. 15 - Consider the following equilibrium for which H<0...Ch. 15 - Prob. 62ECh. 15 - 15.63 How do the following changes affect the...Ch. 15 - Prob. 64ECh. 15 - Consider the following equilibrium between oxides...Ch. 15 - Prob. 66ECh. 15 - Ozone, O3, decomposes to molecular oxygen in the...Ch. 15 - Prob. 68ECh. 15 - Prob. 69ECh. 15 - 15.70 True or false: When the temperature of an...Ch. 15 - Prob. 71AECh. 15 - Prob. 72AECh. 15 - 15.73 A mixture of CH4 and H2O is passed over a...Ch. 15 - Prob. 74AECh. 15 - Prob. 75AECh. 15 - Prob. 76AECh. 15 - Prob. 77AECh. 15 - Prob. 78AECh. 15 - Prob. 79AECh. 15 - For the equilibrium PH3BCI3 (s) PH3 (g) + BCI3...Ch. 15 - Prob. 81AECh. 15 - Prob. 82AECh. 15 - Prob. 83AECh. 15 - At 900 o C, Kc = 0.0108 for the reaction CaCO3(g) ...Ch. 15 - Prob. 85AECh. 15 - The equilibrium constant Kc for C(s) +CO2 2CO(g)...Ch. 15 - Prob. 87AECh. 15 - Le Chatelier noted that many industrial processes...Ch. 15 - Prob. 89AECh. 15 - Prob. 90AECh. 15 - [15.91] An equilibrium mixture of H2, I2, and HI...Ch. 15 - Consider the hypothetical reaction A(g) + 2B(g) 2...Ch. 15 - Prob. 93AECh. 15 - Prob. 94AECh. 15 - Prob. 95IECh. 15 - The following equilibria were measured at 823 K:...Ch. 15 - Prob. 97IECh. 15 - Prob. 98IECh. 15 - At 800 K, the equilibrium constant for the...Ch. 15 - Prob. 100IECh. 15 - Prob. 101IECh. 15 - Prob. 102IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Can you please explain this prooblem to me, show me how the conjugation is added, did I add them in the correct places and if so please show me. Thanks!arrow_forwardBasic strength of organic bases.arrow_forwardNucleophilic Aromatic Substitution: What is the product of the reaction? What is the name of the intermediate complex? *See imagearrow_forward
- The answer here says that F and K have a singlet and a doublet. The singlet and doublet are referring to the H's 1 carbon away from the carbon attached to the OH. Why don't the H's two carbons away, the ones on the cyclohexane ring, cause more peaks on the signal?arrow_forwardDraw the Birch Reduction for this aromatic compound and include electron withdrawing groups and electron donating groups. *See attachedarrow_forwardShow the correct sequence to connect the reagent to product. * see imagearrow_forward
- Blocking Group are use to put 2 large sterically repulsive group ortho. Show the correct sequence toconnect the reagent to product with the highest yield possible. * see imagearrow_forwardElimination-Addition: What molecule was determined to be an intermediate based on a “trapping experiment”? *please solve and see imagearrow_forwardShow the correct sequence to connect the reagent to product. * see imagearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Chemical Equilibria and Reaction Quotients; Author: Professor Dave Explains;https://www.youtube.com/watch?v=1GiZzCzmO5Q;License: Standard YouTube License, CC-BY