
Concept explainers
Draw the organic products formed when allylic alcoholA is treated with each reagent.
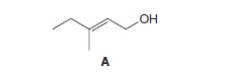
a.
b.
c.
d.
e.
f.
g. [1]
h.
(a)
Interpretation: The product formed when A is treated with
Concept introduction: The addition of
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
When A is treated with
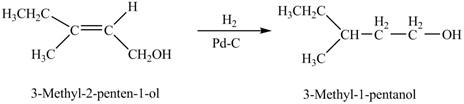
Figure 1
The product formed when A is treated with
(b)
Interpretation: The product formed when A is treated with
Concept introduction: In presence of peroxide alkene is oxidized to epoxide this is known as epoxidation. The weak pi bond of alkene and weak
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
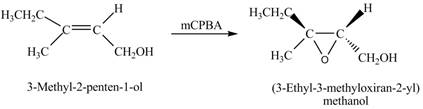
Figure 2
The product formed when A is treated with
(c)
Interpretation: The product formed when A is treated with
Concept introduction: Alcohols are oxidized to different carbonyl compounds depending upon the reagents and alcohol used. In presence of strong oxidizing reagents such as
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
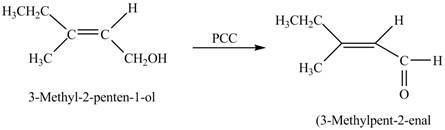
Figure 3
The product formed when A is treated with
(d)
Interpretation: The product formed when A is treated with
Concept introduction: Alcohols are oxidized to different carbonyl compounds depending upon the reagents and alcohol used. In the presence of strong oxidizing reagents such as
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
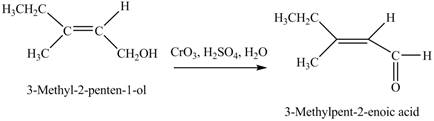
Figure 4
The product formed when A is treated with
(e)
Interpretation: The product formed when A is treated with Sharpless reagent
Concept introduction: Sharpless epoxidation involves the oxidation of double bond between carbon atoms to epoxide. This oxidation occurs only in allylic alcohol. This is an enantioselective oxidation, which means predominantly one enantiomer is formed. Sharpless reagents are
Answer to Problem 12.39P
The product formed when A is treated with Sharpless reagent
Explanation of Solution
There are two different chiral diethyl tartrate isomers,
When epoxidation is done using
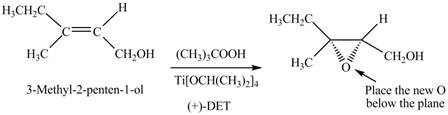
Figure 5
The product formed when A is treated with Sharpless reagent
(f)
Interpretation: The product formed when A is treated with Sharpless reagent
Concept introduction: Sharpless epoxidation involves the oxidation of double bond between carbon atoms to epoxide. This oxidation occurs only in allylic alcohol. This is an enantioselective oxidation, which means predominantly one enantiomer is formed. Sharpless reagents are
Answer to Problem 12.39P
The product of formed when A is treated with Sharpless reagent
Explanation of Solution
There are two different chiral diethyl tartrate isomers,
When epoxidation is done using
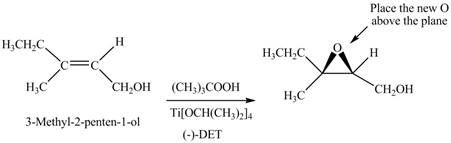
Figure 6
The product formed when A is treated with Sharpless reagent
(g)
Interpretation: The product formed when A is treated with given reagent is shown in Figure 7.
Concept introduction: Alcohols on treatment with phosphorus tribromide gives alkyl bromide. The
Answer to Problem 12.39P
The product formed in the given reaction is shown in Figure 7.
Explanation of Solution
The given reaction is,
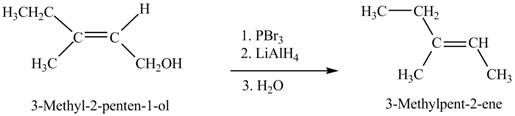
Figure 7
When the given alcohol is treated with
The product formed when A is treated with given reagent is shown in Figure 7.
(h)
Interpretation: The product formed when A is treated with
Concept introduction:
Answer to Problem 12.39P
The product formed when A is treated with
Explanation of Solution
In the given reaction, when A is treated with
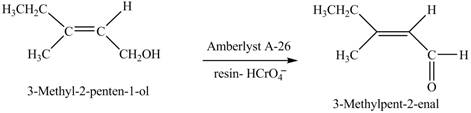
Figure 8
The product formed when A is treated with
Want to see more full solutions like this?
Chapter 12 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Human Physiology: An Integrated Approach (8th Edition)
Fundamentals Of Thermodynamics
Organic Chemistry
Genetics: From Genes to Genomes
Physics of Everyday Phenomena
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electrons-pushing arrows for the following reaction or mechanistic step(s).arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. I I I H Select to Add Arrows HCI, CH3CH2OHarrow_forwardCurved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and the follow the arrows to draw the intermediate and product in this reaction or mechanistic step(s).arrow_forward
- Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the intermediates and product of the following reaction or mechanistic step(s).arrow_forwardCurved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the arrows to draw the intermediate and the product in this reaction or mechanistic step(s).arrow_forwardLook at the following pairs of structures carefully to identify them as representing a) completely different compounds, b) compounds that are structural isomers of each other, c) compounds that are geometric isomers of each other, d) conformers of the same compound (part of structure rotated around a single bond) or e) the same structure.arrow_forward
- Given 10.0 g of NaOH, what volume of a 0.100 M solution of H2SO4 would be required to exactly react all the NaOH?arrow_forward3.50 g of Li are combined with 3.50 g of N2. What is the maximum mass of Li3N that can be produced? 6 Li + N2 ---> 2 Li3Narrow_forward3.50 g of Li are combined with 3.50 g of N2. What is the maximum mass of Li3N that can be produced? 6 Li + N2 ---> 2 Li3Narrow_forward
- Concentration Trial1 Concentration of iodide solution (mA) 255.8 Concentration of thiosulfate solution (mM) 47.0 Concentration of hydrogen peroxide solution (mM) 110.1 Temperature of iodide solution ('C) 25.0 Volume of iodide solution (1) used (mL) 10.0 Volume of thiosulfate solution (5:03) used (mL) Volume of DI water used (mL) Volume of hydrogen peroxide solution (H₂O₂) used (mL) 1.0 2.5 7.5 Time (s) 16.9 Dark blue Observations Initial concentration of iodide in reaction (mA) Initial concentration of thiosulfate in reaction (mA) Initial concentration of hydrogen peroxide in reaction (mA) Initial Rate (mA's)arrow_forwardDraw the condensed or line-angle structure for an alkene with the formula C5H10. Note: Avoid selecting cis-/trans- isomers in this exercise. Draw two additional condensed or line-angle structures for alkenes with the formula C5H10. Record the name of the isomers in Data Table 1. Repeat steps for 2 cyclic isomers of C5H10arrow_forwardExplain why the following names of the structures are incorrect. CH2CH3 CH3-C=CH-CH2-CH3 a. 2-ethyl-2-pentene CH3 | CH3-CH-CH2-CH=CH2 b. 2-methyl-4-pentenearrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

