
Organic Chemistry, Books a la Carte Edition (9th Edition)
9th Edition
ISBN: 9780134160382
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.13, Problem 13.29P
A bottle of allyl bromide was found to contain a large amount of an impurity. A careful distillation separated the impurity, which has the molecular formula C3H6O. The folowing 13C NMR spectrum of the impurity was obtained:
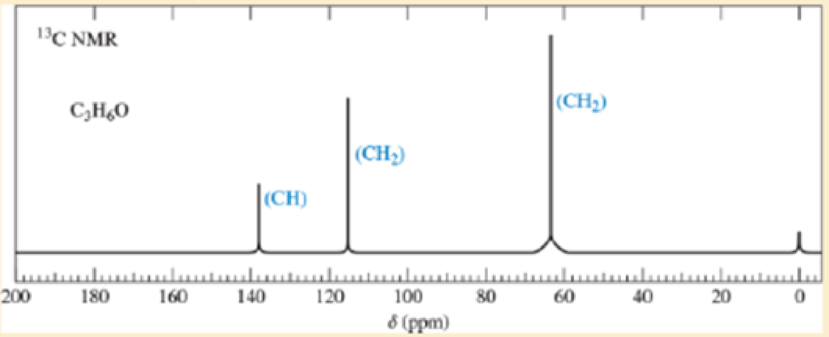
- a. Propose a structure for this impurity.
- b. Assign the peaks in the 13C NMR spectrum to the carbon atoms in the structure.
- c. Suggest how this impunty arose in the allyl bromide sample.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can you please explain this problem to me? I'm very confused about it. Please provide a detailed, step-by-step explanation for me! (ME EX1) Prblm 22
Can you please explain this problems to me? I'm very confused about it. Please provide a detailed, step-by-step explanation for me! (ME EX1) Prblm 30
This organic molecule is dissolved in a basic aqueous solution:
O
?
olo
RET
A short time later sensitive infrared spectroscopy reveals the presence of a new C-OH stretch absorption. That is, there Ar
must now be a new molecule present with at least one C - OH bond.
In the drawing area below, show the detailed mechanism that could convert the molecule above into the new molecule.
$
Add/Remove step
Chapter 13 Solutions
Organic Chemistry, Books a la Carte Edition (9th Edition)
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- So the thing is im trying to memorize VESPR Shapes in order to be able to solve problems like so, and I need help with making circles like the second image that's in blue or using an x and y axis plane in order to memorize these and be able to solve those type of problems. Especially like the ones given in the top / first image. (180 , 120 , 109.5) Can you help me with this.arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward2. (15 points) Draw an appropriate mechanism for the following reaction. H N. H* + H₂Oarrow_forward
- Draw a tripeptide of your choosing at pH 7. Have the N-terminus on the left and the C-terminus on the right. Then: Draw a triangle around the α-carbons. Draw a box around the R-groups. Circle the atoms capable of hydrogen bonding. Highlight the atoms involved in the formation of the peptide bonds. What type of structure have you drawn? (primary, secondary, tertiary or quaternary protein structure). make sure its a tripeptidearrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- Don't used Ai solution and don't used hand raitingarrow_forward> Organic Functional Groups Naming and drawing alkyl halides structure CI Br CI CI Explanation Check 2 name 1-chloro-2,4,9-trimethylnonane CI 2-iodo-2,3-dimethylbutane FEB 19 € E M tv MacBook Airarrow_forwardCan you please explain to me this problem im very confused and lost. Help me step by step and in detail im soo lost.arrow_forward
- 2) There are many forms of cancer, all of which involve abnormal cell growth. The growth and production of cells, called cell proliferation, is known to involve an enzyme called protein farnesyltransferase (PFTase). It is thought that inhibitors pf PFTase may be useful as anticancer drugs. The following molecule showed moderate activity as a potential PFTase inhibitor. Draw all stereoisomers of this compound. HO OHarrow_forwardConsidering rotation around the bond highlighted in red, draw the Newman projection for the most stable and least stable conformations when viewed down the red bond in the direction of the arrow. Part 1 of 2 H₁₂C H H Draw the Newman projection for the most stable conformation. Select a template to begin. Part 2 of 2 Draw the Newman projection for the least stable conformation. G 心arrow_forwardpersonality of each of them in terms of nucleophile vs. electrophile (some can be considered acids/bases but we are not looking at that here). Note you may have to use your growing intuition to figure out the personality of one of the molecules below but I believe in you! Rationalize it out based on what we have called strong versus weak electrophiles in past mechanisms. Consider using the memes below to help guide your understanding! A OH O B CH3 C Molecule A: [Select] Molecule B: [Select] Molecule C: [Select] Molecule D: [Select] > H D OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY