
Chemistry: Atoms First
2nd Edition
ISBN: 9780073511184
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Question
Chapter 12, Problem 12.1KSP
Interpretation Introduction
Interpretation:
The compound(s) that are more soluble in water has to be predicted from the following options –
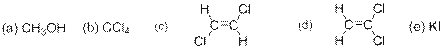
Concept Introduction:
Solubility of a substance depends on various factors like – chemical structure of the substance, temperature, pH etc,
Like dissolves like is a Universal rule which states that polar compounds dissolve more in polar solvents and non-polar compounds dissolve more in non-polar compounds.
Dipole moment is the measure of polarity in polar covalent compounds.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Is nucleophilic acyl substitution an SN1 or SN2 reaction?
Draw product A, indicating what type of reaction occurs.
NH2
F3C
CF3
NH
OMe
NH2-NH2, ACOH
A
Photochemical smog is formed in part by the action of light on nitrogen dioxide. The wavelength of radiation absorbed by NO2 in this reaction is 197 nm.(a) Draw the Lewis structure of NO2 and sketch its π molecular orbitals.(b) When 1.56 mJ of energy is absorbed by 3.0 L of air at 20 °C and 0.91 atm, all the NO2 molecules in this sample dissociate by the reaction shown. Assume that each absorbed photon leads to the dissociation (into NO and O) of one NO2 molecule. What is the proportion, in parts per million, of NO2 molecules in this sample? Assume that the sample behaves ideally.
Chapter 12 Solutions
Chemistry: Atoms First
Ch. 12.2 - Prob. 12.1WECh. 12.2 - Prob. 1PPACh. 12.2 - Prob. 1PPBCh. 12.2 - The diagram on the left depicts a system at room...Ch. 12.2 - Prob. 12.2.1SRCh. 12.2 - Prob. 12.2.2SRCh. 12.2 - Prob. 12.2.3SRCh. 12.2 - Prob. 12.2.4SRCh. 12.3 - Prob. 12.2WECh. 12.3 - When silver crystallizes, it forms face-centered...
Ch. 12.3 - The density of sodium metal is 0.971 g/cm3 and the...Ch. 12.3 - Prob. 12.3.1SRCh. 12.3 - Prob. 12.3.2SRCh. 12.4 - How many of each ion are contained within a unit...Ch. 12.4 - Referring to Figure 12.23, determine how many of...Ch. 12.4 - Referring to Figure 12.23, determine how many of...Ch. 12.4 - Prob. 3PPCCh. 12.4 - The edge length of the NaCl unit cell is 564 pm....Ch. 12.4 - Prob. 4PPACh. 12.4 - NiO also adopts the face-centered cubic...Ch. 12.4 - The metal iridium (Ir) crystallizes with a...Ch. 12.4 - Prob. 5PPACh. 12.4 - Copper crystallizes in a face-centered cubic...Ch. 12.4 - Given that the diameter and average mass of a...Ch. 12.5 - (a) Calculate the amount of heat deposited oil the...Ch. 12.5 - Calculate the amount of energy (in kilojoules)...Ch. 12.5 - Determine the final state and temperature of 100 g...Ch. 12.5 - Prob. 6PPCCh. 12.5 - Prob. 12.5.1SRCh. 12.5 - Prob. 12.5.2SRCh. 12.6 - Using the following phase diagram, (a) determine...Ch. 12.6 - Use the following phase diagram to (a) determine...Ch. 12.6 - Prob. 7PPBCh. 12.6 - Prob. 7PPCCh. 12.6 - Prob. 12.6.1SRCh. 12 - Explain why liquids, unlike gases, are virtually...Ch. 12 - What is surface tension? What is the relationship...Ch. 12 - Prob. 12.3QPCh. 12 - Prob. 12.4QPCh. 12 - Prob. 12.5QPCh. 12 - Prob. 12.6QPCh. 12 - Prob. 12.7QPCh. 12 - Why does the viscosity of a liquid decrease with...Ch. 12 - Why is ice less dense than water?Ch. 12 - Prob. 12.10QPCh. 12 - Prob. 12.11QPCh. 12 - Prob. 12.12QPCh. 12 - Prob. 12.13QPCh. 12 - Predict which of the following liquids has greater...Ch. 12 - Prob. 12.15QPCh. 12 - Vapor pressure measurements at several different...Ch. 12 - The vapor pressure of liquid X is lower than that...Ch. 12 - Prob. 12.18QPCh. 12 - Prob. 12.19QPCh. 12 - Define the following terms: crystalline solid,...Ch. 12 - Prob. 12.21QPCh. 12 - Classify the solid states in terms of crystal...Ch. 12 - Prob. 12.23QPCh. 12 - Define X-ray diffraction. What are the typical...Ch. 12 - Prob. 12.25QPCh. 12 - What is the coordination number of each sphere in...Ch. 12 - Calculate the number of spheres that would be...Ch. 12 - Prob. 12.28QPCh. 12 - Barium metal crystallizes in a body-centered cubic...Ch. 12 - Prob. 12.30QPCh. 12 - Europium crystallizes in a body-centered cubic...Ch. 12 - Crystalline silicon has a cubic structure. The...Ch. 12 - Prob. 12.33QPCh. 12 - Prob. 12.34QPCh. 12 - Prob. 12.35QPCh. 12 - Prob. 12.36QPCh. 12 - Shown here is a zinc oxide unit cell. What is the...Ch. 12 - Describe and give examples of the following types...Ch. 12 - Prob. 12.39QPCh. 12 - A solid is hard, brittle, and electrically...Ch. 12 - A solid is soft and has a low melting point (below...Ch. 12 - Prob. 12.42QPCh. 12 - Which of the following are molecular solids and...Ch. 12 - Prob. 12.44QPCh. 12 - Prob. 12.45QPCh. 12 - What is a phase change? Name all possible changes...Ch. 12 - What is the equilibrium vapor pressure of a...Ch. 12 - Use any one of the phase changes to explain what...Ch. 12 - Define the following terms: (a) molar heat of...Ch. 12 - Prob. 12.50QPCh. 12 - What can we learn about the intermolecular forces...Ch. 12 - The greater the molar heat of vaporization of a...Ch. 12 - Prob. 12.53QPCh. 12 - A closed container of liquid pentane (bp = 36.1C)...Ch. 12 - What is critical temperature? What is the...Ch. 12 - Prob. 12.56QPCh. 12 - How do the boiling points and melting points of...Ch. 12 - Prob. 12.58QPCh. 12 - Prob. 12.59QPCh. 12 - Prob. 12.60QPCh. 12 - Which of the following phase transitions gives off...Ch. 12 - Prob. 12.62QPCh. 12 - Prob. 12.63QPCh. 12 - Prob. 12.64QPCh. 12 - Prob. 12.65QPCh. 12 - Prob. 12.66QPCh. 12 - Prob. 12.67QPCh. 12 - How is the rate of evaporation of a liquid...Ch. 12 - Explain why steam at 100C causes more serious bums...Ch. 12 - The following compounds, listed with then- boiling...Ch. 12 - Prob. 12.71QPCh. 12 - Prob. 12.72QPCh. 12 - Explain how waters phase diagram differs from...Ch. 12 - The blades of ice skates are quite thin, so the...Ch. 12 - A length of wire is placed on top of a block of...Ch. 12 - Prob. 12.76QPCh. 12 - A phase diagram of water is shown. Label the...Ch. 12 - Prob. 12.78QPCh. 12 - Prob. 12.79QPCh. 12 - Prob. 12.80QPCh. 12 - Prob. 12.81QPCh. 12 - Prob. 12.82QPCh. 12 - The average distance between base pairs measured...Ch. 12 - A CO2 fire extinguisher is located on the outside...Ch. 12 - What is the vapor pressure of mercury at its...Ch. 12 - Prob. 12.86QPCh. 12 - The liquid-vapor boundary line in the phase...Ch. 12 - Prob. 12.88QPCh. 12 - Prob. 12.89QPCh. 12 - A student is given four solid samples labeled W,...Ch. 12 - Prob. 12.91QPCh. 12 - The diagram shows a kettle of boiling water....Ch. 12 - The south pole of Mars is covered with solid...Ch. 12 - The properties of gases, liquids, and solids...Ch. 12 - The standard enthalpy of formation of gaseous...Ch. 12 - Prob. 12.96QPCh. 12 - Under the same conditions of temperature and...Ch. 12 - The distance between Li+ and Cl is 257 pm in solid...Ch. 12 - Heat of hydration, that is, the heat change that...Ch. 12 - The fluorides of the second period elements and...Ch. 12 - Calculate the H for the following processes at...Ch. 12 - Prob. 12.102QPCh. 12 - Prob. 12.103QPCh. 12 - Ozone (O3) is a strong oxidizing agent that can...Ch. 12 - A sample of limestone (CaCO3) is heated in a...Ch. 12 - Carbon and silicon belong to Group 4A of the...Ch. 12 - Prob. 12.107QPCh. 12 - A 1.20-g sample of water is injected into an...Ch. 12 - What are the advantages of cooking the vegetable...Ch. 12 - A quantitative measure of how efficiently spheres...Ch. 12 - The phase diagram of helium is shown. Helium is...Ch. 12 - The phase diagram of sulfur is shown. (a) How many...Ch. 12 - Prob. 12.113QPCh. 12 - Argon crystallizes in the face-centered cubic...Ch. 12 - Given the phase diagram of carbon, answer the...Ch. 12 - Prob. 12.116QPCh. 12 - Swimming coaches sometimes suggest that a drop of...Ch. 12 - Prob. 12.118QPCh. 12 - Why do citrus growers spray their trees