
Identify each of the following compounds as an
a.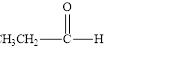 d.
d.
b.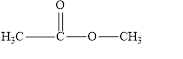 e.
e.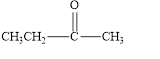
c.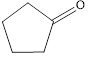 f.
f.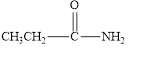
(a)
Interpretation:
The given compound is to be identified as an aldehyde, a ketone, or neither.
Concept introduction:
An aldehyde consists of a carbonyl group that is single bonded to minimum one hydrogen atom, whereas a ketone consists of a carbonyl group that is single bonded to two carbon atoms. The structural formula of an aldehyde is
Answer to Problem 14.4E
The given compound is an aldehyde.
Explanation of Solution
The given compound is shown below.
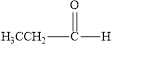
Figure 1
The given compound contains carbonyl group that is connected to one carbon atom and one hydrogen atom. Hence, it is an aldehyde.
The given compound is an aldehyde.
(b)
Interpretation:
The given compound is to be identified as an aldehyde, a ketone, or neither.
Concept introduction:
An aldehyde consists of a carbonyl group that is single bonded to minimum one hydrogen atom, whereas a ketone consists of a carbonyl group that is single bonded to two carbon atoms. The structural formula of an aldehyde is
Answer to Problem 14.4E
The given compound is neither aldehyde nor ketone.
Explanation of Solution
The given compound is shown below.
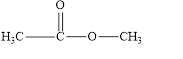
Figure 2
In the given compound, the carbonyl group is not attached with two carbon atoms. Therefore, it is not a ketone. Also, the carbonyl group is not attached with at least one hydrogen atom. Thus, it is not an aldehyde. Hence, it is neither aldehyde nor ketone.
The given compound is neither aldehyde nor ketone.
(c)
Interpretation:
The given compound is to be identified as an aldehyde, a ketone, or neither.
Concept introduction:
An aldehyde consists of a carbonyl group that is single bonded to minimum one hydrogen atom, whereas a ketone consists of a carbonyl group that is single bonded to two carbon atoms. The structural formula of an aldehyde is
Answer to Problem 14.4E
The given compound is a ketone.
Explanation of Solution
The given compound is shown below.
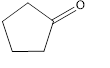
Figure 3
The given compound contains carbonyl group that is connected to two carbon atoms in the ring. Hence, it is a ketone.
The given compound is a ketone.
(d)
Interpretation:
The given compound is to be identified as an aldehyde, a ketone, or neither.
Concept introduction:
An aldehyde consists of a carbonyl group that is single bonded to minimum one hydrogen atom, whereas a ketone consists of a carbonyl group that is single bonded to two carbon atoms. The structural formula of an aldehyde is
Answer to Problem 14.4E
The given compound is neither aldehyde nor ketone.
Explanation of Solution
The given compound is shown below.
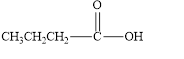
Figure 4
In the given compound, the carbonyl group is not attached with two carbon atoms. Therefore, it is not a ketone. Also, the carbonyl group is not attached with at least one hydrogen atom. Thus, it is not an aldehyde. Hence, it is neither aldehyde nor ketone.
The given compound is neither aldehyde nor ketone.
(e)
Interpretation:
The given compound is to be identified as an aldehyde, a ketone, or neither.
Concept introduction:
An aldehyde consists of a carbonyl group that is single bonded to at least one hydrogen atom, whereas a ketone consists of a carbonyl group that is single bonded to two carbon atoms. The structural formula of an aldehyde is
Answer to Problem 14.4E
The given compound is a ketone.
Explanation of Solution
The given compound is shown below.
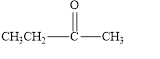
Figure 5
The given compound contains carbonyl group that is connected to two carbon atoms. Hence, it is a ketone.
The given compound is a ketone.
(f)
Interpretation:
The given compound is to be identified as an aldehyde, a ketone, or neither.
Concept introduction:
An aldehyde consists of a carbonyl group that is single bonded to minimum one hydrogen atom, whereas a ketone consists of a carbonyl group that is single bonded to two carbon atoms. The structural formula of an aldehyde is
Answer to Problem 14.4E
The given compound is neither aldehyde nor ketone.
Explanation of Solution
The given compound is shown below.
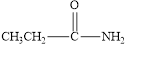
Figure 6
In the given compound, the carbonyl group is not attached with two carbon atoms. Therefore, it is not a ketone. Also, the carbonyl group is not attached with at least one hydrogen atom. Thus, it is not an aldehyde. The carbonyl group is attached to one nitrogen atom and one carbon atom. Hence, it is neither aldehyde nor ketone.
The given compound is neither aldehyde nor ketone.
Want to see more full solutions like this?
Chapter 14 Solutions
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
- Please correct answer and don't used hand raitingarrow_forwardCalculate the packing factor of CaTiO3. It has a perovskite structure. Data: ionic radii Co²+ = 0.106 nm, Ti4+ = 0.064 nm, O² = 0.132 nm; lattice constant is a = 2(rTi4+ + ro2-). Ca2+ 02- T14+ Consider the ions as rigid spheres. 1. 0.581 or 58.1% 2. -0.581 or -58.1 % 3. 0.254 or 25.4%arrow_forwardGeneral formula etherarrow_forward
- Please provide the retrosynthetic analysis and forward synthesis of the molecule on the left from the starting material on the right. Please include hand-drawn structures! will upvote! Please correct answer and don't used hand raitingarrow_forwardPlease provide the retrosynthetic analysis and forward synthesis of the molecule on the left from the starting material on the right. Please include hand-drawn structures! will upvote!arrow_forward(please correct answer and don't used hand raiting) Please provide the retrosynthetic analysis and forward synthesis of the molecule on the left from the starting material on the right. Please include hand-drawn structures! will upvote!arrow_forward
- CaTiO3 has a perovskite structure. Calculate the packing factor.Data: ionic radii Co+2 = 0.106 nm, Ti+4 = 0.064 nm, O-2 = 0.132 nm; lattice constant is a = 2(rTi4+ + rO-2).(a) 0.581(b) -0.581(c) 0.254(d) -0.254arrow_forwardIn the initial linear section of the stress-strain curve of a metal or alloy. Explain from the point of view of atomic structure?(a) No, the atomic level properties of the material can never be related to the linear section.(b) The elastic zone is influenced by the strength of the bonds between atoms.(c) The stronger the bond, the less rigid and the lower the Young's Modulus of the material tested.(d) The stronger the bond, the less stress is necessary to apply to the material to deform it elastically.arrow_forwardThe degree of polymerization of polytetrafluoroethylene (Teflon) is 7500 (mers/mol). If all polymer chains have equal length, state the molecular weight of the polymer and the total number of chains in 1000 g of the polymer(a) 50 000 g/mol; 0.03·1020 chains(b) 100 000 g/mol; 1.03·1020 chains(c) 750 000 g/mol; 8.03·1020 chainsarrow_forward
- In natural rubber or polyisoprene, the trans isomer leads to a higher degree of crystallinity and density than the cis isomer of the same polymer, because(a) it is more symmetrical and regular.(b) it is less symmetrical.(c) it is irregular.arrow_forwardMost ceramic materials have low thermal conductivities because:(a) Electron mobility is strongly restricted due to their strong ionic-covalent bonding.(b) False, in general they are excellent thermal conductors (they are used in ovens).(c) Electron mobility is dependent on T and therefore they are poor conductors at high temperatures.(d) Electron mobility is very restricted by secondary bonds.arrow_forwardResistivity and electrical conductivity.(a) In metals, resistivity decreases.(b) In metals, resistivity decreases and conductivity in semiconductors also decreases with increasing temperature.(c) With increasing temperature, resistivity in metals and conductivity in semiconductors also increases.(d) None of the above.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning




