
Concept explainers
(a)
Interpretation:
The compound that is more soluble in water should be determined.
CH3 OH or CH3 OCH3.
Concept Introduction:
Water solubility is known as the measurement of the chemical substance which can dissolve into the water at the specific temperature. The solubility’s unit is mg/L or ppm i.e. parts per million. Water solubility is the most vital properties which affects the environmental fate and bioavailability of the chemical substances.
(b)
Interpretation:
The compound that is more soluble in water should be determined.
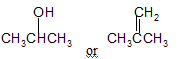
Concept Introduction:
Water solubility is known as the measurement of the chemical substance which can dissolve into the water at the specific temperature. The solubility’s unit is mg/L or ppm i.e. parts per million. Water solubility is the most vital properties which affects the environmental fate and bioavailability of the chemical substances.
(c)
Interpretation:
The compound that is more soluble in water should be determined.
CH3 CH2 CH2 SH or CH3 CH2 CH2 OH.
Concept Introduction:
Water solubility is known as the measurement of the chemical substance which can dissolve into the water at the specific temperature. The solubility’s unit is mg/L or ppm i.e. parts per million. Water solubility is the most vital properties which affects the environmental fate and bioavailability of the chemical substances.
(d)
Interpretation:
The compound that is more soluble in water should be determined.
CH3 CH2 Cl or NaCl.
Concept Introduction:
Water solubility is known as the measurement of the chemical substance which can dissolve into the water at the specific temperature. The solubility’s unit is mg/L or ppm i.e. parts per million. Water solubility is the most vital properties which affects the environmental fate and bioavailability of the chemical substances.
(e)
Interpretation:
The compound that is more soluble in water should be determined.
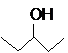 or
or 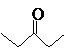
Concept Introduction:
Water solubility is known as the measurement of the chemical substance which can dissolve into the water at the specific temperature. The solubility’s unit is mg/L or ppm i.e. parts per million. Water solubility is the most vital properties which affects the environmental fate and bioavailability of the chemical substances.
Trending nowThis is a popular solution!

Chapter 13 Solutions
Introduction to General, Organic and Biochemistry
- 17-26 Account for the fact that acetone has a higher boiling point (56°C) than ethyl methyl ether (11°C) even though their molecular weights are almost the same.arrow_forward17-69 Propanal (bp 49°C) and 1-propanol (bp 97°C) have about the same molecular weight, yet their boiling points differ by almost 50°C. Explain this fact.arrow_forward16-17 Propylamine (bp 48°C), ethylmethylamine (bp 37°C), and trimethylamine (bp 3°C) are constitutional isomers with the molecular formula C3HgN. Account for the fact that trimethylamine has the lowest boiling point of the three and propylamine has the highest.arrow_forward
- Kindly answer this questionarrow_forward12 Write the IUPAC names for these compounds. O (a) CHO CHOH (c) | CHOH I CH₂OH OH (b) (d) CHO NH₂ Oarrow_forward15-18 Give IUPAC names for the following compounds: (a) (c) Br CH3 (b) CO₂H طة Br CH3 -CHCH2CH2CHCH3 (d) Br CH2CH2CH3 (e) F NO2 CH3 (f) NH2 NO2arrow_forward
- 19. Which of the following compounds has the LOWEST solubility in water at 25 °C? a) CH;CH;CH;OH b) CH-CH:CH:CH.ОН c) CH3CH2CH2CH;CH2OH d) same solubility for allarrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forwardIdentify the 5-7 uses of benzoic acidarrow_forward
- 17-42 What Grignard reagent and what carbonyl compound might you start with to prepare the following alcohols? (a) OH (b) CH3CHCH₂CH3 (d) HO (e) OH ī CH3CH₂CHCH2CH3 HO CH3 CH3 (c) (f) CH3 H₂C= CH₂OH CH₂OHarrow_forwardPQ-17. What is the major product of this reaction? (A) H3CO њсоян (B) OH OH (C) OH O H OH OH 1) CH,MgBr (1 eq.) 2) NHÀ -OH (D) онянarrow_forwardI really need help with thisarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


