
Concept explainers
(a)
Interpretation: The number of peaks present in the given NMR signal of labeled proton is to be calculated.
Concept introduction: In NMR spectrum, peaks are known as resonances, lines or absorptions. The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The labeled proton splits into septet in NMR spectrum of the given compound.
Explanation of Solution
The given compound is
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton gives seven peaks in NMR spectrum.
The labeled proton splits into septet in NMR spectrum of the given compound.
(b)
Interpretation: The number of peaks present in the given NMR signal of labeled proton is to be calculated.
Concept introduction: In NMR spectrum, peaks are known as resonances, lines or absorptions. The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The labeled proton (a), (b), and (c) splits into triplet, multiplet and quintet in NMR spectrum of the given compound.
Explanation of Solution
The given compound is
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton (a) gives three peaks (triplet) in NMR spectrum.
Proton (b) is bonded to one methylene group that has two hydrogen atoms and one methyl group that has three hydrogen atoms. The number of peaks is calculated by the formula,
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton (b) splits into a multiplet in NMR spectrum.
Proton (c) is bonded to two methylene groups that have four hydrogen atoms. The number of peaks is calculated by the formula,
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton (c) gives quintet in NMR spectrum.
Therefore, protons (a), (b), and (c) give
The labeled proton (a), (b), and (c) splits into triplet, multiplet and quintet in NMR spectrum of the given compound.
(c)
Interpretation: The number of peaks present in the given NMR signal of labeled proton is to be calculated.
Concept introduction: In NMR spectrum, peaks are known as resonances, lines or absorptions. The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The labeled proton (a) and (b) splits into doublet and sextet in NMR spectrum of the given compound.
Explanation of Solution
The given compound is shown below.
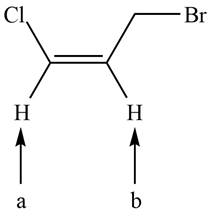
Figure 1
There are two labeled protons, (a) and (b). Proton (b) is bonded to a carbon atom that has one hydrogen and one methylene group. The number of peaks is calculated by the formula,
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton (b) splits into a sextet in NMR spectrum.
Proton (a) is bonded to a carbon atom that has only one hydrogen atom. The number of peaks is calculated by the formula,
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton (a) gives two peaks (doublet) in NMR spectrum.
Therefore, protons (a) and (b) give
The labeled proton (a) and (b) splits into doublet and sextet in NMR spectrum of the given compound.
(d)
Interpretation: The number of peaks present in the given NMR signal of labeled proton is to be calculated.
Concept introduction: In NMR spectrum, peaks are known as resonances, lines or absorptions. The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The labeled proton (a) and (b) splits into triplet, doublet and doublet in NMR spectrum of the given compound.
Explanation of Solution
The given compound is shown below.
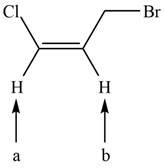
Figure 2
There are two labeled protons, (a) and (b). Proton (a) is bonded to a carbon atom that has two hydrogen atoms. The number of peaks is calculated by the formula,
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled proton (a) gives three peaks (triplet) in NMR spectrum.
Both protons (b) are bonded to a carbon atom that has only one hydrogen atom. The number of peaks is calculated by the formula,
Where,
➢
➢
Here,
Substitute the value of
Hence, the labeled protons (b) give two peaks (doublet) in NMR spectrum.
Therefore, protons (a) and (b) give
The labeled proton (a) and (b) splits into triplet, doublet and doublet in NMR spectrum of the given compound.
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry
- Part I. Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff: Pinacol (2,3-dimethyl, 1-3-butanediol) on treatment w/ acid gives a mixture of pina colone and (3,3-dimethyl-2-butanone) 2,3-dimethyl-1,3-butadiene. Give reasonable mechanism the formation of the products Forarrow_forwardShow the mechanism for these reactionsarrow_forwardDraw the stepwise mechanismarrow_forward
- Draw a structural formula of the principal product formed when benzonitrile is treated with each reagent. (a) H₂O (one equivalent), H₂SO₄, heat (b) H₂O (excess), H₂SO₄, heat (c) NaOH, H₂O, heat (d) LiAlH4, then H₂Oarrow_forwardDraw the stepwise mechanism for the reactionsarrow_forwardDraw stepwise mechanismarrow_forward
- Part I. Draw reaction mechanism for the transformations of benzophenone to benzopinacol to benzopinaco lone and answer the ff: a) Give the major reason for the exposure of benzophenone al isopropyl alcohol (w/acid) to direct sunlight of pina colone Mechanism For b) Pinacol (2,3-dimethy 1, 1-3-butanediol) on treatment w/ acid gives a mixture (3,3-dimethyl-2-butanone) and 2, 3-dimethyl-1,3-butadiene. Give reasonable the formation of the productsarrow_forwardwhat are the Iupac names for each structurearrow_forwardWhat are the IUPAC Names of all the compounds in the picture?arrow_forward
- 1) a) Give the dominant Intermolecular Force (IMF) in a sample of each of the following compounds. Please show your work. (8) SF2, CH,OH, C₂H₂ b) Based on your answers given above, list the compounds in order of their Boiling Point from low to high. (8)arrow_forward19.78 Write the products of the following sequences of reactions. Refer to your reaction road- maps to see how the combined reactions allow you to "navigate" between the different functional groups. Note that you will need your old Chapters 6-11 and Chapters 15-18 roadmaps along with your new Chapter 19 roadmap for these. (a) 1. BHS 2. H₂O₂ 3. H₂CrO4 4. SOCI₂ (b) 1. Cl₂/hv 2. KOLBU 3. H₂O, catalytic H₂SO4 4. H₂CrO4 Reaction Roadmap An alkene 5. EtOH 6.0.5 Equiv. NaOEt/EtOH 7. Mild H₂O An alkane 1.0 2. (CH3)₂S 3. H₂CrO (d) (c) 4. Excess EtOH, catalytic H₂SO OH 4. Mild H₂O* 5.0.5 Equiv. NaOEt/EtOH An alkene 6. Mild H₂O* A carboxylic acid 7. Mild H₂O* 1. SOC₁₂ 2. EtOH 3.0.5 Equiv. NaOEt/E:OH 5.1.0 Equiv. NaOEt 6. NH₂ (e) 1. 0.5 Equiv. NaOEt/EtOH 2. Mild H₂O* Br (f) i H An aldehyde 1. Catalytic NaOE/EtOH 2. H₂O*, heat 3. (CH,CH₂)₂Culi 4. Mild H₂O* 5.1.0 Equiv. LDA Br An ester 4. NaOH, H₂O 5. Mild H₂O* 6. Heat 7. MgBr 8. Mild H₂O* 7. Mild H₂O+arrow_forwardLi+ is a hard acid. With this in mind, which if the following compounds should be most soluble in water? Group of answer choices LiBr LiI LiF LiClarrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

