
Chemistry (7th Edition)
7th Edition
ISBN: 9780321943170
Author: John E. McMurry, Robert C. Fay, Jill Kirsten Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 13, Problem 13.8A
Interpretation Introduction
Interpretation:
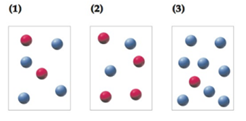
Determine the initial
Concept Introduction:
Relationship of rate of the reaction to the rate constant and concentration of the reactantsraised to some powers which are experimentally determined are called as rate law.
Given:
Initial rate of the reaction in the vessel 1 is 0.01 M/s, Rate law for the Reaction
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write the complete common (not IUPAC) name of each molecule below.
Note: if a molecule is one of a pair of enantiomers, be sure you start its name with D- or L- so we know which enantiomer it is.
molecule
C=O
H3N
CH3
common name
(not the IUPAC
name)
H
☐
C=O
H
O-C-CH2-CH2
010
NH3
☐
H3N
☐
HO
5
Write the systematic name of each organic molecule:
structure
CI
CH3
HO-C-CH-CH-CH2 – CH— CH3
CH3
name
X
O
☐
CH3-CH-CH2-CH2-C-OH
CH3
11
HO-C-CH-CH2-OH
CH3
☐
Check the box under each a amino acid.
If there are no a amino acids at all, check the "none of them" box under the table.
Note for advanced students: don't assume every amino acid shown must be found in nature.
CH3
CH2
0 C=O
+
CH-CH3
H₂N C-COOH
H₂N
H
H
H3N C COO¯
NH, O
HO C C H
CH3-CH
HO C=O
H2N-CH-COOH
CH2
NH3
HO
CH3
none of them
O
NH3
Chapter 13 Solutions
Chemistry (7th Edition)
Ch. 13 - Prob. 13.1PCh. 13 - Prob. 13.2ACh. 13 - Prob. 13.3PCh. 13 - Prob. 13.4ACh. 13 - Prob. 13.5PCh. 13 - Prob. 13.6ACh. 13 - Prob. 13.7PCh. 13 - Prob. 13.8ACh. 13 - Prob. 13.9PCh. 13 - Prob. 13.10A
Ch. 13 - Prob. 13.11PCh. 13 - Prob. 13.12ACh. 13 - Prob. 13.13PCh. 13 - Prob. 13.14ACh. 13 - Prob. 13.15PCh. 13 - Prob. 13.16ACh. 13 - Prob. 13.17PCh. 13 - Prob. 13.18ACh. 13 - Prob. 13.19PCh. 13 - Prob. 13.20ACh. 13 - Prob. 13.21PCh. 13 - Apply 13.22 The rate of the reaction...Ch. 13 - Prob. 13.23PCh. 13 - Prob. 13.24ACh. 13 - Prob. 13.25PCh. 13 - Prob. 13.26ACh. 13 - Prob. 13.27PCh. 13 - Prob. 13.28ACh. 13 - Prob. 13.29PCh. 13 - Prob. 13.30ACh. 13 - Prob. 13.31PCh. 13 - Prob. 13.32ACh. 13 - Prob. 13.33PCh. 13 - Prob. 13.34PCh. 13 - Prob. 13.35PCh. 13 - Prob. 13.36PCh. 13 - Prob. 13.37PCh. 13 - Prob. 13.38PCh. 13 - Prob. 13.39CPCh. 13 - Prob. 13.40CPCh. 13 - Prob. 13.41CPCh. 13 - Prob. 13.42CPCh. 13 - Prob. 13.43CPCh. 13 - Prob. 13.44CPCh. 13 - Prob. 13.45CPCh. 13 - Prob. 13.46CPCh. 13 - Prob. 13.47CPCh. 13 - Prob. 13.48CPCh. 13 - Prob. 13.49CPCh. 13 - Use the data in Table 13.1 to calculate the...Ch. 13 - 13.50 Use the data in Table 13.1 to calculate the...Ch. 13 - Prob. 13.52SPCh. 13 - Prob. 