
Introduction to General, Organic and Biochemistry
12th Edition
ISBN: 9780357119303
Author: Bettelheim, Frederick A., Brown, William H., Campbell, Mary K., FARRELL, Shawn O., Torres, Omar
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 42P
15-46 Consider Lunesta, a nonbenzodiazepine hypnotic agent (i.e., sleep-inducing drug) that is frequently advertised on TV commercials. Answer the following questions with respect to the given structure:
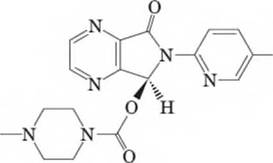
Lunesta
- Determine the molecular formula for Lunesta.
stereocenter(s) and therefore
possible stereoisomer(s). Of the possible
stereocenter(s),
is/are R and
is/are S.
- Does Lunesta have an enantiomer? Does it have a diastereomer?
- Which of the following is true about an enantiomer of Lunesta? Identify all that apply:
Draw an enantiomer of Lunesta.
Examine the derivative of the representation of the six-membered ring found in Lunesta. Draw the alternative chair conformations of this ring and label the more stable chair conformation. (Chapter 11)
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
SH
SH
0
2.
Please consider the two all 'cis' isomers of trimethylcyclohexane drawn below. Draw the
two chair conformers of each stereoisomer below (1 and 2) and calculate their torsional interaction
energies in order to identify the lower energy conformer for each stereoisomer. Based on your
calculations, state which of the two stereoisomers 1 and 2 is less stable and which is more stable.
[1,3-diaxial CH3 CH3 = 3.7kcal/mol; 1,3-diaxial CH3 H = 0.88kcal/mol; cis-1,2 (axial:equatorial) CH3
CH3 = 0.88kcal/mol; trans-1,2-diequatorial CH3 CH3 = 0.88kcal/mol)
all-cis-1,2,3-
1
all-cis-1,2,4-
2
Chapter 14 Solutions
Introduction to General, Organic and Biochemistry
Ch. 14.1 - Prob. 14.1QCCh. 14.2 - Problem 15-2 Assign priorities to the groups in...Ch. 14.2 - Problem 15-3 Assign an R or S configuration to the...Ch. 14.3 - Problem 15-4 3-Amino-2-butanol has two...Ch. 14.3 - Prob. 14.5QCCh. 14.3 - Prob. 14.6QCCh. 14 - 15-7 Answer true or false. The cis and trans...Ch. 14 - 15-8 What does the term “chiral” mean? Give an...Ch. 14 - 15-9 What does the term “achiral” mean? Give an...Ch. 14 - 15-10 Define the term “stereoisomer.” Name three...
Ch. 14 - 15-11 In what way are constitutional isomers...Ch. 14 - 15-12 Which of the following objects are chiral...Ch. 14 - Prob. 7PCh. 14 - Prob. 8PCh. 14 - Prob. 9PCh. 14 - Prob. 10PCh. 14 - 15-15 Explain why the carbon of a carbonyl group...Ch. 14 - 15-16 Which of the following compounds contain...Ch. 14 - 15-17 Which of the following compounds contain...Ch. 14 - Prob. 14PCh. 14 - 15-19 Draw the mirror image for each molecule: OH...Ch. 14 - Prob. 16PCh. 14 - 15-21 Answer true or false. For a molecule with...Ch. 14 - Prob. 18PCh. 14 - Prob. 19PCh. 14 - Prob. 20PCh. 14 - Prob. 21PCh. 14 - 15-26 For centuries, Chinese herbal medicine has...Ch. 14 - Prob. 23PCh. 14 - Prob. 24PCh. 14 - Prob. 25PCh. 14 - Prob. 26PCh. 14 - Prob. 27PCh. 14 - Prob. 28PCh. 14 - Prob. 29PCh. 14 - Prob. 30PCh. 14 - 15-35 Following are structural formulas for three...Ch. 14 - Prob. 32PCh. 14 - 15-37 Consider a cyclohexane ring substituted with...Ch. 14 - Prob. 34PCh. 14 - Prob. 35PCh. 14 - Prob. 36PCh. 14 - 15-41 Compound A(C5Hh, is not optically active and...Ch. 14 - Prob. 38PCh. 14 - 15-43 Triamcinolone acetonide, the active...Ch. 14 - 15-44 Consider the structure of the...Ch. 14 - Prob. 41PCh. 14 - 15-46 Consider Lunesta, a nonbenzodiazepine...Ch. 14 - Prob. 43PCh. 14 - Prob. 44P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonearrow_forwardWhat is the mechanism by which the 1,4 product is created? Please draw it by hand with arrows and stuff.arrow_forwardWhat is the relationship between A and B? H3C A Br Cl H3C B Br relationship (check all that apply) O same molecule O enantiomer O diastereomer structural isomer O stereoisomer isomer O need more information to decide O same molecule ☐ enantiomer Br Br Br CH3 Br CI CH3 O diastereomer ☐ structural isomer ☐ stereoisomer isomer O need more information to decide O same molecule O enantiomer Odiastereomer structural isomer O stereoisomer ☐ isomer O need more information to decidearrow_forward
- b. Please complete the zig-zag conformation of the compound (3R,4S)-3,4-dichloro-2,5-dimethylhexane by writing the respective atoms in the boxes. 4arrow_forwardc. Serricornin, the female-produced sex pheromone of the cigarette beetle, has the following structure. OH What is the maximum number of possible stereoisomers? Is this structure a meso compound? d. Please consider the natural product alkaloids shown below. Are these two structures enantiomers, diastereomers or conformers? H HO H H HN HO HN R R с R=H cinchonidine R=ET cinchonine Harrow_forwardNail polish remover containing acetone was spilled in a room 5.23 m × 3.28 m × 2.76 m. Measurements indicated that 2,250 mg of acetone evaporated. Calculate the acetone concentration in micrograms per cubic meter.arrow_forward
- Please help me answer number 1. 1. If your graphs revealed a mathematical relationship between specific heat and atomic mass, write down an equation for the relationship. I also don't understand, is the equation from the line regression the one that I'm suppose use to show the relationship? If so could you work it all the way out?arrow_forwardDescribe the principle of resonance and give a set of Lewis Structures to illustrate your explanation.arrow_forwardDon't used hand raitingarrow_forward
- It is not unexpected that the methoxyl substituent on a cyclohexane ring prefers to adopt the equatorial conformation. OMe H A G₂ = +0.6 kcal/mol OMe What is unexpected is that the closely related 2-methoxytetrahydropyran prefers the axial conformation: H H OMe OMe A Gp=-0.6 kcal/mol Methoxy: CH3O group Please be specific and clearly write the reason why this is observed. This effect that provides stabilization of the axial OCH 3 group in this molecule is called the anomeric effect. [Recall in the way of example, the staggered conformer of ethane is more stable than eclipsed owing to bonding MO interacting with anti-bonding MO...]arrow_forward206 Pb 82 Express your answers as integers. Enter your answers separated by a comma. ▸ View Available Hint(s) VAΣ ΜΕ ΑΣΦ Np, N₁ = 82,126 Submit Previous Answers ? protons, neutronsarrow_forwardPlease draw the inverted chair forms of the products for the two equilibrium reactions shown below. Circle the equilibrium reaction that would have a AG = 0, i.e., the relative energy of the reactant (to the left of the equilibrium arrows) equals the relative energy of the product? [No requirement to show or do calculations.] CH3 CH3 HH CH3 1 -CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY