
(a)
Interpretation:
An IUPAC name for the alcohol,
Concept introduction:
The systematic naming of organic compound is given by
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene, -ol, -al, -oic and so on.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 13.6E
The IUPAC name for the alcohol,
Explanation of Solution
In
The IUPAC name for the alcohol,
(b)
Interpretation:
An IUPAC name for the given alcohol is to be predicted.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene, -ol, -al, -oic and so on.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 13.6E
An IUPAC name for the given alcohol is propan-
Explanation of Solution
The alcohol for which the IUPAC name has to be predicted is shown below.

Figure 1
The longest carbon chain in this alcohol is made up of three carbon atoms. The three carbon atoms chain makes this alcohol a propanol. The hydroxyl group is attached to the second carbon atom. Therefore, the IUPAC name for this alcohol is propan-
An IUPAC name for the given alcohol is propan-
(c)
Interpretation:
An IUPAC name for the alcohol,
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene, -ol, -al, -oic and so on.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 13.6E
An IUPAC name for the alcohol,
Explanation of Solution
In
An IUPAC name for the alcohol,
(d)
Interpretation:
An IUPAC name for the given alcohol is to be predicted.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene, -ol, -al, -oic and so on.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 13.6E
An IUPAC name for the given alcohol is
Explanation of Solution
The alcohol for which the IUPAC name has to be predicted is shown below.
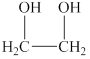
Figure 2
The longest carbon chain in this alcohol is made up of two carbon atoms. The hydroxyl groups are attached to the first and second carbon atom. Therefore, the IUPAC name for the given alcohol is
An IUPAC name for the given alcohol is
(e)
Interpretation:
An IUPAC name for the given alcohol is to be predicted.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene, -ol, -al, -oic and so on.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 13.6E
An IUPAC name for the given alcohol is propane-
Explanation of Solution
The alcohol for which the IUPAC name has to be predicted is shown below.
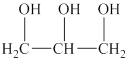
Figure 3
The longest carbon chain in this alcohol is made up of three carbon atoms. One hydroxyl group is attached to each carbon atom. Therefore, the alcohol will be propanetriol. The hydroxyl group is attached to the first, second and third carbon atom. Therefore, the IUPAC name for the given alcohol is propane-
An IUPAC name for the given alcohol is propane-
Want to see more full solutions like this?
Chapter 13 Solutions
Study Guide with Student Solutions Manual for Seager/Slabaugh/Hansen's Chemistry for Today: General, Organic, and Biochemistry, 9th Edition
- Indicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol.arrow_forward2,2-Dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol. Indicate the products obtained.arrow_forwardAdd conditions above and below the arrow that turn the reactant below into the product below in a single transformationADS fint anditions 百 Abl res condinese NC ง Add on condtions 1.0 B H,N.arrow_forward
- Steps on how to solve. Thank you!arrow_forward3. Name this ether correctly. H₁C H3C CH3 CH3 4. Show the best way to make the ether in #3 by a Williamson Ether Synthesis. Start from an alcohol or phenol. 5. Draw the structure of an example of a sulfide.arrow_forward1. Which one(s) of these can be oxidized with CrO3 ? (could be more than one) a) triphenylmethanol b) 2-pentanol c) Ethyl alcohol d) CH3 2. Write in all the product(s) of this reaction. Label them as "major" or "minor". 2-methyl-2-hexanol H2SO4, heatarrow_forward
- 3) Determine if the pairs are constitutional isomers, enantiomers, diastereomers, or mesocompounds. (4 points)arrow_forwardIn the decomposition reaction in solution B → C, only species C absorbs UV radiation, but neither B nor the solvent absorbs. If we call At the absorbance measured at any time, A0 the absorbance at the beginning of the reaction, and A∞ the absorbance at the end of the reaction, which of the expressions is valid? We assume that Beer's law is fulfilled.arrow_forward> You are trying to decide if there is a single reagent you can add that will make the following synthesis possible without any other major side products: 1. ☑ CI 2. H3O+ O Draw the missing reagent X you think will make this synthesis work in the drawing area below. If there is no reagent that will make your desired product in good yield or without complications, just check the box under the drawing area and leave it blank. Click and drag to start drawing a structure. Explanation Check ? DO 18 Ar B © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilityarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



