
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
5th Edition
ISBN: 9780321967466
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 12.7, Problem 12.39QAP
Interpretation Introduction
a)
To determine:
The structural formula of product for the given reaction using Markownikov’s rule if necessary
The product is Pentane.
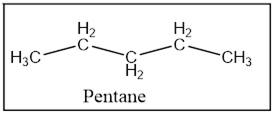
Interpretation Introduction
b)
To determine:
The structural formula of product for the given reaction using Markownikov’s rule if necessary
The product is 2-Methylbutan-2-ol.
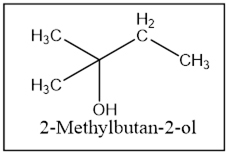
Interpretation Introduction
c)
To determine:
The structural formula of product for the given reaction using Markownikov’s rule if necessary
The product is Butane.
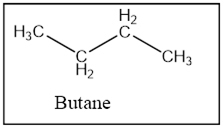
Interpretation Introduction
d)
To determine:
The structural formula of product for the given reaction using Markownikov’s rule if necessary
The product is 1-Methylcyclohexan-1-ol.
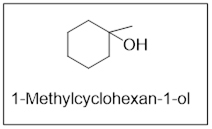
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Laser. Indicate the relationship between metastable state and stimulated emission.
The table includes macrostates characterized by 4 energy levels (&) that are
equally spaced but with different degrees of occupation.
a) Calculate the energy of all the macrostates (in joules). See if they all have
the same energy and number of particles.
b) Calculate the macrostate that is most likely to exist. For this macrostate,
show that the population of the levels is consistent with the Boltzmann
distribution.
macrostate 1 macrostate 2 macrostate 3
ε/k (K) Populations
Populations
Populations
300
5
3
4
200
7
9
8
100
15
17
16
0
33
31
32
DATO: k = 1,38×10-23 J K-1
Don't used Ai solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Ch. 12.1 - Identify each of the following as a formula of an...Ch. 12.1 - Prob. 12.2QAPCh. 12.1 - Prob. 12.3QAPCh. 12.1 - Prob. 12.4QAPCh. 12.1 - Prob. 12.5QAPCh. 12.1 - Prob. 12.6QAPCh. 12.1 - Prob. 12.7QAPCh. 12.1 - Prob. 12.8QAPCh. 12.2 - Give the IUPAC name for each of the following...Ch. 12.2 - Prob. 12.10QAP
Ch. 12.2 - Prob. 12.11QAPCh. 12.2 - Prob. 12.12QAPCh. 12.3 - Prob. 12.13QAPCh. 12.3 - Prob. 12.14QAPCh. 12.3 - Give the IUPAC name for each of the following:Ch. 12.3 - Give the IUPAC name for each of the following:Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the condensed structural formula for each of...Ch. 12.3 - Draw the line-angle structural formula for each of...Ch. 12.3 - Draw the line-angle structural formula for each of...Ch. 12.4 - Heptane, used as a solvent for rubber cement, has...Ch. 12.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 12.4 - Prob. 12.23QAPCh. 12.4 - Prob. 12.24QAPCh. 12.4 - Prob. 12.25QAPCh. 12.4 - Prob. 12.26QAPCh. 12.5 - Prob. 12.27QAPCh. 12.5 - Prob. 12.28QAPCh. 12.5 - Prob. 12.29QAPCh. 12.5 - Prob. 12.30QAPCh. 12.5 - Prob. 12.31QAPCh. 12.5 - Prob. 12.32QAPCh. 12.6 - 12.33 Give the IUPAC name for each of the...Ch. 12.6 - Prob. 12.34QAPCh. 12.6 - Prob. 12.35QAPCh. 12.6 - Prob. 12.36QAPCh. 12.6 - Prob. 12.37QAPCh. 12.6 - Prob. 12.38QAPCh. 12.7 - Prob. 12.39QAPCh. 12.7 - Prob. 12.40QAPCh. 12.7 - Prob. 12.41QAPCh. 12.7 - Prob. 12.42QAPCh. 12.7 - Prob. 12.43QAPCh. 12.7 - Prob. 12.44QAPCh. 12.7 - Prob. 12.45QAPCh. 12.7 - Prob. 12.46QAPCh. 12.8 - Prob. 12.47QAPCh. 