
What products would result from the following processes?
Write an equation for each reaction.
a.
b.
c.
d.
e.
(a)
Interpretation:
The product formed when
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed when
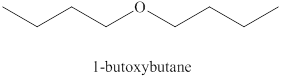
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Alcohols on heating at
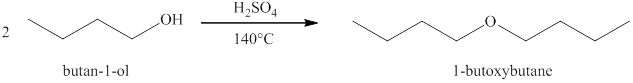
Figure 1
The product formed when
(b)
Interpretation:
The product formed by the excess oxidation of
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed by the excess oxidation of
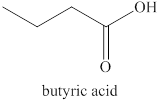
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
The primary alcohol,

Figure 2
The product formed by the excess oxidation of
(c)
Interpretation:
The product formed by the controlled oxidation of
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed when
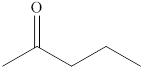
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
The compound
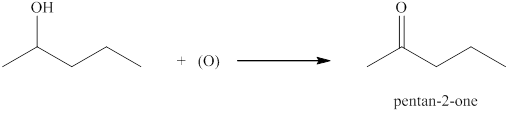
Figure 3
The product formed when
(d)
Interpretation:
The product formed when
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
The product formed when
![]()
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Alcohols on heating at temperature
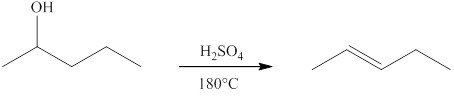
Figure 4
Interpretation:
The product formed when
(e)
Interpretation:
The product formed by the controlled oxidation of
Concept introduction:
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
Answer to Problem 13.29E
No product is formed by the controlled oxidation of
Explanation of Solution
Ketones can be prepared by the oxidation reaction of secondary alcohol. On the other hand, primary alcohols on oxidation with weak oxidant give aldehyde and with strong oxidant give carboxylic acid. No product is obtained by the oxidation of tertiary alcohol.
The controlled oxidation of
No product is formed by the controlled oxidation of
Want to see more full solutions like this?
Chapter 13 Solutions
Bundle: Chemistry For Today: General, Organic, And Biochemistry, 9th + Owlv2 With Mindtap Reader, 1 Term (6 Months) Printed Access Card
- Don't used Ai solutionarrow_forwardDraw a Lewis dot structure for C2H4Oarrow_forward3.3 Consider the variation of molar Gibbs energy with pressure. 3.3.1 Write the mathematical expression for the slope of graph of molar Gibbs energy against 3.3.2 pressure at constant temperature. Draw in same diagram graphs showing variation with pressure of molar Gibbs energies of a substance in gaseous, liquid and solid forms at constant temperature. 3.3.3 Indicate in your graphs melting and boiling points. 3.3.4 Indicate for the respective phases the regions of relative stability.arrow_forward
- In 2-chloropropane, the signal for the H on the C next to Cl should be split into how many peaks?arrow_forward4.4 Consider as perfect gas 3.0 mol of argon gas to which 229 J of energy is supplied as heat at constant pressure and temperature increases by 2.55 K. Calculate 4.4.1 constant pressure molar heat capacity. 4.4.2 constant volume molar heat capacity.arrow_forward3.2 32 Consider calibrating a calorimeter and measuring heat transferred. A sample of compound was burned in a calorimeter and a temperature change of 3.33°C recorded. When a 1.23 A current from a 12.0 V source was passed through a heater in the same calorimeter for 156 s, the temperature changed of 4.47°C was recorded. 3.2.1 Calculate the heat supplied by the heater. 3.2.2 Calculate the calorimeter constant. 3.2.3 Calculate the heat released by the combustion reaction.arrow_forward
- -.1 Consider the standard enthalpy of formation of gaseous water at 25°C as -241.82 kJ/mol and calculate the standard enthalpy of formation of gaseous water at 100°C.arrow_forward3.5 Complete the following sentences to make correct scientific meaning. 3.5.1 The entropy of a perfect gas. 3.5.2 when it expands isothermally. The change in entropy of a substance accompanying a change of state at its transition 3.5.3 temperature is calculated from its of transition. The increase in entropy when a substance is heated is calculated from itsarrow_forward3.4 Consider the internal energy of a substance 3.4.1 Draw a graph showing the variation of internal energy with temperature at constant volume 3.4.2 Write the mathematical expression for the slope in your graph in 3.4.1arrow_forward
- For a system, the excited state decays to the ground state with a half-life of 15 ns, emitting radiation of 6000 Å. Determine the Einstein coefficients for stimulated absorption and spontaneous emission and the dipole moment of the transition. Data: epsilon 0 = 8.85419x10-12 C2m-1J-1arrow_forwardProblem a. The following compounds have the same molecular formula as benzene. How many monobrominated products could each form? 1. HC =CC=CCH2CH3 2. CH2=CHC = CCH=CH₂ b. How many dibrominated products could each of the preceding compounds form? (Do not include stereoisomers.)arrow_forwardDon't used Ai solutionarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