with water...Ch. 12 - Calcium metal crystallizes in a face-centered...Ch. 12 - A student heated a beaker of cold water (on a...Ch. 12 - The compound diclilorodifluoromethane (CCl2F2) has...Ch. 12 - Iron crystallizes in a body-centered cubic...Ch. 12 - Sketch the cooling curves of water from about 110C...Ch. 12 - Prob. 12.125QPCh. 12 - A sampleof water shows the following behavior as...Ch. 12 - A closed vessel of volume 9.6 L contains 2.0 g of...Ch. 12 - The electrical conductance of copper metal...Ch. 12 - Assuming ideal behavior, calculate the density of...Ch. 12 - Explain why drivers are advised to use motor oil...Ch. 12 - Prob. 12.131QPCh. 12 - Silicon used in computer chips must have an...Ch. 12 - Prob. 12.133QPCh. 12 - Prob. 12.1KSPCh. 12 - Prob. 12.2KSPCh. 12 - Prob. 12.3KSPCh. 12 - Prob. 12.4KSP
Knowledge Booster
Similar questions
- Correct each molecule in the drawing area below so that it has the skeletal ("line") structure it would have if it were dissolved in a 0.1 M aqueous solution of HCI. If there are no changes to be made, check the No changes box under the drawing area. No changes. HO Explanation Check NH, 2 W O :□ G ©2025 M unter Accessibilityarrow_forwardAn expression for the root mean square velocity, vrms, of a gas was derived. Using Maxwell’s velocity distribution, one can also calculate the mean velocity and the most probable velocity (mp) of a collection of molecules. The equations used for these two quantities are vmean=(8RT/πM)1/2 and vmp=(2RT/M)1/2 These values have a fixed relationship to each other.(a) Arrange these three quantities in order of increasing magnitude.(b) Show that the relative magnitudes are independent of the molar mass of the gas.(c) Use the smallest velocity as a reference for establishing the order of magnitude and determine the relationship between the larger and smaller values.arrow_forwardThe reaction of solid dimethylhydrazine, (CH3)2N2H2, and liquefied dinitrogen tetroxide, N2O4, has been investigated for use as rocket fuel. The reaction produces the gases carbon dioxide (CO2), nitrogen (N2), and water vapor (H2O), which are ejected in the exhaust gases. In a controlled experiment, solid dimethylhydrazine was reacted with excess dinitrogen tetroxide, and the gases were collected in a closed balloon until a pressure of 2.50 atm and a temperature of 400.0 K were reached.(a) What are the partial pressures of CO2, N2, and H2O?(b) When the CO2 is removed by chemical reaction, what are the partial pressures of the remaining gases?arrow_forward
- One liter of chlorine gas at 1 atm and 298 K reacts completely with 1.00 L of nitrogen gas and 2.00 L of oxygen gas at the same temperature and pressure. A single gaseous product is formed, which fills a 2.00 L flask at 1.00 atm and 298 K. Use this information to determine the following characteristics of the product:(a) its empirical formula;(b) its molecular formula;(c) the most favorable Lewis formula based on formal charge arguments (the central atom is N);(d) the shape of the molecule.arrow_forwardHow does the square root mean square velocity of gas molecules vary with temperature? Illustrate this relationship by plotting the square root mean square velocity of N2 molecules as a function of temperature from T=100 K to T=300 K.arrow_forwardDraw product B, indicating what type of reaction occurs. F3C CF3 NH2 Me O .N. + B OMearrow_forward
- Benzimidazole E. State its formula. sState the differences in the formula with other benzimidazoles.arrow_forwardDraw product A, indicating what type of reaction occurs. F3C CN CF3 K2CO3, DMSO, H₂O2 Aarrow_forward19) Which metal is most commonly used in galvanization to protect steel structures from oxidation? Lead a. b. Tin C. Nickel d. Zinc 20) The following molecule is an example of a: R₁ R2- -N-R3 a. Secondary amine b. Secondary amide c. Tertiary amine d. Tertiary amidearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning