
(a)
Interpretation:
The statement “benzoic acid is an aliphatic
(a)
Answer to Problem 6MCP
The given statement is false because benzoic acid is an
Explanation of Solution
Benzoic acid is the simplest aromatic carboxylic acid. It is aromatic due to the presence of a benzene ring in its chemical structure.
The structure of benzoic acid is,
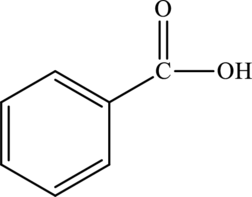
The statement “benzoic acid is an aliphatic carboxylic acid” is false because it is an aromatic carboxylic acid due to the presence of benzene ring.
(b)
Interpretation:
The statement “benzoic acid has a lower boiling point than benzaldehyde” has to be indicated as true or false. The reason for the false statement has to be described.
(b)
Answer to Problem 6MCP
The given statement is false because benzoic acid has higher boiling point than benzaldehyde due to strong intermolecular hydrogen bonding and strong dipole-dipole interactions.
Explanation of Solution
Benzoic acid is a carboxylic acid and benzaldehyde is an aldehyde.
Carboxylic acids have strong intermolecular hydrogen bonding with each other and strong dipole-dipole attractions.
The presence of polar carboxyl group and intermolecular hydrogen bonding makes these acids have higher boiling points. The presence of dimers increases the strength of the van der Waals dispersion forces, which makes them have high boiling points.
Therefore, benzoic acid has higher boiling point than benzaldehyde due to strong intermolecular hydrogen bonding and strong dipole-dipole interactions.
Hence, the statement “benzoic acid has a lower boiling point than benzaldehyde” is false.
(c)
Interpretation:
The statement “benzoic acid reacts with ethanol to form ethyl benzoate” has to be indicated as true or false. The reason for the false statement has to be described.
(c)
Answer to Problem 6MCP
The given statement is true.
Explanation of Solution
Benzoic acid on reaction with ethyl alcohol produces ethyl benzoate and water.
The
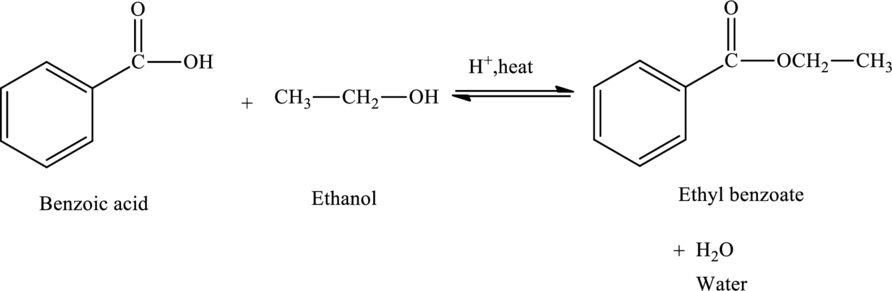
The statement “benzoic acid reacts with ethanol to form ethyl benzoate” is true.
(d)
Interpretation:
The statement “benzoic acid has molecular formula of
(d)
Answer to Problem 6MCP
The given statement is false because the molecular formula of
Explanation of Solution
Benzoic acid is an organic compound with molecular formula of
Hence, the statement “benzoic acid has molecular formula of
(e)
Interpretation:
The statement “benzoic acid is structural isomer of xylene” has to be indicated as true or false. The reason for the false statement has to be described.
(e)
Answer to Problem 6MCP
The given statement is false because the structural isomers of benzoic acids are tropolone, salicyladehyde,
Explanation of Solution
Structural isomers are those with same molecular formula but differ in arrangement of atoms.
The molecular formula of benzoic acid
The molecular formula of xylene is
Both these are not structural isomers with each other.
Hence, the statement “benzoic acid is structural isomer of xylene” is false because the structural isomers of benzoic acids are tropolone, salicyladehyde,
(f)
Interpretation:
The statement “benzoic acid is more water soluble in phenol” has to be indicated as true or false. The reason for the false statement has to be described.
(f)
Answer to Problem 6MCP
The given statement is false because phenol is more water soluble than benzoic acid due to its ability to form hydrogen bond with water molecules.
Explanation of Solution
Phenols are water soluble to some extent. This is due to its ability to form hydrogen bonding with water molecules. However, the large part of phenol molecule is the phenyl group that is non-polar and hence its solubility is limited in water.
Whereas, benzoic acid is insoluble in water because of the presence larger nonpolar benzene ring in the structure that cannot be water-soluble.