12.8 - Give the IUPAC name and any common name for each...Ch. 12.8 - Prob. 12.49QAPCh. 12.8 - Prob. 12.50QAPCh. 12.8 - Prob. 12.51QAPCh. 12.8 - Prob. 12.52QAPCh. 12.8 - Prob. 12.53QAPCh. 12.8 - Prob. 12.54QAPCh. 12 - Prob. 12.55UTCCh. 12 - Prob. 12.56UTCCh. 12 - Prob. 12.57UTCCh. 12 - Prob. 12.58UTCCh. 12 - Prob. 12.59UTCCh. 12 - Prob. 12.60UTCCh. 12 - Prob. 12.61UTCCh. 12 - Prob. 12.62UTCCh. 12 - Give the number of carbon atoms and the types of...Ch. 12 - Prob. 12.64UTCCh. 12 - Prob. 12.65AQAPCh. 12 - Give the IUPAC name for each of the following:...Ch. 12 - Prob. 12.67AQAPCh. 12 - Prob. 12.68AQAPCh. 12 - Prob. 12.69AQAPCh. 12 - 12.70 Give the IUPAC name for each of the...Ch. 12 - Prob. 12.71AQAPCh. 12 - Prob. 12.72AQAPCh. 12 - Prob. 12.73AQAPCh. 12 - Prob. 12.74AQAPCh. 12 - Prob. 12.75AQAPCh. 12 - Prob. 12.76AQAPCh. 12 - Prob. 12.77AQAPCh. 12 - Prob. 12.78AQAPCh. 12 - Prob. 12.79AQAPCh. 12 - Prob. 12.80AQAPCh. 12 - Prob. 12.81AQAPCh. 12 - Prob. 12.82AQAPCh. 12 - Prob. 12.83AQAPCh. 12 - Prob. 12.84AQAPCh. 12 - Prob. 12.85AQAPCh. 12 - Prob. 12.86AQAPCh. 12 - Prob. 12.87AQAPCh. 12 - Prob. 12.88AQAPCh. 12 - Prob. 12.89CQCh. 12 - Prob. 12.90CQCh. 12 - Prob. 12.91CQCh. 12 - Prob. 12.92CQCh. 12 - Prob. 12.93CQCh. 12 - Prob. 12.94CQ
Knowledge Booster
Similar questions
- In an experiment, the viscosity of water was measured at different temperatures and the table was constructed from the data obtained. a) Calculate the activation energy of viscous flow (kJ/mol). b) Calculate the viscosity at 30°C. T/°C 0 20 40 60 80 η/cpoise 1,972 1,005 0,656 0,469 0,356arrow_forwardDon't used Ai solutionarrow_forwardLet's see if you caught the essentials of the animation. What is the valence value of carbon? a) 4 b) 2 c) 8 d) 6arrow_forward
- A laser emits a line at 632.8 nm. If the cavity is 12 cm long, how many modes oscillate in the cavity? How long does it take for the radiation to travel the entire cavity? What is the frequency difference between 2 consecutive modes?(refractive index of the medium n = 1).arrow_forwardA laser emits a line at 632.8 nm. If the cavity is 12 cm long, how many modes oscillate in the cavity? How long does it take for the radiation to travel the entire cavity? What is the frequency difference between 2 consecutive modes?(refractive index of the medium n = 1).arrow_forwardThe number of microstates corresponding to each macrostate is given by N. The dominant macrostate or configuration of a system is the macrostate with the greatest weight W. Are both statements correct?arrow_forward
- For the single step reaction: A + B → 2C + 25 kJ If the activation energy for this reaction is 35.8 kJ, sketch an energy vs. reaction coordinate diagram for this reaction. Be sure to label the following on your diagram: each of the axes, reactant compounds and product compounds, enthalpy of reaction, activation energy of the forward reaction with the correct value, activation energy of the backwards reaction with the correct value and the transition state. In the same sketch you drew, after the addition of a homogeneous catalyst, show how it would change the graph. Label any new line "catalyst" and label any new activation energy.arrow_forwardHow many grams of C are combined with 3.75 ✕ 1023 atoms of H in the compound C5H12?arrow_forwarde. f. CH3O. יון Br NaOCH3 OCH 3 Br H₂Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY