
Interpretation:
To determine the retrosynthetic analysis and synthesis of the following compounds.
Concept introduction:
Grignard reagents and organolithium compounds are strong bases. They will react with compounds containing hydrogen, attached to electronegative atoms like oxygen, nitrogen, and sulfur. They can be denoted as follows:
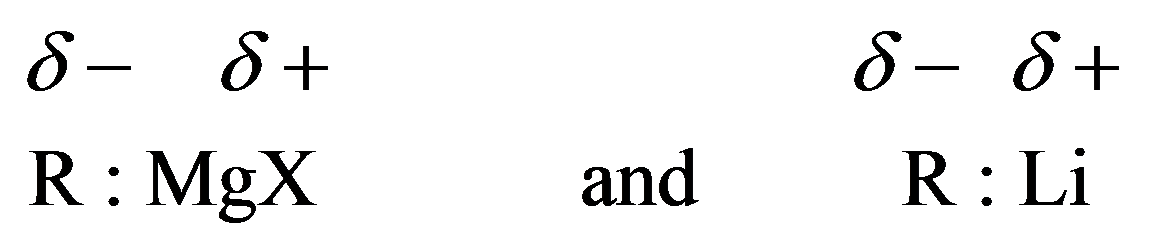
Grignard reagent reactions with carbonyl groups are treated as acid–base reactions. Grignard reagents are strong bases. When a reaction between a strong base and strong acid occurs, weak acid and weak base are formed. The side of the reaction that has the weak acid and base is favored.
Lithium aluminum hydride or sodium borohydride are used as reducing agents. Sodium borohydride is better for reducing
Retrosynthetic analysis is backtracking the reactants in a series of reactions. For synthesis of a compound using Grignard reagents, we have to select the correct reagents, ethers, aldehydes, ketones, or esters. There might be multiple ways to synthesize the alcohol and depending on the availability of reagents, we can choose a specific method.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
- Nonearrow_forward4. Draw and label all possible isomers for [M(py)3(DMSO)2(CI)] (py = pyridine, DMSO dimethylsulfoxide).arrow_forwardThe emission data in cps displayed in Table 1 is reported to two decimal places by the chemist. However, the instrument output is shown in Table 2. Table 2. Iron emission from ICP-AES Sample Blank Standard Emission, cps 579.503252562 9308340.13122 Unknown Sample 343.232365741 Did the chemist make the correct choice in how they choose to display the data up in Table 1? Choose the best explanation from the choices below. No. Since the instrument calculates 12 digits for all values, they should all be kept and not truncated. Doing so would eliminate significant information. No. Since the instrument calculates 5 decimal places for the standard, all of the values should be limited to the same number. The other decimal places are not significant for the blank and unknown sample. Yes. The way Saman made the standards was limited by the 250-mL volumetric flask. This glassware can report values to 2 decimal places, and this establishes our number of significant figures. Yes. Instrumental data…arrow_forward
- 7. Draw a curved arrow mechanism for the following reaction. HO cat. HCI OH in dioxane with 4A molecular sievesarrow_forwardTry: Convert the given 3D perspective structure to Newman projection about C2 - C3 bond (C2 carbon in the front). Also, show Newman projection of other possible staggered conformers and circle the most stable conformation. Use the template shown. F H3C Br Harrow_forwardNonearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