13.53SPCh. 13 - From the plot of concentrationtime data in Figure...Ch. 13 - Prob. 13.55SPCh. 13 - Prob. 13.56SPCh. 13 - Prob. 13.57SPCh. 13 - Prob. 13.58SPCh. 13 - Prob. 13.59SPCh. 13 - Prob. 13.60SPCh. 13 - Prob. 13.61SPCh. 13 - Prob. 13.62SPCh. 13 - Prob. 13.63SPCh. 13 - Prob. 13.64SPCh. 13 - Prob. 13.65SPCh. 13 - Prob. 13.66SPCh. 13 - Prob. 13.67SPCh. 13 - Prob. 13.68SPCh. 13 - Prob. 13.69SPCh. 13 - Prob. 13.70SPCh. 13 - Prob. 13.71SPCh. 13 - Prob. 13.72SPCh. 13 - Prob. 13.73SPCh. 13 - Prob. 13.74SPCh. 13 - Prob. 13.75SPCh. 13 - Prob. 13.76SPCh. 13 - Prob. 13.77SPCh. 13 - Prob. 13.78SPCh. 13 - Prob. 13.79SPCh. 13 - Prob. 13.80SPCh. 13 - Prob. 13.81SPCh. 13 - Prob. 13.82SPCh. 13 - Prob. 13.83SPCh. 13 - Prob. 13.84SPCh. 13 - Prob. 13.85SPCh. 13 - Prob. 13.86SPCh. 13 - Prob. 13.87SPCh. 13 - Prob. 13.88SPCh. 13 - Prob. 13.89SPCh. 13 - Prob. 13.90SPCh. 13 - Prob. 13.91SPCh. 13 - Prob. 13.92SPCh. 13 - Prob. 13.93SPCh. 13 - Prob. 13.94SPCh. 13 - Prob. 13.95SPCh. 13 - Prob. 13.96SPCh. 13 - Prob. 13.97SPCh. 13 - Prob. 13.98SPCh. 13 - Prob. 13.99SPCh. 13 - Prob. 13.100SPCh. 13 - Prob. 13.101SPCh. 13 - Prob. 13.102SPCh. 13 - Prob. 13.103SPCh. 13 - Prob. 13.104SPCh. 13 - Prob. 13.105SPCh. 13 - Prob. 13.106SPCh. 13 - Prob. 13.107SPCh. 13 - Prob. 13.108SPCh. 13 - Prob. 13.109SPCh. 13 - Prob. 13.110SPCh. 13 - Prob. 13.111SPCh. 13 - Prob. 13.112SPCh. 13 - Prob. 13.113SPCh. 13 - Prob. 13.114SPCh. 13 - Prob. 13.115CPCh. 13 - Prob. 13.116CPCh. 13 - Prob. 13.117CPCh. 13 - Prob. 13.118CPCh. 13 - Prob. 13.119CPCh. 13 - Prob. 13.120CPCh. 13 - Prob. 13.121CPCh. 13 - Prob. 13.122CPCh. 13 - Prob. 13.123CPCh. 13 - Prob. 13.124CPCh. 13 - Prob. 13.125CPCh. 13 - Prob. 13.126CPCh. 13 - Prob. 13.127CPCh. 13 - Prob. 13.128CPCh. 13 - Prob. 13.129CPCh. 13 - Prob. 13.130CPCh. 13 - Prob. 13.131CPCh. 13 - Prob. 13.132CPCh. 13 - Prob. 13.133CPCh. 13 - Prob. 13.134CPCh. 13 - Prob. 13.135CPCh. 13 - Prob. 13.136CPCh. 13 - Prob. 13.137CPCh. 13 - Prob. 13.138CPCh. 13 - Prob. 13.139CPCh. 13 - Prob. 13.140CPCh. 13 - Prob. 13.141CPCh. 13 - Prob. 13.142CPCh. 13 - Prob. 13.143CPCh. 13 - Prob. 13.144MPCh. 13 - Prob. 13.145MPCh. 13 - Prob. 13.146MPCh. 13 - Prob. 13.147MPCh. 13 - Prob. 13.148MPCh. 13 - Prob. 13.149MPCh. 13 - Prob. 13.150MP
Knowledge Booster
Similar questions
- handwritten answer please!arrow_forwardConsider the following SN 2 reaction: مار + Br H₂O acetone + Br OH What effect would each of the following changes have on the rate of this reaction. Select the single best answer for each part. Part 1 of 3 If the substrate was changed to: The rate would Br O increase O decrease O remain unchanged Part 2 of 3 × S If the nucleophile was changed to OH, the rate would: O increase O decrease O remain unchanged Part 3 of 3 If the solvent was changed to ethanol, the rate would: Increase O decrease O remain unchanged 2 ol Ararrow_forwardConsider the following nucleophilic substitution reaction. The compound listed above the arrow is the solvent for the reaction. If nothing is listed over the arrow, then the nucleophile is also the solvent for the reaction. Part: 0/2 Part 1 of 2 Br acetone + I What is the correct mechanism for the reaction? Select the single best answer. OSN 1 OSN 2 X Part: 1/2 Part 2 of 2 Draw the products for the reaction. Include both the major organic product and the inorganic product. If more than one stereoisomer is possible, draw only one stereoisomer. Include stereochemistry where relevant. Click and drag to start drawing a structure. Х 5 ☐arrow_forward
- Triethyloxonium tetrafluoroborate reacts with ethanol (CH3CH2OH) to give diethyl ether (CH3CH2OCH2CH3). BF triethyloxonium tetrafluoroborate Which equation, including the curved arrows, best represents the rate-determining step in the mechanism? Select the single best answer. O OH CH3CH2 OH + H. 0+ CH₂H₂ :0 + 0+ ж + H + :0: 0 Carrow_forwardCH3CH2CH=CH2 + H₂O − H+arrow_forwardГ C-RSA CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0.0 b.092 0.797 1.088 1.813 C-RSA CHROMATOPAC CH=1 Report No. =13 ** CALCULATION REPORT ** DATA=1: @CHRM1.000 11/03/05 08:09:52 CH PKNO TIME 1 2 0.797 3 1.088 4 1.813 AREA 1508566 4625442 2180060 HEIGHT 207739 701206 V 287554 V MK IDNO CONC NAME 18.1447 55.6339 26.2213 TOTAL 8314067 1196500 100 C-R8A CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0. 0 087 337. 0.841 1.150 C-R8A CHROMATOPAC CH=1 Report No. =14 DATA=1: @CHRM1.000 11/03/05 08:12:40 ** CALCULATION REPORT ** CH PKNO TIME AREA 1 3 0.841 1099933 41.15 4039778 HEIGHT MK IDNO 170372 649997¯¯¯ CONC NAME 21.4007 78.5993 TOTAL 5139711 820369 100 3 C-R8A CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0.100 0:652 5.856 3 1.165 C-RSA CHROMATOPAC CH-1 Report No. =15 DATA=1: @CHRM1.000 11/03/05 08:15:26 ** CALCULATION REPORT ** CH PKNO TIME AREA HEIGHT MK IDNO CONC NAME 1 3 3 0.856 4 1.165 TOTAL 1253386 4838738 175481 708024 V 20.5739 79.4261 6092124…arrow_forward
- break down both reactions shown and explain it correctly using the bromonium ion mechanism, instead of the (disproven) carbocation-based mechanism.arrow_forwardIndicate how from 1,2-diaminobenzene to obtain 1-metilbenzotriazol.arrow_forward-C = C - C - + Br₂ + I" -> -C-C-c -C = C -C- + Br² + I₂ -C=C Br I + Brū + Iz -7- C - C-C- I Br Mechanism; - C = c - c - + Br - Br > - C-c-c- Br -C-C-C- + 1 - - -Ċ-Ċ'-c' - Br Br Iarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY