
EBK THERMODYNAMICS: AN ENGINEERING APPR
9th Edition
ISBN: 8220106796979
Author: CENGEL
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1.11, Problem 72P
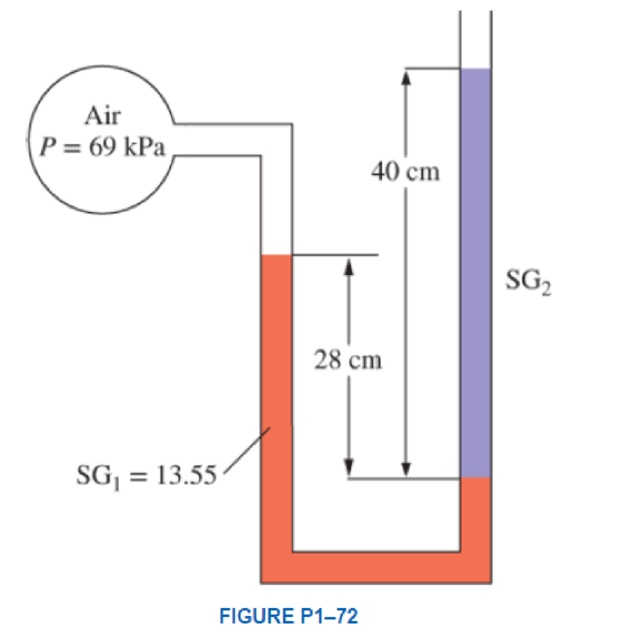
Consider a double-fluid manometer attached to an air pipe shown in Fig. P1–72. If the specific gravity of one fluid is 13.55, determine the specific gravity of the other fluid for the indicated absolute pressure of air. Take the atmospheric pressure to be 100 kPa. Answer: 1.59
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Determine which of the following flow fields represent a possible
incompressible flow?
(a) u= x²+2y+z; v=x-2y+z;w= -2xy + y² + 2z
a
(b) V=U cose
U coso 1 (9)
[1-9]
Usino |1 (4)]
[+]
V=-Usin 1+1
3. Determine the flow rate through the pipe line show in the figure in ft³/s,
and determine the pressures at A and C, in psi.
5'
B
C
12°
20'
D
6"d
2nd-
Water
A
5. A flow is field given by V = x²₁³+xy, and determine
3
·y³j-
(a) Whether this is a one, two- or three-dimensional flow
(b) Whether it is a possible incompressible flow
(c) Determine the acceleration of a fluid particle at the location (X,Y,Z)=(1,2,3)
(d) Whether the flow is rotational or irrotational flow?
Chapter 1 Solutions
EBK THERMODYNAMICS: AN ENGINEERING APPR
Ch. 1.11 - The value of the gravitational acceleration g...Ch. 1.11 - One of the most amusing things a person can...Ch. 1.11 - An office worker claims that a cup of cold coffee...Ch. 1.11 - What is the difference between the classical and...Ch. 1.11 - Explain why the light-year has the dimension of...Ch. 1.11 - What is the difference between pound-mass and...Ch. 1.11 - What is the net force acting on a car cruising at...Ch. 1.11 - What is the weight, in N, of an object with a mass...Ch. 1.11 - If the mass of an object is 10 lbm, what is its...Ch. 1.11 - The acceleration of high-speed aircraft is...
Ch. 1.11 - The value of the gravitational acceleration g...Ch. 1.11 - A 3-kg plastic tank that has a volume of 0.2 m3 is...Ch. 1.11 - A 2-kg rock is thrown upward with a force of 200 N...Ch. 1.11 - Solve Prob. 113 using appropriate software. Print...Ch. 1.11 - A 4-kW resistance heater in a water heater runs...Ch. 1.11 - A 150-lbm astronaut took his bathroom scale (a...Ch. 1.11 - The gas tank of a car is filled with a nozzle that...Ch. 1.11 - How would you define a system to determine the...Ch. 1.11 - A large fraction of the thermal energy generated...Ch. 1.11 - A can of soft drink at room temperature is put...Ch. 1.11 - How would you define a system to determine the...Ch. 1.11 - How would you describe the state of the air in the...Ch. 1.11 - What is the difference between intensive and...Ch. 1.11 - The specific weight of a system is defined as the...Ch. 1.11 - Is the number of moles of a substance contained in...Ch. 1.11 - Is the state of the air in an isolated room...Ch. 1.11 - What is a quasi-equilibrium process? What is its...Ch. 1.11 - Define the isothermal, isobaric, and isochoric...Ch. 1.11 - What is specific gravity? How is it related to...Ch. 1.11 - What are the ordinary and absolute temperature...Ch. 1.11 - Consider an alcohol and a mercury thermometer that...Ch. 1.11 - Consider two dosed systems A and B. System A...Ch. 1.11 - Consider a system whose temperature is 18C....Ch. 1.11 - Steam enters a heat exchanger at 300 K. What is...Ch. 1.11 - The temperature of a system rises by 130C during a...Ch. 1.11 - The temperature of a system drops by 45F during a...Ch. 1.11 - The temperature of the lubricating oil in an...Ch. 1.11 - Heated air is at 150C. What is the temperature of...Ch. 1.11 - What is the difference between gage pressure and...Ch. 1.11 - Explain why some people experience nose bleeding...Ch. 1.11 - A health magazine reported that physicians...Ch. 1.11 - Someone claims that the absolute pressure in a...Ch. 1.11 - Consider two identical fans, one at sea level and...Ch. 1.11 - The absolute pressure in a compressed air tank is...Ch. 1.11 - A manometer measures a pressure difference as 40...Ch. 1.11 - A vacuum gage connected to a chambee reads 35 kPa...Ch. 1.11 - The maximum safe air pressure of a tire is...Ch. 1.11 - A pressure gage connected to a tank reads 50 psi...Ch. 1.11 - A pressure gage connected to a tank reads 500 kPa...Ch. 1.11 - A 200-pound man has a total foot imprint area of...Ch. 1.11 - The gage pressure in a liquid at a depth of 3 m is...Ch. 1.11 - The absolute pressure in water at a depth of 9 m...Ch. 1.11 - Consider a 1.75-m-tall man standing vertically in...Ch. 1.11 - The barometer of a mountain hiker reads 750 mbars...Ch. 1.11 - The basic barometer can be used to measure the...Ch. 1.11 - A gas is contained in a vertical, frictionless...Ch. 1.11 - Reconsider Prob. 158. Using appropriate software,...Ch. 1.11 - The piston of a vertical piston-cylinder device...Ch. 1.11 - Both a gage and a manometer are attached to a gas...Ch. 1.11 - Reconsider Prob. 161. Using appropriate software,...Ch. 1.11 - A manometer containing oil ( = 850 kg/m3) is...Ch. 1.11 - A manometer is used to measure the air pressure in...Ch. 1.11 - A mercury manometer ( = 13.600 kg/m3) is connected...Ch. 1.11 - Repeat Prob. 165 for a differential mercury height...Ch. 1.11 - The pressure in a natural gas pipeline is measured...Ch. 1.11 - Repeat Prob. 167E by replacing air with oil with a...Ch. 1.11 - Blood pressure is usually measure by wrapping a...Ch. 1.11 - The maximum blood pressure in the upper arm of a...Ch. 1.11 - Consider a U-tube whose arms are open to the...Ch. 1.11 - Consider a double-fluid manometer attached to an...Ch. 1.11 - Calculate the absolute pressure. P1, of the...Ch. 1.11 - Consider the manometer in Fig. 173. If the...Ch. 1.11 - Consider the manometer in Fig. 173. If the...Ch. 1.11 - The hydraulic lift in a car repair shop has an...Ch. 1.11 - Consider the system shown in Fig. 177. If a change...Ch. 1.11 - The gage pressure of the air in the tank shown in...Ch. 1.11 - Repeat Prob. 178 for a gage pressure of 40 kPa.Ch. 1.11 - What is the value of the engineering software...Ch. 1.11 - Determine a positive real root of this equation...Ch. 1.11 - Solve this system of two equations with two...Ch. 1.11 - Solve this system of three equations with three...Ch. 1.11 - Solve this system of three equations with three...Ch. 1.11 - The reactive force developed by a jet engine to...Ch. 1.11 - The reactive force developed by a jet engine to...Ch. 1.11 - A man goes to a traditional market to buy a steak...Ch. 1.11 - What is the weight of a 1-kg substance in N, kN,...Ch. 1.11 - The pressure in a steam boiler is given to be 92...Ch. 1.11 - A hydraulic lift is to be used to lift a 1900-kg...Ch. 1.11 - The average atmosphere pressure on earth is...Ch. 1.11 - Hyperthermia of 5C (i.e., 5C rise above the normal...Ch. 1.11 - The boiling temperature of water decreases by...Ch. 1.11 - A house is losing heat at a rate of 1800 kJ/h per...Ch. 1.11 - The average body temperature of a person rises by...Ch. 1.11 - The average temperature of the atmosphere in the...Ch. 1.11 - A vertical, frictionless pistoncylinder device...Ch. 1.11 - A vertical pistoncylinder device contains a gas at...Ch. 1.11 - The force generated by a spring is given by F =...Ch. 1.11 - An air-conditioning system requires a 35-m-long...Ch. 1.11 - Balloons are often filled with helium gas because...Ch. 1.11 - Reconsider Prob. 1101. Using appropriate software,...Ch. 1.11 - Determine the maximum amount of load, in kg, the...Ch. 1.11 - The lower half of a 6-m-high cylindrical container...Ch. 1.11 - A pressure cooker cooks a lot faster than an...Ch. 1.11 - The pilot of an airplane reads the altitude 6400 m...Ch. 1.11 - A glass tube is attached to a water pipe, as shown...Ch. 1.11 - Consider a U-tube whose arms are open to the...Ch. 1.11 - A water pipe is connected to a double-U manometer...Ch. 1.11 - A gasoline line is connected to a pressure gage...Ch. 1.11 - Repeat Prob. 1110 for a pressure gage reading of...Ch. 1.11 - When measuring small pressure differences with a...Ch. 1.11 - Pressure transducers are commonly used to measure...Ch. 1.11 - Consider the flow of air through a wind turbine...Ch. 1.11 - The drag force exerted on a car by air depends on...Ch. 1.11 - It is well known that cold air feels much colder...Ch. 1.11 - Reconsider Prob. 1116E. Using appropriate...Ch. 1.11 - During a heating process, the temperature of an...Ch. 1.11 - An apple loses 3.6 kJ of heat as it cools per C...Ch. 1.11 - At sea level, the weight of 1 kg mass in SI units...Ch. 1.11 - Consider a fish swimming 5 m below the free...Ch. 1.11 - The atmospheric pressures at the top and the...Ch. 1.11 - Consider a 2.5-m-deep swimming pool. The pressure...
Additional Engineering Textbook Solutions
Find more solutions based on key concepts
A byte is made up of eight a. CPUs b. addresses c. variables d. bits
Starting Out with Java: From Control Structures through Objects (7th Edition) (What's New in Computer Science)
How is the hydrodynamic entry length defined for flow in a pipe? Is the entry length longer in laminar or turbu...
Fluid Mechanics: Fundamentals and Applications
Using your text editor, enter (that is, type in) the C++ program shown in Display 1.8. Be certain to type the f...
Problem Solving with C++ (10th Edition)
What is an uninitialized variable?
Starting Out with Programming Logic and Design (5th Edition) (What's New in Computer Science)
The solid steel shaft AC has a diameter of 25 mm and is supported by smooth bearings at D and E. It is coupled ...
Mechanics of Materials (10th Edition)
How does a computers main memory differ from its auxiliary memory?
Java: An Introduction to Problem Solving and Programming (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- Solve this problem and show all of the workarrow_forwardSolve this problem and show all of the workarrow_forwarddraw the pneumatic circuit to operate a double-acting cylinder with: 1. Extension: Any of two manual conditions plus cylinder fully retracted, → Extension has both meter-in and meter-out, 2. Retraction: one manual conditions plus cylinder fully extended, → Retraction is very fast using quick exhaust valve.arrow_forward
- Correct answer is written below. Detailed and complete solution with fbd only. I will upvote, thank you. Expert solution plsarrow_forwardCorrect answer is written below. Detailed and complete solution with fbd only. I will upvote, thank you.arrow_forwardCorrect answer is written below. Detailed and complete solution with fbd only. I will upvote, thank you.arrow_forward
- Correct answer is written below. Detailed and complete solution only with fbd. I will upvote, thank you.arrow_forwardCorrect answer is written below. Detailed and complete solution only. I will upvote, thank you.arrow_forwardCorrect answer is written below. Detailed and complete solution with fbd only. I will upvote, thank you.arrow_forward
- Correct answer is written below. Detailed and complete solution only. I will upvote, thank you.arrow_forwardCorrect answer is written below. Detailed and complete solution with fbd only. I will upvote, thank you.arrow_forwardCorrect answer is written below. Detailed and complete solution only. I will upvote, thank you.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
First Law of Thermodynamics, Basic Introduction - Internal Energy, Heat and Work - Chemistry; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=NyOYW07-L5g;License: Standard youtube license