The statement “benzoic acid is more water soluble in phenol” is false because phenol is more water soluble than benzoic acid due the ability of phenols to form hydrogen bond with water molecules.
(g)
Interpretation:
The statement “sodium benzoate is food preservative and is produced by neutralization of benzoic acid with sodium hydroxide” has to be described as true or false. If the statement is false, the reason that it is false has to be described.
(g)
Answer to Problem 6MCP
The given statement is true.
Explanation of Solution
The reaction of benzoic acid with sodium hydroxide yields sodium salt of benzoic acid and water. The protons of the acid are removed by the
The reaction is,
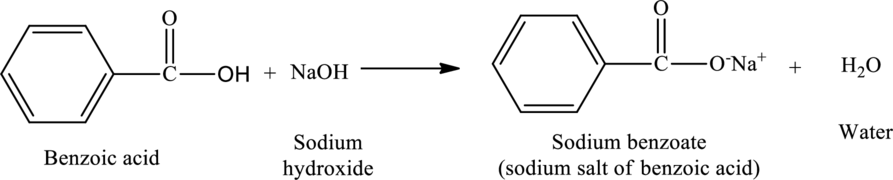
The statement “sodium benzoate is food preservative and is produced by neutralization of benzoic acid with sodium hydroxide” is true.
Want to see more full solutions like this?
Chapter 14 Solutions
GENERAL, ORGANIC, BIOCHEM (LL W/ ACCESS)
- The rate coefficient of the gas-phase reaction 2 NO2 + O3 → N2O5 + O2 is 2.0x104 mol–1 dm3 s–1 at 300 K. Indicate whether the order of the reaction is 0, 1, or 2.arrow_forward8. Draw all the resonance forms for each of the following molecules or ions, and indicate the major contributor in each case, or if they are equivalent. (4.5 pts) (a) PH2 سمةarrow_forward3. Assign absolute configuration (Rors) to each chirality center. a. H Nitz C. он b. 0 H-C. C H 7 C. ་-4 917-417 refs H 1つ ८ ડુ d. Но f. -2- 01 Ho -OH 2HNarrow_forward
- How many signals do you expect in the H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red. Note for advanced students: In this question, any multiplet is counted as one signal. Number of signals in the 'H NMR spectrum. For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in top molecule For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. No additional Hs to color in bottom moleculearrow_forwardIn the drawing area below, draw the major products of this organic reaction: 1. NaOH ? 2. CH3Br If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area instead. No reaction. Click and drag to start drawing a structure. ☐ : A คarrow_forwardPredict the major products of the following organic reaction: NC Δ ? Some important Notes: • Draw the major product, or products, of the reaction in the drawing area below. • If there aren't any products, because no reaction will take place, check the box below the drawing area instead. • Be sure to draw bonds carefully to show important geometric relationships between substituents. Note: if your answer contains a complicated ring structure, you must use one of the molecular fragment stamps (available in the menu at right) to enter the ring structure. You can add any substituents using the pencil tool in the usual way. Click and drag to start drawing a structure. Х аarrow_forward
- Predict the major products of this organic reaction. Be sure you use dash and wedge bonds to show stereochemistry where it's important. + ☑ OH 1. TsCl, py .... 文 P 2. t-BuO K Click and drag to start drawing a structure.arrow_forwardConsider this organic reaction: ( Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. Click and drag to start drawing a structure. Х : а ค 1arrow_forwardIn the drawing area below, draw the major products of this organic reaction: If there are no major products, because nothing much will happen to the reactant under these reaction conditions, check the box under the drawing area instead. 1. NaH 2. CH3Br ? Click and drag to start drawing a structure. No reaction. : ☐ Narrow_forward
- + Predict the major product of the following reaction. : ☐ + ☑ ค OH H₂SO4 Click and drag to start drawing a structure.arrow_forwardConsider this organic reaction: ... OH CI Draw the major products of the reaction in the drawing area below. If there won't be any major products, because this reaction won't happen at a significant rate, check the box under the drawing area instead. ☐ No Reaction. Click and drag to start drawing a structure. : аarrow_forwardConsider the following reactants: Br Would elimination take place at a significant rate between these reactants? Note for advanced students: by significant, we mean that the rate of elimination would be greater than the rate of competing substitution reactions. yes O no If you said elimination would take place, draw the major products in the upper drawing area. If you said elimination would take place, also draw the complete mechanism for one of the major products in the lower drawing area. If there is more than one major product, you may draw the mechanism that leads to any of them. Major Products:arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





